1000/1000
Hot
Most Recent

Peretinoin, an retinoid acid (RA), a metabolite of vitamin A and its related analogues (termed retinoids) has been suggested as a promising chemotherapeutic agent in cancer treatment. The synthetic oral retinoid peretinoin is the only agent for the secondary chemoprevention of HCC after curative therapy that is currently well applied into clinical development.
Liver cancer is the fifth most common neoplasm in men and the seventh most common neoplasm in women worldwide [1][2]. The incidence and mortality rates of liver cancer are particularly high in Asia, with over 75% of patients with liver cancer being from Asian countries [1][3]. Hepatocellular carcinoma (HCC) is the most common type of liver cancer. The prognosis of patients with HCC has improved recently owing to developments in various therapeutic methods, including surgical resection, ablation therapy [mostly radiofrequency ablation (RFA), percutaneous-ethanol-injection (PEI)], transarterial chemoembolization (TACE), and molecularly targeted agents such as sorafenib, lenvatinib, and regorafenib, liver transplantation [4][5][6][7][8][9]. Despite successful treatment, however, HCC has a high risk of recurrence. The mode of recurrence is identical in patients with HCC caused by hepatitis B virus (HBV) and hepatitis C virus (HCV) and is thought to be due to manifestations of intrahepatic metastases and metachronous multicentric carcinogenesis. Moreover, although it is difficult to determine the mode of recurrence of individual lesions, the timing of recurrence is believed to differ. For example, recurrence from intrahepatic metastases is predominant within 2 years after radical treatment of the primary tumor, whereas metachronous multicentric recurrence occurs predominantly after 2 years [10][11]. Most patients ultimately die of HCC, due to the occurrence of non-responding lesions, such as intraportal tumor thrombi, diffuse multiple cancers, and distant metastases.
In addition to early detection and early treatment, the prognosis of patients with HCC may be improved by aggressively suppressing HCC recurrence. Antiviral therapy, mainly with nucleic acid analogs and Direct acting antivirals (DAAs), may inhibit the recurrence of HBV- and HCV-positive HCC after radical treatment of patients [12][13]. However, there is no established non-viral strategy to prevent recurrence of virus associated HCC after curative therapy. The incidence of HCC in patients with non-alcoholic fatty liver disease (NAFLD) and metabolic syndrome has recently increased [14]. Because HCC can develop in patients with NAFLD, even in the absence of established cirrhosis, to prevent recurrence of non-virus associated HCC after curative therapy, there is an urgent unmet need for secondary chemoprevention of HCC recurrence after curative therapy. This review investigated the current status and prospects of peretinoin, an acyclic retinoid (ACR) compound developed for secondary prevention of HCC.
Peretinoin is a synthetic polyprenoic acid that binds to cellular retinoic acid-binding protein [15] and has retinoid-like properties. Initially, it was developed under the name E-5166 (Eisai Co., Ltd.) to treat dermal diseases. A randomized placebo controlled study showed that oral administration of peretinoin (600 mg/day, twice daily) for one year to patients who had undergone curative resection or percutaneous ethanol injection therapy for viral and non-viral HCC showed good tolerability, inhibited HCC recurrence, and improved patient survival rate [16][17]. Based on these promising results, peretinoin was developed under the name NIK 333 (Kowa Company, Ltd.) in 1997, clinical studies were started in February 2012, and the name of the compound was changed to K-333. The mechanism of action of peretinoin includes transcriptional activation via the retinoic acid receptor (RAR) and retinoid X receptor (RXR), promoting, along with other transcriptional complexes, the transcription of target genes. Peretinoin was found to modulate genes involved in the regulation of cellular proliferation, cellular differentiation and apoptosis in HCC cells [18][19][20]. Several pharmacologic studies have indicated that peretinoin inhibits the recurrence of HCC by inhibiting carcinogenesis of precancerous lesions in the liver and/or by inhibiting the growth of subclinical cancers [19][21].
The chemical name of peretinoin is (2E,4E,6E,10E)-3,7,11,15-tetramethylhexadeca-2,4,6,10,14-pentaenoic acid (Scheme 1). Peretinoin consists of crystals or crystalline powder, pale yellow to yellow in color, with a molecular formula of C20H30O2 and a molecular weight of 302.45 g/mol. Peretinoin was highly soluble in dimethylsulfoxide, soluble in ethanol (99.5%) and acetone, sparingly soluble in methanol, slightly soluble in acetonitrile and hexane, and practically insoluble in water.
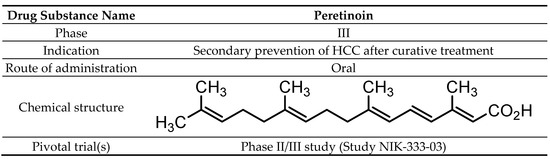
Scheme 1. Drug substance summary.
Peretinoin was found to activate transcription through retinoic acid receptor (RAR) and retinoid X receptor (RXR), and could activate the differentiation of human acute promyelocytic leukemia in HL-60 cells [22][23]. Peretinoin significantly inhibited hepatocarcinogenesis in rat chemical carcinogenesis models using 3′-methyl-4-(dimethylamino)azobenzene (3′-MeDAB) and diethylnitrosamine (DEN) [24][25], suggesting that peretinoin was also involved in suppressing hepatocarcinogenesis. Furthermore, peretinoin was reported to reduce the numbers of oval cells with stem cell-like properties that appear during early stages of 3′-MeDAB-induced hepatocarcinogenesis in rats [26][27]. All-trans-RA was found to induce the differentiation of hepatic precursor cells derived from mouse fetal liver cells [28], and peretinoin is thought to act similarly. Peretinoin treatment also prevented obesity-related liver carcinogenesis and attenuated liver steatosis and inflammation [29], thereby being expected to reduce HCC incidence in patients with liver cirrhosis. These results suggest that, due to its retinoid-dependent and -independent effects, peretinoin prevents the recurrence of HCC by inhibiting oncogenesis of precancerous lesions in the liver and/or by inhibiting the growth of occult hepatic cancer. In addition to preclinical study, the effect of peretinoin in human was investigated through clinical study (NIK-333-02) [18]. In this study, the change of gene expression profile after 8 weeks of peretinoin treatment was examined in liver biopsy for patients who had received curative treatment for HCC. The levels of expression of genes encoding interferon, tumor suppressors, negative regulators of Wnt and insulin-like growth factor (IGF) signaling, hepatocyte differentiation and retinoid-induced genes were higher after 8 weeks of peretinoin treatment than before treatment. In contrast, genes related to mammalian target of rapamycin (mTOR), tumor progression, the cell cycle and metastasis/angiogenesis were downregulated. Through retinoid target gene, peretinoin inhibit HCC proliferation, suppress tumor growth or induce tumor apoptosis [30][31][32][33].
It is well known that abnormalities in the genes regulating Wnt signaling, IGF signaling, interferon, mTOR, and the cell cycle have been indicated to play a crucial role in the development of HCC [34][35]. Gene expression profiles in the liver identified candidate drug-response genes among the genes which exhibited changes in expression before and after peretinoin administration. These genes included Wnt signal-related, IGF signal-related, interferon-related, mTOR-related, and cell cycle-related genes known to be involved in the process of carcinogenesis of hepatocellular carcinoma, in addition to the retinoid-related genes which are the targets of peretinoin, suggesting that peretinoin directly or indirectly regulate many signal transduction systems in the carcinogenic process. Furthermore, with a hierarchical cluster analysis showing the feasibility of differentiating within-2-year-recurrence and non-recurrence groups, a comparative analysis of the two groups was performed. The results showed that, in the non-recurrence group, differentiation of hepatocytes and expression of the genes involved in tumor suppression were accelerated, while expression of the genes involved in promotion of liver fibrosis and lipidation, as well as the genes which can become markers for the stem cells of hepatic cancer was reduced. Changes in the expression of these genes may not reflect the direct action of peretinoin, but they were assumed to become eventual candidates for the genes related to drug efficacy. Furthermore, genes with a potential to distinguish the two doses were extracted by a comparison of the two dose groups of 300 mg and 600 mg of peretinoin. These genes included retinoid-related genes as well as genes that play a significant role in hepatic cancer and hepatic cirrhosis. The exact modes of action are not certain. Through these mechanisms, peretinoin suppresses HCC cell proliferation, and therefore could prevent HCC recurrence by modulating multiple signaling cascades involved in carcinogenesis, either directly or indirectly (Figure 1) [18][20].
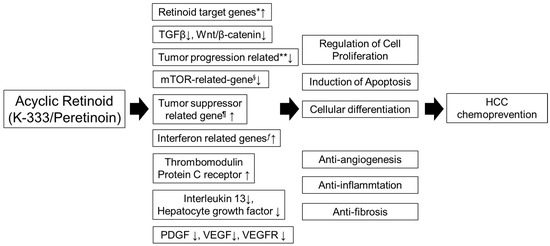
Figure 1. Molecular pathway of peretinoin. Peretinoin enhances the expression of multiple retinoid target genes. Through the retinoid target gene, peretinoin inhibits HCC proliferation, suppresses tumor growth and induces tumor apoptosis. In addition to enhancing retinoid target gene expression, peretinoin induces changes in the expression levels of a variety of genes involved in hepatocarcinogenesis, such as those related to Wnt signaling, IGF signaling, interferon, mTOR, and cell cycle regulation. Therefore, peretinoin modulates multiple signaling cascades involved in carcinogenesis, either directly or indirectly. HCC, hepatocellular carcinoma; IGF, insulin-like growth factor; mTOR, mammalian target of rapamycin; VEGF, vascular endothelial growth factor; PDGF, platelet-derived growth factor. * cytochrome P450, family 26, subfamily B, polypeptide 1, insulin-like growth factor binding protein 6, regulatory factor X-associated ankyrin-containing protein, putative lymphocyte G0/G1 switch gene, retinol binding protein 1, retinol binding protein 4, retinoic acid induced 3, transglutaminase 2, CCAAT/enhancer binding protein (C/EBP). ** junctional adhesion molecule 3, V-myc myelocytomatosis viral oncogene homolog, Src-like-adaptor, cell division cycle 2, G1 to S and G2 to M, BCL2-associated athanogene, chemokine (C-C motif) receptor 9, pre-B-cell leukemia transcription factor 1. § FK506 binding protein 12-rapamycin associated protein 1. ¶ Jumonji domain containing 3, jumping translocation breakpoint, protein kinase, AMP-activated, alpha 2 catalytic subunit. ƒ guanylate binding protein 1, interferon-inducible, 67kDa, interferon-induced protein 44, chemokine (C-X-C motif) ligand 9.
The Wnt/β-catenin carcinogenesis pathway is frequently activated in HCC associated with hepatitis virus, especially HCV. As peretinoin has been shown to upregulate genes associated with the negative regulation of Wnt/β-catenin signaling, studies have investigated the chemopreventive activities of peretinoin in HCV- and HBV-associated HCCs [18][36]. In contrast, the β-catenin pathway is not activated in metabolic syndrome associated HCCs. Rather, carcinogenic mediators include insulin, lipid peroxidation and oxidative stress induced by free radicals, all of which stimulate cellular proliferation, activate hepatic progenitor cells and induce p53 mutations and epigenetic aberrations [37][38]. The non-activation of the Wnt/β-catenin pathway in metabolic syndrome associated HCCs suggested that peretinoin may not be effective as a chemopreventive agent in these patients. However, peretinoin also downregulates the expression of genes related to inflammation [18]. In a rat model, peretinoin targeted platelet-derived growth factor (PDGF) signaling, preventing hepatic fibrosis, steatosis and HCC development [20] and suggesting that peretinoin may be useful in the chemoprevention of metabolic syndrome related HCCs. It was reported that peretinoin activates the autophagy pathway by increasing Atg16L1 expression to prevent the progression of HCC in NAFLD mouse model [39]. Indeed, a recent preclinical study in obese and diabetic db/db mice with DEN-induced HCC showed that peretinoin significantly reduced the incidence of obesity related HCC [29]. Thus, peretinoin may be useful in preventing metabolic syndrome related HCC in humans, a hypothesis requiring testing in clinical trials.
Administration of single doses (300, 600 and 900 mg) of peretinoin to human subjects confirmed dose proportionality, with the lipid form of peretinoin reaching saturation at high doses. The route of drug administration is per oral. At all dose levels, urinary excretion of peretinoin was not observed [40]. Results of 24-week repeated-dose administration (150 mg × 2/day, 300 mg × 2/day, and 450 mg × 2/day) confirmed that peretinoin and its lipid form reached and maintained steady state concentrations in plasma for up to 24 weeks. Assessments of the elimination phase showed that peretinoin concentrations near the quantitation limit were present in some subjects after completion of administration at each dose. However, the level was lower than 1% of the Cmax after a single dose at each dose level. In most subjects, the plasma concentrations of peretinoin and peretinoin lipids were lower after completion of administration than the quantitation limit at 12 weeks. These results suggested that peretinoin does not accumulate in plasma, and that the steady state can be maintained for up to 24 weeks after administration. Moreover, food intake had no effect on plasma peretinoin concentration, as these concentrations did not differ in subjects administered a single of 300 mg peretinoin in the presence or absence of food [40]. A study of its clinical pharmacology showed dose-dependent increases in plasma peretinoin concentrations following 8-week repeated administration at doses of 300 mg/day and 600 mg/day. Peretinoin concentrations in the liver were below the lower quantitation limit (0.050 μg/g) in all six subjects administered 300 mg/day peretinoin and in four of the six subjects administered 600 mg/day peretinoin. The other two subjects administered 600 mg/day peretinoin had plasma peretinoin concentrations of 0.052 μg/g and 0.059 μg/g, respectively. Repeated-dose administration for up to 96 weeks showed no evidence of drug accumulation in the liver [18].
Peretinoin, a synthetic oral ACR discovered in 1980, has been specifically developed for secondary chemoprevention in patients who had undergone curative surgical or local ablative treatment for HCC. Clinical trials on the tolerability and efficacy of peretinoin indicated that twice daily administration of 600 mg/day peretinoin would be effective in preventing HCC recurrence and that peretinoin was safe and tolerable. Its long-term safety and marginal efficacy with hepatitis C virus- or hepatitis B virus-associated HCC for its clinical use as adjuvant therapy in patients is of concern. Subsequent placebo-controlled phase III clinical trials are underway. The outcome of these phase III trials will determine whether peretinoin is approved for clinical use. Therefore, the dose of peretinoin (2 × 600 mg/day) might be considered high as for the anticancer drug under clinical development. To exert more effective and satisfactory outcomes of ACR to enable HCC chemoprevention, the combination therapy of ACR with more potent anticancer agents would be considered [41][42].