1000/1000
Hot
Most Recent

Unlike in normal epithelium, dysregulated overactivation of various proteases have been reported in cancers. Degradation of pericancerous extracellular matrix leading to cancer cell invasion by matrix metalloproteases is well known evidence. On the other hand, several cell-surface proteases, including type II transmembrane serine proteases (TTSPs), also induce progression through activation of growth factors, protease activating receptors and other proteases. Hepatocyte growth factor (HGF) known as a multifunctional growth factor that upregulates cancer cell motility, invasiveness, proliferative, and anti-apoptotic activities through phosphorylation of MET (a specific receptor of HGF). HGF secreted as inactive zymogen (pro-HGF) from cancer associated stromal fibroblasts, and the proteolytic activation by several TTSPs including matriptase and hepsin is required. The activation is strictly regulated by HGF activator inhibitors (HAIs) in physiological condition. However, downregulation is frequently observed in cancers. Indeed, overactivation of MET by upregulation of matriptase and hepsin accompanied by the downregulation of HAIs in urological cancers (prostate cancer, renal cell carcinoma, and bladder cancer) are also reported, a phenomenon observed in cancer cells with malignant phenotype, and correlated with poor prognosis.
MET, encoded by Met proto-oncogene located on chromosome 7q31, is a tyrosine kinase-type specific receptor of HGF, which forms disulfide-inked heterodimer consisting of an extracellular alpha chain and single-pass transmembrane beta chain [1][2][3][4]. As shown in Figure 1, the intracellular domain of the beta chain comprises a juxtamembrane domain and catalytic kinase domain containing an activation loop and carboxy-terminal multifunctional docking site. The juxtamembrane domain downregulates the kinase activity by phosphorylation of Ser975, while the catalytic kinase domain upregulates the activity by phosphorylation of Tyr1234 and Tyr1235. The multifunctional docking sites contain Tyr1349 and Tyr1356, which lead to downstream signaling through several intracellular adaptor proteins [1][2][3][4][5]. Increased expression of MET with worse prognosis has been reported in various cancer cells, and phosphorylation (activation) potently promotes invasion and metastasis [5][6][7][8]. Activation of HGF/MET signaling axis in cancer cells also plays a significant role in proliferation, angiogenesis, epithelial-mesenchymal transition (EMT), and drug resistance [1][2][3]. Activation is introduced by: 1) ligand (HGF)-dependent activation, 2) reciprocal activation by overexpression-induced MET oligomerization, 3) activating point mutation of tyrosine kinase domain, and 4) transactivation by heterodimerization with another receptor tyrosine kinase [1][2][3]. In the ligand-dependent activation, proteolytic activation of pro-HGF is necessary. As mentioned above, two major activating protease families were reported: 1) a serum serine protease, HGFA; and 2) type II transmembrane serine proteases (TTSPs) such as matriptase, hepsin, and transmembrane protease/serine (TMPRSS) 2 [1][9][10][11]. Although these pro-HGF activating proteases are tightly regulated by two transmembrane serine protease inhibitors, HAI-1 and HAI-2, downregulation of HAIs has been observed in several cancers and has been shown to induce progression [10][11].
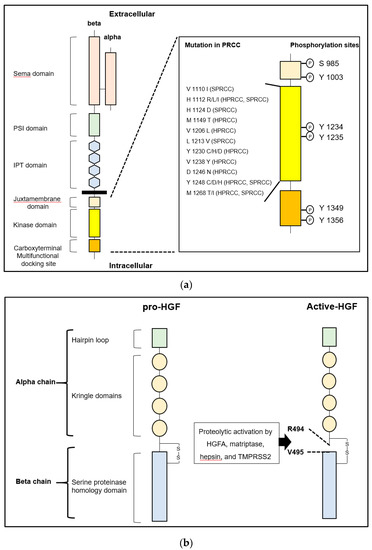
Figure 1. (a) Left: The structure of human MET is shown. MET consists of extracellular alpha and single-pass transmembrane beta chain, which are disulfide-linked heterodimer. The beta chain is composed of six major domains including Sema (semaphorin), PSI (plexin, semaphorin, integrin), IPT (immunoglobulin-like regions in plexins and transcription factors), juxtamembrane, tyrosine kinase domain, and multifunctional docking site. Right: Sites of point mutation in hereditary and sporadic papillary renal cell carcinoma (HPRCC and SPRCC) and conventional phosphorylation sites in intracellular domains are shown. (b) Left: The structure of human pro-hepatocyte growth factor (HGF) is shown. HGF consists of four Kringle domains and a serine proteinase homology domain. Right: The active form of HGF is shown. HGFA, hepsin, matriptase, and TMPRSS2 proteolytically cleave between Arg 494 and Val 495 to convert to a two-chain heterodimeric active form. One-letter abbreviation of amino acids is used.
The TTSP family in humans consists of 17 serine proteases [1][6][9]. The structures are specified as a single-pass hydrophobic transmembrane domain near the N-terminus with a short intracellular domain and a large extracellular portion including a carboxy-terminal serine protease domain [1][6][9]. All TTSPs are divided into the four subfamilies of hepsin, matriptase, human airway trypsin-like protease (HAT) and corin (Table 1) [1][6][9]. All TTSPs belong to the S1 peptidase family (noted in MEROPS as clan PA, family S1), and a catalytic triad consists of serine, aspartate, and histidine residues, as shown in Figure 2 [12][13]. Hepsin, matriptase and TMPRSS2 shows a strong cleavage preference for substrate with arginine in the P1 position [12][13]. In urogenital cancers, the expression of matriptase, hepsin and TMPRSS2 has been reported (Figure 2). Therefore, we focused on these TTSPs in this review.
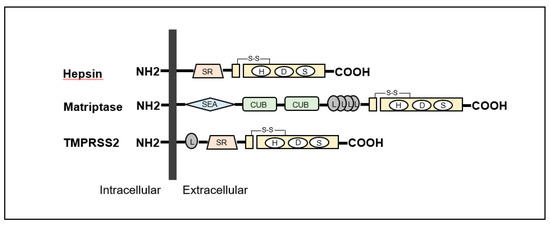
Figure 2. Structures of hepsin, matriptase and transmembrane protease serine (TMPRSS) 2 are shown. All type II transmembrane serine proteases (TTSPs) show single-pass transmembrane proteins with intracellular NH2-terminus and extracellular carboxy-terminal serine protease domains. Hepsin is composed of scavenger receptor (SR) and serine protease domains. Matriptase contains sea urchin sperm protein/enteropeptidase/agrin (SEA) domain, Cls/Cls, urchin embryonic growth factor, bone morphologic protein-1 (CUB) domain, four low-density lipoprotein receptor (L) domains class A and serine protease domains. TMPRSS2 is consists of an L domain, SR, and serine protease domains.
Table 1. Type II transmembrane serine protease (TTSP) family content.
| Subfamily | Protease |
|---|---|
| HAT/DESC | HAT |
| DESC1 | |
| TMPRSS 11A | |
| HAT-like 4 | |
| HAT-like 5 | |
| Hepsin/TMPRSS | Hepsin (TMPRSS1) |
| TMPRSS 2 | |
| TMPRSS 3 | |
| TMPRSS 4 | |
| TMPRSS 13 | |
| Enteropeptidase | |
| Spinesin | |
| Matriptase | Matriptase |
| Matriptase 2 | |
| Matriptase 3 | |
| Polyserase | |
| Corin | Corin |
Matriptase (MT-ST1, ST14) gene is located on human chromosome 11q24-25, and 855 amino acids are encoded in the gene [1][6][7][9]. The molecular weight of matriptase is 80–90-kda. Matriptase was first discovered in breast cancer cell line (T-47D) and purified from human milk [1][6][7][9]. It is expressed in human epithelial cells of various organs to maintain the formation of epithelial barrier formation [1][8][9][10][11]. In addition, the major enzymatic functions are reported as follows: 1) activation of hepatocyte growth factor zymogen (pro-HGF), pro-platelet-derived growth factor (PDGF)-C, -D, and pro-macrophage stimulating protein (MSP); 2) activation of protease-activated receptor (PAR)-2; 3) activation of urokinase-type plasminogen activator; 4) degradation of extracellular matrix; and 5) activation of prostasin, which is a glycosylphosphatidylinositol (GPI)-anchored protease known to activate epithelial sodium channel (ENaC) [1][6][7][9]. Among TTSPs, matriptase has been reported as the most efficient activator of pro-HGF [1][9][10][11]. HAIs are major regulators of matriptase, and deregulation of matriptase activity facilitates cancer progression [1][9][10][11]. Indeed, matriptase expression is reported to be upregulated in various cancers (breast, ovarian, uterine, colon, cervical, skin, pancreatic, esophageal, head and neck, prostate, bladder and renal cell carcinoma: RCC) with poor prognosis [1][9][10][11][14].
Hepsin (HPN, TMPRSS1) gene is located on human chromosome 19q13.11, and 417 amino acids are encoded [1][6][7][9]. The molecular weight of hepsin protein is 45-kda. Although mRNA is highly expressed in liver and kidney, ubiquitous expression of the protein is reported [1][9][15]. The functions are reported as follows: 1) activation of pro-HGF; 2) activation of pro-MSP; 3) activation of pro-urokinase-type plasminogen activator; and 4) cleavage of laminin-332 [16]. Similar to matriptase, the catalytic activities of hepsin are regulated by HAI-1 and HAI-2 [1][9][10][11]. In cancer, overexpression of hepsin mRNA is reported in prostate, ovary, kidney, and breast [1][6][7][9]. Increased expression of the protein is also reported in prostate, ovarian, breast, and endometrial cancer [1][6][7][9].
HAI-1 (SPINT-1) gene is located at 15q 15.1 and HAI-2 (SPINT-2) is located at 19q 13.2 [1][10][11]. Both proteins were initially identified in conditioned medium of human gastric cancer cell line MKN45 [1][10][11[17][18]. HAI-2 was also purified as placental bikunin from placenta [1][10][11][19]. The proteins have two specific extracellular Kunitz-type serine protease inhibitor domains, (KD)-1 and KD-2, except for a splicing variant of HAI-2 (isoform B has single KD) (Figure 3), which can inhibit several trypsin-like serine proteases, including all pro-HGF-activating enzymes [1][10][11][20][21]. Whereas, HAIs were initially discovered as HGFA inhibitors, they also inhibit matriptase and hepsin [1][10][11]. In addition, HAIs are required for intracellular transport and cell surface localization of matriptase in several types of cells [1][9][10][11]. HAI-1 is reported to express in the majority of normal epithelial cells [1][10][11][22]. In physiological condition, HAI-1 maintains epithelial integrity through regulation of matriptase activity [1][10][11][22]. HAI-1 is also required for placental differentiation, embryonic development and postnatal survival [10][11][23]. However, it has been reported that insufficient expression revealed dysregulation of pro-HGF activating enzymes in various cancers leading to progression [10][11]. Indeed, decreased expression of HAI-1 induced carcinogenesis (skin, intestine) and progression with worse prognosis (gastrointestinal, breast, ovarian, endometrial cancers and RCC) [10][11][24][25][26][27][28][29][30][31][32][33][34]. In addition, HAI-1 is also known as a suppressor of epithelial mesenchymal transition (EMT) [35].
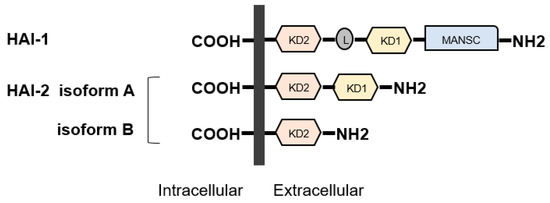
Figure 3. Structures of hepatocyte growth factor activator inhibitor (HAI)-1 and HAI-2 are shown. HAIs show single-pass transmembrane protein with intracellular carboxy-terminus and extracellular specific protease inhibitor domains, the so-called Kunitz domain (KD). HAI-1 is composed of two KDs, L domain, and motif at N terminus with seven cysteines (MANSC) domains. There are two isoforms in HAI-2. Similar to HAI1, HAI-2 isoform A has two KDs, whereas isoform B has a single KD.
HAI-2 is ubiquitously expressed in normal cells, including epithelial, mesenchymal, blood cells and trophoblasts [11]. HAI-2 is reported to maintain the integrity of intestinal epithelium through regulation of matriptase-induced epithelial cell adhesion molecule (EpCAM) cleavage [36]. Downregulation by hypermethylation of SPINT2 gene has been reported in several cancers, including hepatocellular carcinoma, RCC, melanoma, gastric carcinoma, and esophageal squamous cell carcinoma [37][38][39][40]. Expression of HAI-2 is also decreased in PC. However, no apparent SPINT2 promoter methylation has been observed in either clinical samples or cell lines [41]. In this report, the authors suggest that posttranslational regulation of HAI-2 expression is essential in prostate cancer. The regulatory role of HAI-2 in the activation of pro-HGF by inhibiting the activating proteases (including matriptase), which induces HGF/MET signaling axis, has been considered a major suppressive function in cancer progression [10][11]. Additionally, an alternative function such as the activation of caspase 3 in esophageal squamous cell carcinoma leading to the promotion of apoptosis and inhibition of proliferation was also reported [39][42]. However, HAI-2 has also been reported to be required for invasive growth in oral squamous cell carcinoma, which suggests that the role of HAI-2 may be tissue or cell-type specific and dependent on targeting TTSPs [43].