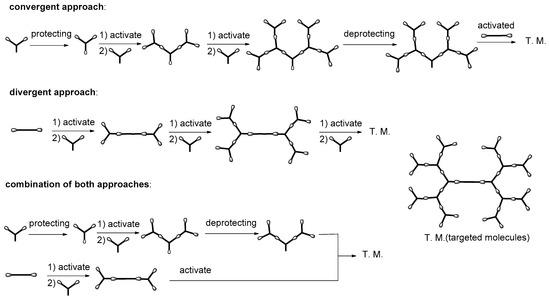
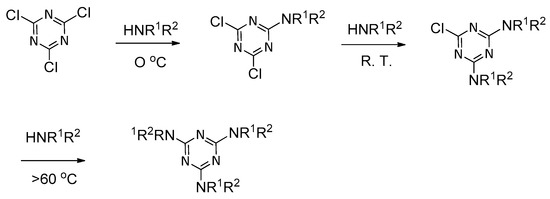
2.4. LC Dendrimers with the Loss of C2 Symmetry by Changing the Central Cores
Partially changing the peripheral group to break
C2-symmetry of molecules can lead dendrimers to contain isomers, and face-to-face π–π interactions between dendritic molecules are thus modulated to form the LC phase with thermal treatment. This strategy can also be applied to central linkers of dendrimers. With the peripheral group fixed by the dialkylamino units, the central linker is modulated as C
5H
10, C
4H
8, C
3H
6, and C
2H
4 (
Figure 5)
[63]. Dendrimers
9 and
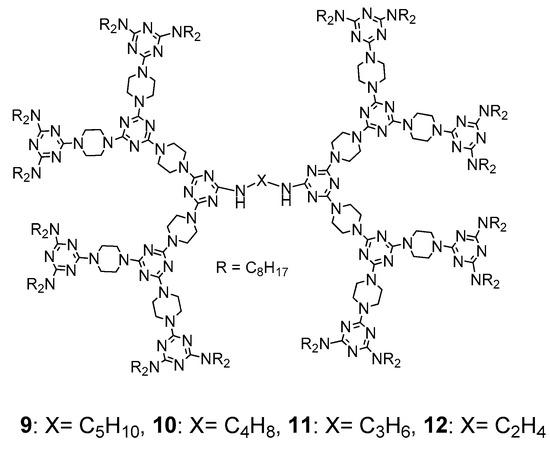
11, with the odd-number carbon linker in the dendritic core, are LCs. Dendrimer
9 was observed to exhibit a columnar phase from ~109 to ~168 °C on heating and from ~146 to ~87 °C on cooling. Dendrimer
11 showed a columnar phase between ~107 and ~144 °C on heating and between ~115 and ~81 °C on cooling. Dendrimers
10 and
12 with the even-number carbon linker in the dendritic core are not liquid crystalline. The solid-phase-to-isotropic-phase (solid-to-iso) transition temperatures for dendrimers
10 and
12 are ~132 and ~147 °C, respectively, on heating, and the isotropic-phase-to-solid-phase (iso-to-solid) transition temperatures for
10 and
12 are ~100 and ~90 °C, respectively, on cooling.
Figure 5. Structures of dendrimers (9–12).
Molecular weights for dendrimers
9–
12 are similar: ~6050, ~6036, ~6022, and ~6008, respectively. A computer simulation for molecular conformations of dendrimers
9 and
10 showed that
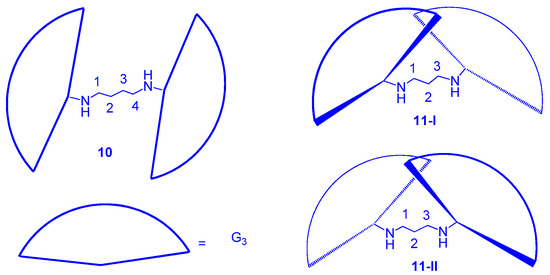
9 is asymmetrical and
10 is symmetrical along with the central linkers, and a diagram can show their differences
[63]. As shown in
Figure 6, dendrimer
10, with a 4-carbon linker in the dendritic core, allows the dendronic halves to be located at the
anti-position of the C
4H
8 linker to reduce repulsions between Hs (at C
2 and C
3) and the dendronic halves. Therefore, the molecular conformation of
10 is almost symmetrical. Accordingly, dendrimer
11 with a 3-carbon linker leads both the dendronic halves to exist in the
syn-position of the C
3H
6 linker to reduce corresponding repulsions; in this conformation, two isomers of dendrimer
11 should exist since the left dendronic half may be on the top or bottom of the right dendronic half (
Figure 6). Because of the isomeric effect, the intermolecular face-to-face π–π interactions are reduced in the solid stacking, and the columnar phases of dendrimers
9 and
11 are thus formed on thermal treatment. To further understand the isomeric effect on influencing their stackings in LC state, their molecular volumes (
Vm) and cell volumes (
Vcell) were calculated on the basis of powder-XRD data of dendrimers
9 and
11 [63]. Since the calculated volumes are temperature-dependent, and the XRD data for
9 and
11 were collected at different temperatures, the
Vm/
Vcell ratios, instead of
Vm or
Vcell, were compared to remove the temperature factor. The
Vm/
Vcell ratios of
9 and
11 were calculated to be ~1.03 and ~0.95, respectively. For the non-mesogenic compounds
10 and
12, crystalline XRD data were also used for calculation, assuming that both formed the columnar hexagonal crystalline phase; the
Vm/
Vcell ratios of
10 and
12 were estimated to be ~0.92 and ~0.87, respectively.
Figure 6. Isomers of dendrimers (10) and (11).
The ratios of Vm/Vcell for 9 and 11 are larger than those for 10 and 12, indicating that the stackings of 9 and 11 in the solid state are somewhat looser than those of 10 and 12. This further indicates that the isomeric effect on dendrimers can probably modulate the face-to-face π–π interactions between dendritic molecules.
As indicated previously, the repulsion between dendronic halves and the Hs (at C
2 or C
3) significantly influences the conformation of dendrimer
10. If the central linker of
10 was methylated on nitrogen, then the repulsion between Me and the Hs (at C
2 or C
3) should increase, although the repulsion should be smaller than that between the dendronic halves and the corresponding Hs (at C
2 or C
3). However, this should convert several of the dendronic halves at the
anti-position of C
4H
8 linker to the
syn-position. Specifically, dendrimer
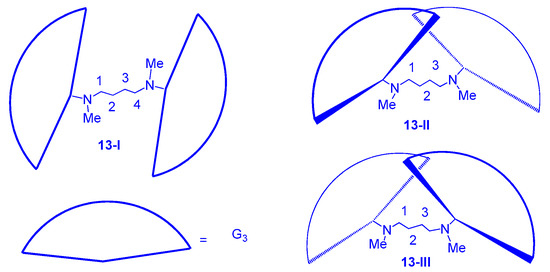
13 should have three isomers in the solid state, as shown in
Figure 7 [64], although most of the
13 exists as the form of isomer
13-I and, as expected, dendrimer
13 exhibited a columnar phase on thermal treatment (
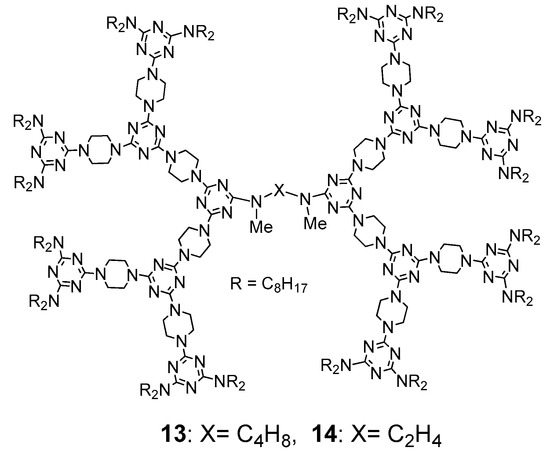
Figure 8). The mesogenic range is from ~111 to ~123 °C on heating and from ~115 to ~94 °C on cooling. Accordingly, dendrimer
14 has three isomers in the solid state but showed only a columnar phase between ~94 and ~72 °C on cooling without showing any mesogenic behavior on heating.
Figure 7. Isomers of dendrimers (13-I), (13-II), and (13-III).
Figure 8. Structures of dendrimers (13) and (14).
The solid-to-iso transition temperatures of dendrimers 10 and 12 on heating are ~132 and ~147 °C, respectively, which are higher than those of corresponding dendrimers 13 and 14 (~118 and 123 °C, respectively). This indicates that methylation of nitrogen in the central linker affects their molecular morphology and somewhat loosens their solid stacking. Based on the powder-XRD data of dendrimers 13 and 14, their ratios of Vm/Vcell were also calculated. The Vm/Vcell ratio of 13 was ~0.92, similar to that of 10 (~0.92) and the Vm/Vcell ratio of 14 was ~0.80, which is smaller to that of 12 (~0.87).
Although methylation of nitrogens in the central linker of dendrimers converts non-mesogenic
10 and
12 to become mesogenic
13 and
14, respectively, the isomeric effect seems to not be significant in reducing intermolecular face-to-face π–π interactions, allowing dendrimers
13 and
14 to be looser in the solid stacking than dendrimers
10 and
12. Based on the optimal conformation of
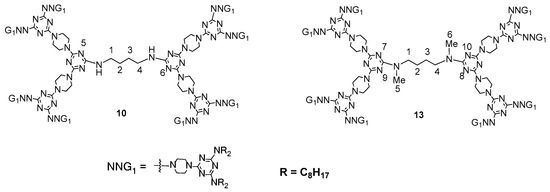
13 [64], it was discovered that four intramolecular H-bond interactions arise between N
a (a = 7, 8, 9, 10) and H (at C
b; b = 1, 4, 5, 6), respectively, as shown in
Figure 9. Accordingly, only two intramolecular H-bond interactions arise between N
a (a = 5, 6) and H (at C
b; b = 1, 4,) in dendrimer
10. Therefore, dendrimer
13 that mostly exists in the form of
13-I should be more rigid and is less distorted than dendrimer
10, and therefore the void space inside the solid stacking of
10 should be larger than that inside the dendritic framework of
13-I. However, because of the isomeric effect, the face-to-face π–π interactions between dendritic molecules of
13 is reduced and therefore its dendritic framework is fluffier. As a result, dendrimers
10 and
13 have almost the same
Vm/
Vcell ratio (~0.92) on the basis of balancing the two conflicting factors. When the central linker is shortened, congestion from dendritic halves of
12 is more vigorous and the distortion allows dendrimer
12 to become more porous. Therefore, the
Vm/
Vcell ratio of
12 (~0.87) is larger than that of
14 (~0.80).
Figure 9. The numbering on carbons for indicating intramolecular H-bond interactions between H and N of dendrimers (10) and (13).
The central linkers in triazine-based dendrimers significantly influence their morphology and consequently affect the formation of the mesophase during the thermal process. Instead of N-alkylation of an even-number carbon central linker, this strategy was also undertaken to an odd-number carbon central linker. As dendrimer
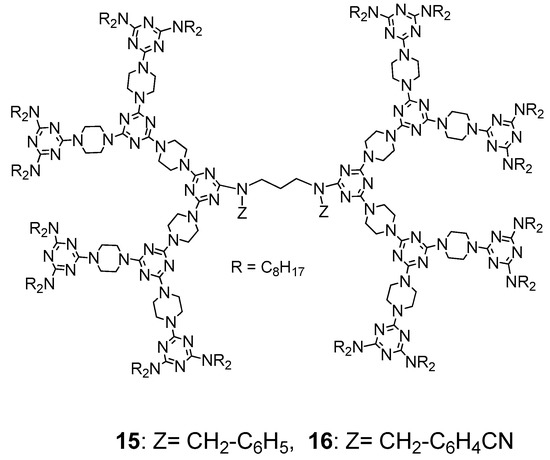
11 has already been observed to exhibit a columnar phase on thermal treatment, a strong CN polar group was incorporated in the central linker to investigate its influence on the mesogenic behavior. Dendrimers
15 and
16 were therefore further prepared for studying their LC properties (
Figure 10)
[65]. Dendrimer
15 exhibited a columnar phase between ~102 and ~118 °C on heating and between ~115 and ~82 °C on cooling. Dendrimer
16 showed a columnar phase from ~88 to ~144 °C on heating and from ~138 to ~68 °C on cooling. Compared with the DSC behaviors of
11, the solid-to-iso transition temperature of
15 (~118 °C) was lower than that of
11 (~144 °C) on heating, indicating that the alkylation on N in the central linker increased the steric congestion of triazine-based dendrimer and somewhat loosened their solid stacking as shown in the study of dendrimers
10,
12,
13 and
14. The solid-to-iso transition temperature of
16 (~144 °C) was similar to that of
11 (~144 °C), and the mesogenic range of
16 on cooling (from ~138 to ~68 °C) was broader than that of
11 (from ~115 to ~81 °C). The CN group in the central linker not only lowered the solidifying temperature but also raised the isotropic-phase-to-columnar-mesophase (iso-to-col) transition temperature on cooling. In principle, the steric congestion from benzyl moiety in the central linker of dendrimers
15 should not differ from and 4-cyanobenzyl moiety in
16, but their mesogenic behaviors on thermal treatment are significantly different. The strong polarity of the CN group strengthens intermolecular interactions between face-to-face dendritic molecules. For further understanding, the
Vm/
Vcell ratios of
15 and
16 were estimated on the basis of their powder-XRD data as ~0.80 and ~0.98, respectively. The
Vm/
Vcell ratio of dendrimer
16 was larger than that of
11 (~0.95), but that of dendrimer
15 was smaller, indicating that the loose order of their stacking is
16 >
11 >
15. As dendrimers
11,
15, and
16 all show a columnar phase in the mesogenic range, the face-to-face distances between two dendritic molecules (
dslice) for
11,
15, and
16 can be obtained from the powder-XRD data in a similar manner and are calculated to be 13.14, 13.28, and 13.68 Å, respectively. The
Vm/
Vcell ratios and
dslice distances show that the strong polarity of CN group significantly increased the face-to-face distance between dendritic molecules within columns. This reasonably explains that the col-to-solid transition temperature on cooling of
16 is lower than those of
11 and
15. However, as indicated previously, the solid-to-iso transition temperature of
16 (~144 °C) is similar to that of
11 (~144 °C) on heating, which can also be ascribed to the strong polarity of the CN group. During the heating process, the strong intermolecular polar-to-polar interactions within columns led dendrimer
16 to have the same solid-to-iso transition temperature as
11, although the stacking of
16 was looser. Neither dendrimer
11 nor
15 contains any functional group in the central linker, but the
dslice distance of
15 within columns is 13.28 Å, only slightly larger than that of
11 (13.14 Å). The more void space between dendritic molecules of
15 is occupied by the benzyl group, which results in the lower
Vm/
Vcell ratio of
15 (0.80) versus that of
11 (0.95). Since the
dslice distances of
11 and
15 do not differ from each other, the mesophase ranges of both dendrimer are also similar to each other.
Figure 10. Structures of dendrimers (15) and (16).