1000/1000
Hot
Most Recent

A 4R’s approach to optimizing post-exercise recovery has been introduced: (i) Rehydration—a fundamental process that will depend on the athlete, environment and sports event; (ii) Refuel—the consumption of carbohydrates is not only important to replenish the glycogen reserves but also to contribute to the energy requirements for the immune system and tissue reparation. Several bioengineered carbohydrates were discussed but further research is needed; (iii) Repair—post-exercise ingestion of high-quality protein and creatine monohydrate benefit the tissue growth and repair; and (iv) Rest—pre-sleep nutrition has a restorative effect that facilitates the recovery of the musculoskeletal, endocrine, immune, and nervous systems. Nutritional consultancy based on the 4R’s is important for the wise stewardship of the hydration, feeding, and supplementation strategies to achieve a timely recovery.
Recovery strategies to optimize post-exercise recovery depend to a large extent on the proximity of the next session, the degree of physiological stress, and the relevance of the next event. This determines how to rehydrate, replenish energy and consume the nutrients needed to improve tissue repair. To improve comprehension regarding the nutritional strategies that impact post-exercise recovery, a mnemonic entitled the 4R’s (Rehydrate, Refuel, Repair, and Rest) is introduced. This approach divides the nutrition intervention into four interrelated scenarios that follow the post-exercise time course in order to optimize the exercise-induced adaptations and recovery (Figure 1).
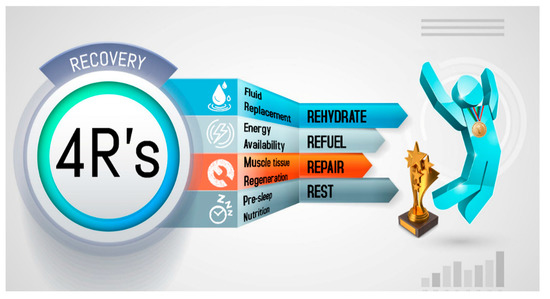
Figure 1. The 4R’s framework of nutritional strategies to optimize post-exercise recovery in athletes.
One of the first goals during recovery is to replace any fluid and electrolyte deficits. Most physically active individuals sweat from 0.3 to 2.4 L·h−1, which depends on exercise intensity, duration, and environmental conditions such as altitude, heat, and humidity [1][2][3][4][5][6][7][18,19]. Moreover, individual characteristics (i.e., body mass, genetic predisposition, heat acclimatization state, physical fitness, and metabolic efficiency) might influence sweat rates for a given activity [3]. For instance, the highest sweat rate was registered at 3.7 L·h−1 for a world-class ultramarathon runner [4]. Thus, measuring pre and post-exercise body mass is a recommended practice to assess fluid status.
Rehydration is important, especially in team, endurance or ultra-endurance sports, where in many cases it is not possible to compensate for the loss of fluids and electrolytes that occur during exercise, particularly in hot and humid environments. As a general advise, for quick rehydration, it is recommended the consumption of 150% of the weight lost after exercise over a short recovery period (less than 4 h) [5][6], with a sodium concentration between 20 and 30 mEq·L−1 [1]. Athletes and practitioners should replenish three cups of fluid per pound of weight lost (~1.5 L·kg−1) and to make sure body mass is back up before the next training session. Furthermore, it has even been shown that consuming a sodium-containing drink between 40 and 60 mEq·L−1 can improve fluid retention and rehydration when there is little time between sessions or when there is moderate dehydration [7].
Rehydration can last between four and 24 h [8]. If recovery time and opportunities allow it, the consumption of sodium-rich foods, such as crackers, peanuts, bread, milk, cheese, ham, kabanos, and soups, may be sufficient to regain the state of euhydration. However, if the recovery time is less than 12 h, more aggressive rehydration strategies and the use of moisturizing beverages (e.g., glycerol) are required before the next training or competition [9]. One practical application to improve both the rate of rehydration and total fluid retention following exercise is the ingestion of glycerol [5][10]; however, professional advice is recommended to avoid potential gastrointestinal discomfort with any hyperhydration agent.
In the rehydration process, it has been found that neither the addition of potassium to the drink [11] nor the way of distributing the volume [12] nor the temperature influence the percentage of liquid preserved to be subsequently used by the body for rehydration [13]; however, it is believed that the delay in early rehydration after exercise is attributed to a reduction in sensations associated with thirst, and it is known that the taste and cool temperature of beverages can positively affect these sensations [14]. On the other hand, some drinks that can be used for rehydration and in turn help recovery has been successfully studied, such as chocolate milk [15][16][17].
Rehydration is a fundamental step in recovery, but what, how, when and how much will depend on the athlete and the particular event. We adhere to the position statement of the National Athletic Trainers’ Association to emphasize that education strategies for athletes should address personal sweat rates, hydration cues, and rehydration strategies that avoid both hypohydration and fluid overload [6].
At the end of the exercise, there are several strategies to maximize muscle and liver glycogen replenishment, especially when two or more sessions are performed on the same day or when competing on consecutive days. For planning, it is necessary to consider the state of training, schedules, and the magnitude of the depletion of reserves, besides the type of exercise [18]. In this sense, the amount of carbohydrates is determined by the need to replenish muscle glycogen stores, and according to Jeukendrup (2017) [19], this is closely related to:
Although certain general recommendations can be given, the carbohydrate intake must be fine-tuned based on individual features, total energy daily expenditure, exercise training requirements, and the respective feedback from training performance in daily recovery [20]. In athletes with high body mass (e.g., basketball and rugby) or players under a weight loss program it might be better to reduce the energy intake to the needs of the previous category [20]. Additionally, resistance/power athletes do not need much carbohydrates as endurance athletes to maintain optimal liver and muscle glycogen; therefore, based on the exercise and sports nutrition review update of the International Society of Sports Nutrition [21], daily carbohydrate needs might be ranked as follows:
Moreover, in the post-exercise period, it takes about four hours for carbohydrates to be digested and absorbed into muscle and liver tissues to be incorporated as glycogen. Hence, if rapid recovery is required due to a limited time period available, the priority should be to consume large amounts of daily carbohydrates (>8 g·kg−1 body mass·day−1) and to eat a high carbohydrate meal within two hours following exercise with at least 1.2 g·kg−1·h−1 for the first four hours of recovery [22]. Ingestion of a glucose polymer or the combination of glucose and fructose (sucrose) results in a fast replenishment of muscle glycogen stores whilst also minimizing gastrointestinal distress [23], and there is no need for protein and/or amino acid ingestion in order to enhance the insulin levels if sufficient carbohydrates are consumed (1.2 g·kg−1·h−1). In fact, higher insulin concentrations do not further increase the rate of muscle glycogen synthesis when carbohydrate intake is sufficient [19]. Slightly less carbohydrate plus protein (e.g., 1 g carbohydrate·kg−1 and 0.5 g protein·kg−1) within 30 min after exercise or carbohydrates along with caffeine may also be used to aid rapid glycogen resynthesis [21]. Additionally, compared to carbohydrate ingestion alone, multiday supplementation with creatine monohydrate along with an adequate amount of carbohydrates has been reported to have a higher positive impact on muscle glycogen synthesis [24].
The consumption of carbohydrates is not only important to replenish the reserves, but also to contribute to cover the energy requirements that are fundamental to help the competition of the immune system and the repair of the tissues [25]. Sports nutritionists, coaches and athletes should be cautious with the potential physiological implications of the relative energy deficiency in sport, which includes impaired metabolic rate, hormonal disruptions, menstrual dysfunction, reduced bone health, immunity, protein synthesis, and cardiovascular health [26][27].
Considering the potential impact of the type of carbohydrates on performance and recovery, some bioengineered formulations with different physicochemical characteristics are described in the next lines. These bioengineering processes refer to the application of theoretical and experimental methods of the basic sciences to produce new scientific knowledge with practical applications, such as the modification of the chemical structure of a molecule by means of biotechnological procedures that use microorganisms (e.g., bacteria) in order to produce substances with different metabolic responses since they are not found regularly in food. We recommend the reader to visit the Carbohydrate Structure Database (CSDB, http://csdb.glycoscience.ru) [28] and the database of Chemical Entities of Biological Interest (ChEBI) [29] to know in depth about advances, structures and different applications of the new generation of carbohydrates produced through different processes and that are of biological interest.
Scientific research has demonstrated that muscle protein synthesis (MPS) can be stimulated by either a physical allostatic challenge (e.g., resistance exercise stimulus) or by the ingestion of dietary protein, with synergistic responses when protein is consumed before and immediately after resistance exercise training [30]. According to the International Society of Sports Nutrition position stand on nutrient timing, post-exercise ingestion (immediately to 2 h) of high-quality protein food represents a robust stimulus that impacts positively on MPS; however, similar increases in MPS have been found when high-quality proteins are ingested immediately before exercise [31]. Indeed, taking into account the sport-specific daily needs, if an insufficient amount of protein is consumed, the athletes will develop and maintain a negative nitrogen balance, which is an indicator of protein catabolism and negatively affect recovery. Over time, this can lead to muscle wasting, injury, disease and intolerance to training [21]. Thus, the peri-exercise ingestion of insulinotropic protein and/or essential amino acid mixtures might stimulate post-exercise net muscle protein anabolism, and this might contribute to faster tissue growth and repair [18]. Similarly, recent findings have provided evidence that marathon runners that consume moderate amounts of protein post-exercise can have recovery benefits [32][33]. With a good grade of evidence, compared to ingestion of carbohydrate alone, co-ingestion of carbohydrate plus protein together during the recovery period have resulted in no difference in the rate of muscle glycogen synthesis but it improves net protein balance [1].
The International Society of Sports Nutrition position stand [30] about proteins for recovery is:
On the other hand, a large body of evidence suggests that creatine monohydrate supplementation (0.1 g·kg−1·day−1) not only optimize exercise adaptations and increase performance but also may reduce muscle damage and/or enhance recovery from intense exercise [34]. These effects are partially due to the optimization of the creatine kinase (CK) system which not only serves as a spatial/temporal buffer of ATP regeneration but also leads to positive regulation of anabolic signaling pathways (such as IGF-I and MAPK) and, hence, might promote faster tissue repair and recovery [35]. Additionally, it has been shown that chronic creatine supplementation prior to an exhaustive exercise bout and glycogen loading promotes greater glycogen resynthesis than just carbohydrate loading alone [36].
The role of antioxidants and anti-inflammatory substances are also highlighted in the post-exercise repair process. An increased antioxidant status leads to the reduction of oxidative stress caused by the production of reactive oxygen species during the inflammatory process [37]; therefore, the use of antioxidants can reduce muscle soreness and help with recovery in the short term, but high doses have also been linked to reduced training benefits in the long term. For example, Levers et al. (2015) found significant beneficial effects on serum markers of muscle catabolism, physiological stress, and inflammatory mechanisms after a short-term supplementation with 480 mg·day−1 of Montmorency powdered tart cherries surrounding a single bout of resistance exercise [38]. More recently, Brown et al. (2019) also reported that supplementation with Montmorency cherry concentrate can be considered as a practical nutritional intervention to reduce symptoms of muscle damage and improve post-exercise recovery on subsequent days in females [39]. It appears that secondary metabolites with antioxidant properties that are found in tart cherry extract might attenuate muscle soreness, strength decrement during recovery, and markers of muscle catabolism in resistance-trained individuals. In this sense, a systematic review and meta-analysis about the effects of antioxidants consumption showed that short-term polyphenol supplementation (e.g., quercetin) may boost athletic performance in predominately-trained males with an average intervention dose of 688 ± 478 mg·day−1 [40]. This has been reinforced by more recent research showing that acute and chronic supplementation with >1000 mg fruit-derived polyphenols per day will enhance recovery following muscle damage via antioxidant and anti-inflammatory mechanisms [41]; which suggest that fruit supplements could be used as part of the post-exercise recovery strategy although the need to educate and encourage athletes to consume more fruits and vegetables in the diet should be associated not only with recovery but also with health [42]. Similarly, curcumin is rich in polyphenols and has shown anti-inflammatory and antioxidant properties. The supplementation with 6 g of curcumin and 60 mg of piperine each day between 48 h before and 48 h after exercise-induced muscle damage have resulted in positive effects on recovery of the muscle function at 24 h and 48 h after the exercise [43].
The use of beetroot juice has been investigated in the management of hypertension [44][45], for the improvement of physical performance [46][47], and also in post-exercise recovery [48]. It has been found that acute supplementation of beetroot juice (250 mL) ×3 servings, two serving 24 h and 48 h following completion of 100-drop jumps attenuated muscle soreness and decrements in countermovement jump performance induced by eccentric exercise, while apparently having no effect on maximal isometric voluntary contractions, CK and some inflammatory markers (IL-6, TNF-alpha and IL-8) [49]. Interestingly, a betalain-rich concentrate of beetroots has shown to improve performance in competitive male and female triathletes and attenuated the increase of CK and fatigue suggesting an increase in recovery [50]. Other herbal and mushroom supplements that have promising effects to improve post-exercise recovery are Zingiber officinale [51], Zingiber officinale + Bixa orellana L. [52], Rhodiola rosea [53], Cordyceps militaris [54], and Rhodiola rosea + Cordyceps sinensis [55]. A root extract that warrants special attention is Withania somnifera (most known as ashwagandha) given some research have found ergogenic effects in athletic [56][57] and physically active individuals [58]. Moreover, the adaptogenic, anti-inflammatory, and antioxidant properties of ashwagandha [59][60][61] turns this herbal extract into a potential strategy to optimize recovery and promote exercise-induced adaptations. Although more research is required, dosages are between 300 and 500 mg of aqueous root extract twice per day.
On the other hand, it appears that supplementation with branched-chain amino acids (BCAA) after high-intensity exercise may favor a hormonal environment that contributes to attenuate the loss of strength, reduce muscle damage, and generate an anabolic environment [62]. Indeed, several systematic reviews and meta-analysis have concluded that BCAA supplementation (>200 mg·kg−1·day−1) may optimize recovery and mitigate muscle soreness following muscle-damaging exercise [63][64][65][66]. Notwithstanding, in resistance-trained males, the attenuation of muscular performance decrements and the observed decrease in plasma CK levels after BCAA supplementation is likely negligible when consumed with a diet consisting of ~1.2 g·kg−1·day−1 of protein[67]. The leucine-derived compound HMB has shown to improve work capacity recovery after high-intensity exercise [68], and may attenuate markers of muscle damage, augment acute immune and endocrine responses while preventing loss of lean body mass in catabolic situations [69]. A recent systematic review and meta-analysis by Rahimi et al. (2018) concluded that HMB may be seen as a recovery agent following exercise-induced muscle damage [70], but more research on recovery from injury that includes periods of extreme inactivity is needed given it does not consistently increase strength and/or lean mass or reduce markers of muscle damage [71]. Finally, it has been recently reviewed systematically if other foods, such as Pomegranate [72], cow’s milk [73], or Chocolate milk [74], might potentially improve exercise-performance and post-exercise recovery but further research is needed to extract definitive conclusions.
There is no doubt that sleep is an absolutely vital physiological function and one of the most important factors in post-exercise recovery [75]. It has been emphasized that naps, sleep extension, and sleep-hygiene practices seem to be advantageous to the performance by optimizing recovery [76]. In spite of the above, von Rosen et al. (2017) reported that the recommended amount of sleep during weekdays (8 h) was not obtained by 19% of 340 Swedish adolescent elite athletes of several disciplines during the autumn semester. Moreover, athletes sleeping more than eight hours and reached the recommended nutrition intake reduced the odds of suffering a new injury [77]. Portuguese elite female gymnasts have also found to have poor sleep habits with consequences on daytime sleepiness, sleep quality, and low energy availability associated with macro and micronutrients’ deficiencies [78]. In fact, according to a recent systematic review by Gupta et al. [79], athletes show a high overall prevalence of insomnia symptoms characterized by increased sleep latency, sleep fragmentation, non-restorative sleep, and excessive daytime fatigue. Currently, there is a lack of evidence and future research should focus on conducting sleep interventions among different athlete populations to address their specific sleep demands and disturbances [80].
It is known that eating the right combination of foods before going to sleep and what foods to avoid in the evening may be beneficial in enhancing sleep [81]. That is the rationale for the pre-sleep nutrition strategies, considering that several nutrients have been shown to improve sleep such as carbohydrates (high-glycemic index dinners), melatonin, tryptophan-rich protein, antioxidant-rich fruits (e.g., tart cherry juice and kiwi), and micronutrients [82]. Casein proteins, a type of secreted calcium (phosphate)-binding phosphoproteins, are among the most common nutrients used for pre-sleep nutrition given they are considered a high-quality protein source with high digestibility and bioavailability but with a slower digestion rate in comparison to whey [83]. Thus, the timing of nutrient intake is as important as the composition to fulfill the nutrition needs of the athletes [31]. Res et al. [84] reported for the first time that casein protein ingestion immediately before sleep was not only effectively digested and absorbed but also increased MPS and net protein balance in healthy young males that performed a resistance-training bout in the evening. Moreover, it has been demonstrated that pre-sleep casein protein ingestion augments the muscle adaptive response in terms of muscle mass and strength after a 12-week resistance exercise training program in young men in comparison to placebo [85]. Therefore, the extended window of opportunity as a result of the additive effects of resistance exercise training and protein ingestion on MPS makes the pre-sleep casein protein supplementation an effective nutrient timing strategy to optimize muscle conditioning and recovery [86] with no need to add extra leucine [87]. Although the positive effects of pre-sleep nutrition have been found particularly in resistance-type exercise training [88], more research is needed in endurance-trained athletes considering recent findings showed no improvement [89]. The available evidence and recommendations under this new paradigm of pre-sleep nutrition are:
The consumption of 40–48 g of casein approximately 30 min before sleep improves post-exercise recovery and positively affect acute protein metabolism during an overnight period in healthy young adults [83][90].
Ashwagandha supplementation (>150 mg aqueous root extract quaque hora somni) seems to be an effective nutritional strategy to improve sleep quality in healthy male and female subjects [91]; consequently, it should be also considered before sleep.