Cadmium (Cd) is an environmental toxicant with serious public health consequences due to its persistence within arable soils, and the ease with which it enters food chains and then, accumulates in human tissues to induce a broad range of adverse health effects. Diet is a primary exposure source for non-smoking populations, whilst cigarette smoke is an additional source of Cd among those who smoked. Cd exists in cigarette smoke as a non-volatile oxide form (CdO), and a volatile metallic form with high transmission rates. Of further concern, the electronegativity of Cd is similar to that of zinc (Zn), a nutritionally essential metal, whereas its ionic radius is similar to calcium (Ca) Thus, Cd can enter the body from the gut and lungs through the metal transporter systems and pathways evolved for acquisition and storage of Zn, Ca, and other nutritionally essential metals such as iron (Fe) and manganese (Mn).
- bladder cancer
- cadmium
- accquired cadmium tolerance
- Zinc homeostasis
- ZnT1 Zn(Cd) efflux transporter
- ZIP8 Zn(Cd) influx transporter
- ZIP14 Zn(Cd) influx transporter
1. Introduction
2. Cadmium-Induced Cell Transformation: An In Vitro Carcinogenicity Test
3. UROtsa Cell Line as a Cell Model to Dissect the Carcinogenicity of Cadmium
4. Zinc Transporters Expressed by Parental UROtsa Cells
| Zinc Transporters | Number of Transcripts in 1000 β-Actin | ||||
|---|---|---|---|---|---|
| Batch I, 0 µM Cd2+ | Batch II, 0 µM Cd2+ | 1 µM Cd2+ | 2 µM Cd2+ | 4 µM Cd2+ | |
| SLC30A family | |||||
| ZnT1 | 181 ± 23 | 365 ± 38 | 3007 ± 465 | 1434 ± 146 | 1216 ± 153 *** |
| ZnT2 | 0.01 ± 0.001 | 0.06 ± 0.01 | 73 ± 15 | 16 ± 1.9 | 11 ± 1.5 *** |
| ZnT3 | 0.03 ± 0.007 | 0.15 ± 0.01 | 0.24 ± 0.05 | 0.10 ± 0.02 | 0.10 ± 0.02 * |
| ZnT4 | 1.6 ± 0.26 | 11.4 ± 0.8 | 10 ± 1 | 8.7 ± 1 | 6.2 ± 0.5 ** |
| ZnT5 | 150 ± 19 | 510 ± 30 | 1038 ± 132 | 495 ± 54 | 568 ± 91 ** |
| ZnT6 | 4.5 ± 0.15 | 65 ± 8 | 77 ± 6 | 63 ± 13 | 57 ± 12 |
| ZnT7 | 734 ± 28 | 758 ± 76 | 1007 ± 136 | 706 ± 44 | 488 ± 63 * |
| ZnT10 | 0.04 ± 0.005 | 1.1 ± 0.2 | 2.4 ± 0.2 | 1.7 ± 0.2 | 1.1 ± 0.1 *** |
| SLC39A family | |||||
| ZIP1 | 19.5 ± 2.0 | 82 ± 9 | 99 ± 15 | 55 ± 10 | 59 ± 12 * |
| ZIP2 | 0.02 ± 0.004 | 1.2 ± 0.1 | 0.8 ± 0.2 | 0.4 ± 0.1 | 0.2 ± 0.03 *** |
| ZIP3A | 9.1 ± 0.4 | 19 ± 1 | 23 ± 2.3 | 17 ± 1.7 | 14 ± 1.3 * |
| ZIP3B | 0.48 ± 0.05 | 4.1 ± 0.2 | 6.2 ± 0.7 | 4.4 ± 0.2 | 4.2 ± 0.4 * |
| ZIP4 | 0.18 ± 0.04 | 0.06 ± 0.01 | 0.06 ± 0.01 | 0.04 ± 0.01 | 0.06 ± 0.01 |
| ZIP5 | 0.01 ± 0.003 | 0.01 ± 0.001 | 0.01 ± 0.003 | 0.01 ± 0.002 | 0.01 ± 0.002 |
| ZIP6 | 18.3 ± 2.6 | 92 ± 8 | 133 ± 12 | 80 ± 9 | 75 ± 10 ** |
| ZIP7 | 121 ± 9.5 | 204 ± 25 | 342 ± 69 | 149 ± 32 | 94 ± 21 *** |
| ZIP8 | 0.09 ± 0.01 | 2.1 ± 0.2 | 2.6 ± 0.3 | 2.0 ± 0.2 | 2.7 ± 0.4 |
| ZIP10 | 5 ± 0.3 | 54 ± 4 | 30 ± 8 | 14 ± 3 | 14 ± 3 *** |
| ZIP14 | 83.4 ± 10.5 | 146 ± 19 | 218 ± 24 | 158 ± 26 | 128 ± 19 * |
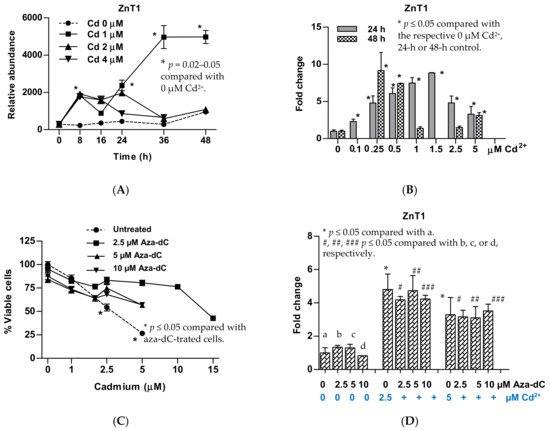
5. Upregulation of ZnT1 and Acquired Resistance to Cadmium
 Encyclopedia
Encyclopedia

