2.1. Cu(I/II) Distributions in the Nervous System
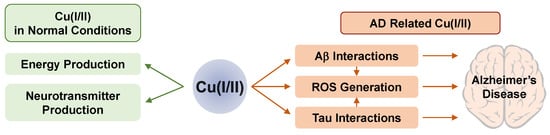
The Cu ion is the third most abundant transition metal ion (ca. 100 mg) in the human body
[20]. It works as a cofactor that binds to various metalloenzymes and assists their activation
[4][21]. Since it usually exists as cuprous ions [Cu(I)] and cupric ions [Cu(II)], Cu(I/II) can serve as an electron transporter. Cu(I) has an electron configuration of [Ar]3d
10, and Cu(II) has [Ar]3d
9 [20][22]. In addition, Cu(I/II) plays a key role in energy metabolism, signal transduction, reproduction, and development that are very important for physiological functions. For example, cytochrome c oxidase in mitochondria needs Cu ions for its activation, and dopamine-β-hydroxylase utilizes it for the cellular secretory pathway
[23][24][25][26]. Cu ions can be detected in various points of the brain such as the soma of cortical pyramidal and cerebellar granular neurons, neuropil within the cerebral cortex, hippocampus, cerebellum, and spinal cord
[27]. On average, ca. 100 μM of Cu ions has been detected in the brain; however, some parts of the brain have a 2- to 3-fold higher concentration than the other regions
[28]. In particular, the ceruleus has a 1.3 mM concentration, which is a part of the brain related to stress and panic. The substania nigra, the dopamine-producing region of the brain, also has a high Cu ion concentration (ca. 400 μM)
[28].
The extracellular level of Cu ions depends on the cellular environment. In cerebrospinal fluid (CSF), only 0.5–2.5 μM of Cu ions exists, while the synaptic cleft contains 30 μM
[29][30][31]. Usually, the Cu ion concentration is 2–3-fold higher in neurons
[32]. In the brain, Cu ions exist in two types: (i) those tightly bound to the proteins, or (ii) those in labile pools
[33]. Several regions of the brain such as the soma of cortical pyramidal and cerebellar granular neurons, the hippocampus, the cerebellum, and the spinal cord have labile copper stores
[27]. There are also labile pools with a low concentration in the extracellular regions
[34].
2.5. Regulators of Cu(I/II)
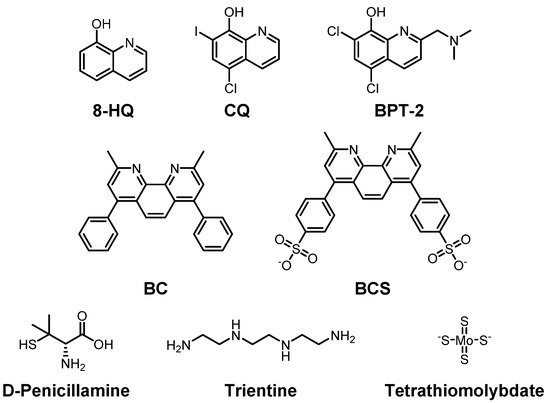
To reduce the risk of onset and/or progression of AD by Cu(I/II), various chemical agents have been developed to regulate the levels of the metal ions (Figure 2). The chemicals were examined by their ability to bind to Cu(I/II) in numerous studies. In this section, we summarize the chemical agents capable of interacting with Cu(I/II).
Figure 2. Selected Cu(I/II)-targeting molecules developed to maintain Cu(I/II) homeostasis. Top row: 8-hydroxyquinoline (8-HQ) and its derivatives, clioquinol (CQ), 5-chloro-7-iodoquinolin-8-ol, and BPT-2, 5,7-dichloro-2-[(dimethylamino)methyl]quinolin-8-ol; middle row: bathocuproine (BC), 2,9-dimethyl-4,7-diphenyl-1,10-phenanthroline, and bathocuproine disulfonate (BCS), 4,40-(2,9-dimethyl-1,10-phenanthroline-4,7-diyl)dibenzenesulfonate; bottom row: D-penicillamine, (2S)-2-amino-3-methyl-3-sulfanylbutanoic acid, trientine, triethylenetetramine, and tetramolybdate.
Clioquinol (
CQ), an 8-hydroxyquinolin (
8-HQ) derivative, is a candidate for treatment of AD by targeting Cu(II) and Aβ (
Figure 2; top row)
[137][138][139]. The
CQ–Cu(II) complex has a square planar structure where two
CQs coordinate Cu(II) in a crystal structure. In the solution phase, its symmetry is broken, and it becomes a tetragonally distorted structure
[140]. Regardless of this, the structure in solution is still closely related to that observed in crystallography. It has nanomolar affinity for Cu(II) and can cross the BBB
[141]. In 2003, Prana Biotechnology reported the results of a phase II trial on
CQ in AD patients due to its abilities of metal chelation and modulation of metal-free and metal-bound Aβ aggregation. However, further processes were stopped during the phase II/III trial since a toxic compound was detected during the manufacturing steps. Although it showed toxicity,
CQ has recently been examined for its potential neuroprotective effect in
C. elegans [142].
PBT-2 is also an
8-HQ derivative and the next generation of
CQ developed for treatment of AD and Huntington’s disease (
Figure 2; top row)
[143][144]. It has improved BBB penetrance and pharmacokinetics compared to
CQ [145]. Interactions between Cu ions and Aβ are disrupted by
PBT-2, preventing the accumulation of toxic Aβ species in the brain. The extracellular Cu ion concentration is reduced since
PBT-2 transports Cu ions to the intracellular space. This effectively reduces the chance for Cu–Aβ interactions which could trigger Aβ aggregation
[146][147]. Prana started the first phase II trial of
PBT-2 for AD in 2007, and the second phase II trial in 2011. In 2014, Prana reported that there is not much difference between treated and non-treated groups
[148]. An additional phase II trial revealed that 250 mg/day, which the patients could tolerate, of
PBT-2 has positive results in terms of cognitive ability
[149][150]. The clinical trial information of
PBT-2 is summarized in
Table 1.
Table 1. Clinical trials of chemical agents. PBT-2 and deferiprone (DFP) have been examined their potential as treatments for neurodegeneration.
| Chemical Agents |
Clinical Trials
(Periods) |
Status |
| PBT-2 |
Phase 2
(December, 2006-December, 2007) |
Completed
(NCT00471211) |
Phase 2
(September, 2011-January, 2014) |
Completed
(ACTRN12611001008910) |
Phase 2
(July, 2013-January, 2015) |
Completed
(ACTRN12613000777796) |
| Deferiprone (DFP) |
Phase 2
(October, 2016-September, 2019) |
Completed
(NCT02728843) |
Phase 2
(November, 2017-October, 2019) |
Completed
(ACTRN12617001578392) |
Phase 2
(January, 2018-December, 2021 (Estimated)] |
Recruiting
(NCT03234686) |
DP-109 and
DP-460 are lipophilic chelators that chelate Cu, Fe, and Zn in the membrane
[151]. In AD mice,
DP-109 administration reduced amyloid plaques and the degree of cerebral amyloid angiopathy in the brain
[152]. Furthermore, both molecules slightly extended (10% and 9%, for
DP-109 and
DP-460, respectively) the life span of G93A-transgenic ALS mice
[153]. However, a recent study presented that
DP-460 has detrimental effects on learning based on the Morris water maze test
[154]. Moreover,
bathocuproine (
BC) and
bathocuproine disulfonate (
BCS), presented in the middle row of
Figure 2, could chelate Cu(I) and Cu(II) by N as electron donor atoms near methyl groups.
BC prefers Cu(I) to Zn(II) due to its size
[155].
BCS has two negative charges which provide water solubility to the molecule. Thus,
BCS can be used to chelate extracellular Cu(II) as well
[156][157][158].
Multiple chelators against Cu have been developed to treat other diseases such as Wilson’s disease.
D-penicillamine is one of those Cu ion chelators (
Figure 2; bottom row)
[159][160][161]. It chelates and reduces Cu(II), which is then excreted in the urine at up to 1.5 mg/day, which is a four-five times increased amount compared to untreated cases in patients
[162]. Additionally, upon treatment of
D-penicillamine in a hydrogel form, it could improve the cognitive ability of APP/PS1 mice through the activation of ADAM10
[163].
Trientine (triethylenetetramine) is a selective Cu(II) chelator that suppresses oxidative stress. Once Cu(II) binds to
trientine, a Cu(II)–
trientine complex is excreted in the urine, but not as much as
D-penicillamine (
Figure 2; bottom row)
[164][165].
Tetrathiomolybdate, shown in the bottom row of
Figure 2, consists of a central molybdenum surrounded by four sulfhydryl groups and has been suggested as a therapeutic agent for Wilson’s disease to control the levels of Cu(I/II) as well. Once it binds to Cu ions in foods, it forms a very stable complex and is excreted in the stool
[166].
Tetrathiomolybdate also forms a complex with serum albumin and labile Cu ions in the blood. Since Cu–
tetrathiomolybdate cannot be reabsorbed, the complex is metabolized in the liver. The resulting metabolized fragments are excreted in bile. Moreover, upon treatment with
tetrathiomolybdate, the production of inflammatory cytokines decreased in APP/PS1 transgenic mice
[167]. If
tetrathiomolybdate is used at more than a specified dose of 120 mg/day, two side effects might appear, namely, anemia, and elevation of the transaminase level
[168].
There are several multifunctional molecules where other scaffolds are conjugated to metal-chelating moieties for reducing possible side effects or improving metal chelation for treatment of AD
[1]. Since there are many factors that cause AD, multifunctional chemical agents that target multiple risk factors of the disease would be an effective strategy to treat/cure AD. In particular, many chemical agents targeting both Cu(I/II) and Aβ or Cu(I/II) and ROS have been developed. Based on multifunctionality, a few chemical agents, although they have firstly been suggested as Cu(I/II) chelators, are still examined for their potentials as therapeutic agents for AD in various animals (
Table 2)
[142][152][153][154][163][167].
Table 2. Recent animal studies conducted with Cu-targeting chemical agents. Results of in vivo experiments performed with clioquinol (CQ), DP-109, DP-460, D-penicillamine, and tetrathiomolybdate are summarized.
| Chemical Agents |
Animal
Models |
Outcomes |
References |
| Clioquinol (CQ) |
C. elegans |
Enhancement of neuroprotective effect |
[142] |
| DP-109 |
Transgenic
mouse |
Extension of life span and
Reduction of amyloid plaques |
[152][153] |
| DP-460 |
Transgenic
mouse |
Extension of life span and
Reduction of amyloid plaques |
[152][153] |
| Rat |
Detrimental effect on learning |
[154] |
| D-Penicillamine |
Transgenic
mouse |
Improvement of cognitive ability |
[163] |
| Tetrathiomolybdate |
Transgenic
mouse |
Decrease of the inflammatory cytokines |
[167] |
More than ten multifunctional molecules based on
8-HQ have been developed. They have tetra-O-benzyl-β-D-glucopyranoside, rasagiline, trehalose, glutathione, and β-cyclodextrine conjugated to
8-HQ [169][170][171][172][173][174][175][176]. By applying
thioflavin-T (
ThT), an imaging agent for aggregated Aβ, several multifunctional molecules were developed by conjugating
CQ,
DTPA,
di-(2-picolyl)amine, and/or
N-(2-pyridylmethyl)amine [177][178][179][180][181][182][183][184]. Multifunctional molecules based on
p-I stilbene (
pISTIB) can chelate Cu(II) to regulate Cu(II)-induced Aβ aggregation
[185][186][187][188][189][190][191][192][193][194]. Even though the exact role of triazole has not yet been elucidated, triazole-based chemicals have been developed having a quinoline ring and a phenol
[195][196][197]. Multifunctional molecules affected metal-free Aβ aggregation, and others reduced Cu(II)-induced Aβ aggregation when
selegiline,
aurone, and
chromone were conjugated
[198][199][200]. An antioxidant molecule,
resveratrol, was used to invent multifunctional molecules by incorporation with
CQ and
deferiprone (
DFP)
[201][202].
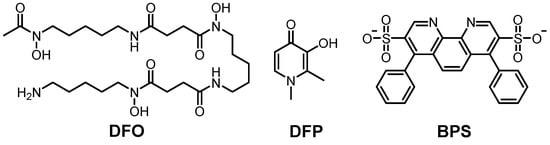
DFP is a well-known Fe(II/III) chelator which is being examined for its potential to treat neurodegeneration. The information of clinical trials (phase II) is summarized in
Table 1, and more information of
DFP is discussed in
Section 3.4 (vide infra).