1000/1000
Hot
Most Recent

Gynaecological cancers can be primary neoplasms, originating either from the reproductive tract or the products of conception, or secondary neoplasms, representative of metastatic disease. For some of these cancers, the exact causes are unknown; however, it is recognised that the precise aetiopathogeneses for most are multifactorial and include exogenous (such as diet) and endogenous factors (such as genetic predisposition), which mutually interact in a complex manner. One factor that has been recognised to be involved in the pathogenesis and progression of gynaecological cancers is the endocannabinoid system (ECS). The ECS consists of endocannabinoids (bioactive lipids), their receptors, and metabolic enzymes responsible for their synthesis and degradation.
Cannabis is the botanical name of an annual herbaceous plant of the Cannabaceae family that is cultivated and distributed all over the world. This genus consists of three major species, C. sativa, C. indica, and C. ruderalis, which, through interbreeding, share similar genetic backgrounds and physical traits [1]. One distinctive trait of Cannabis plants is production of secondary compounds called “phytocannabinoids”, of which over 100 are produced by the female Cannabis inflorescence [2]. The first evidence for the medical use of Cannabis dates to the Han Dynasty in ancient China, where it was recommended for pain, constipation, agitation, hysteria, spasmodic cough, disorders of the female reproductive tract, and other less defined conditions [3]. Of the 100 or so phytocannabinoids, the most potent is Δ-9-tetrahydrocannabinol (THC), which was isolated and identified as a major psychoactive compound in the 1960s [4]. This was followed by the discovery of additional phytocannabinoids, such as cannabidiol (CBD), cannabinol (CBN), cannabichromene (CBC), cannabigerol (CBG), tetrahydrocannabivirin (THCV), and Δ-8-THC [5].
In the early 1990s, two different G-protein-coupled receptors able to interact with phytocannabinoids were discovered in the central nervous system and the spleen; these receptors are now called type 1 and type 2 cannabinoid receptors (CB1 and CB2), respectively [6][7]. Their discovery was shortly followed by that of their ligands—two specific endogenous bioactive lipids, N-arachidonoylethanolamine (also known as anandamide, AEA) and 2-arachidonoylglycerol (2-AG) from animal tissues [8][9]. Later, the metabolic enzymes that regulate the production and degradation of these endogenous cannabinoids (endocannabinoids; eCBs) were discovered, followed by ancillary ligands, receptors, and transporters. These altogether represent the “endocannabinoid system (ECS)”, which is ubiquitously distributed in the body [4][10][11], including both the male and female reproductive tissues [12][13].
The main elements of the ECS are all expressed in human female reproductive tissues, such as the ovaries [14], Fallopian tubes (oviduct) [15], uterus [16], and placenta [17] (Figure 1). They have also been localised to areas of the hypothalamus responsible for producing hormones, which act through the hypothalamic–pituitary–gonadal (HPG) axis to control a great number of reproductive functions [18]. In the human ovaries, CB1 and CB2 have been shown to be expressed in the granulosa cells of primordial, primary, secondary, and tertiary follicles, as well as in theca cells of secondary and tertiary follicles (Figure 1), with the highest expression at the time of ovulation [14]. Additionally, both receptors are expressed in the corpus luteum and corpus albicans, even in the absence of pregnancy [14]. Moreover, FAAH has been shown to be present within theca cells, but NAPE-PLD appears only in the granulosa of secondary and tertiary follicles, the corpus luteum, and corpus albicans [14].
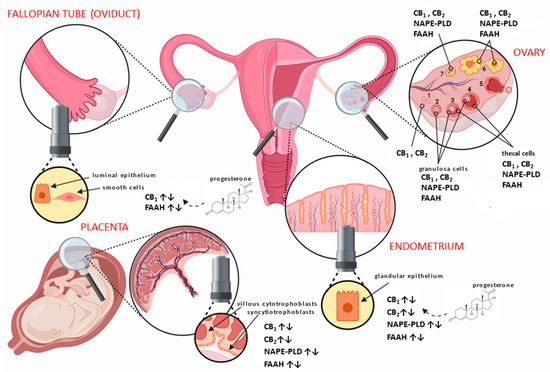
Figure 1. Distribution of the main endocannabinoid system (ECS) components in human female reproductive tissues. In the ovary, the different stages of follicular development from (1) primordial/primary, (2) secondary, (3) tertiary, (4) pre-ovulatory/Graafian, to (5) ovulating follicles are depicted. After ovulation is complete, the condensing granulosa and mural thecal cells form the corpus luteum (6), a structure that produces the progesterone required for continued early pregnancy. In the absence of pregnancy, the corpus luteum degenerates into the corpus albicans (7). Throughout the ovarian cycle, CB1 (type 1 cannabinoid receptor) and CB2 (type 2 cannabinoid receptor), fatty acid amide hydrolase (FAAH), and N-acylphosphatidylethanolamine-specific phospholipase D (NAPE-PLD) are produced in the various cells of the developing follicle and corpus luteum, including the oocyte [14]. Similarly, CB1, CB2, FAAH, and NAPE-PLD are expressed in the Fallopian tube [19] and endometrium [16] throughout the menstrual cycle, where they are regulated by the actions of estradiol and progesterone. The cytotrophoblast and syncytiotrophoblast cells of the early placenta also express CB1, CB2, FAAH, and NAPE-PLD [17], where modulation of protein expression occurs when production of progesterone changes from the corpus luteum to the placenta.
In the Fallopian tube, CB1 is expressed primarily in the smooth muscle cells and in surrounding blood vessels, with lower expression in the cytoplasm of epithelial cells lining the lumen of the tube [15]. In the endometrium, CB1 mRNA and protein levels increase in the secretory phase, probably under the influence of progesterone [20], while CB2 expression is minimal at the beginning of the cycle and increases markedly during the late proliferate phase of the menstrual cycle [16]. Interestingly, CB1 mRNA is only present at low levels in both the Fallopian tube and the endometrium of women with an ectopic pregnancy [15].
There have been several reviews [21] on this, and these are summarised in Table 1. All the components of the ECS are present and active in all parts of the female reproductive tract. Here, they play roles that include oocyte production [14][22][23][24][25], oviductal transport [25][26][27], and blastocyst maturity and implantation [28], as well as in preparing the endometrium for implantation [16][29][30][31][32]. When the ECS is dysfunctional or interfered with by, for example, cannabinoids [33][34], human fertility may be impaired (reviewed in [12][28][35][36]) and there may be associated reproductive-tissue-dependent pathologies, such as endometriosis, miscarriage, ectopic pregnancy, or pre-eclampsia [37][28][35][38][39][40]. Two recent reviews [12][18] on the ECS in the female reproductive tract summarise what is known on this topic, but crucially, these omit some important details on the main ECS components in gynaecological cancers, which we hope to address in this review. In this context, the ECS has been studied the most in the human ovary [14][22][41][42][43][44][45][46], cervix [47][48][49][50][51], and endometrium [16][20][52][53][54][55][56][57][58][59][60], the most common gynaecological cancers ([21][61][62]); however, other female cancers have not been studied, as shown in Figure 2. Although the presence and actions of the most commonly studied endogenous ligands (AEA, 2-AG, OEA, and PEA) in human reproductive tissues have been demonstrated, the presence and actions of others, such as SEA, virodhamine, stearamide, and monoolein [12][63][18][14][19][37][64][65][54][66][21][28][32][35][40][67][68][69][70][71][72][73][74] have not. Studies on receptor expression and function are few, and although there are some published studies on NAPE-PLD and FAAH expression and action in the female reproductive tract, many other (endo)cannabinoid metabolising enzymes have yet to be investigated, either in the normal female reproductive tract or in their related neoplasms (Figure 1 and Figure 2).
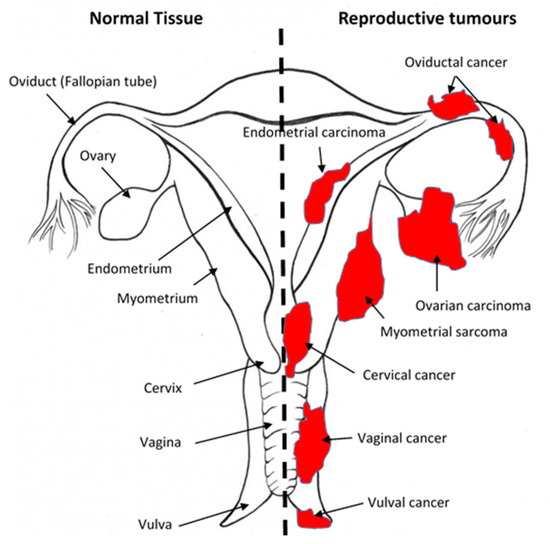
Figure 2. Sites of gynaecological cancers in the female reproductive tract. The diagram indicates the names of the normal tissues of the female reproductive tract (left side) and the sites and names of the cancers (right side) for the corresponding normal tissues. Please add copyright if necessary.
The main cannabinoids that are considered to have pharmaceutical promise in the treatment of cancer are the potent psychoactive and the commonly investigated non-psychoactive components of the Cannabis species, tetrahydrocannabinol (THC) and cannabidiol (CBD). Although there is scant evidence for their effectiveness in the treatment of gynaecological cancers, they are often promoted on medical cannabis production and distribution sites as having proven effectiveness [75][76][77][78][79][80]. Here, we examine the evidence in the scientific and clinical literature to support the current and future use of such compounds in the treatment of gynaecological cancers. These data are summarised in Table 1.
Among gynaecological cancers, those of the ovary have the highest morbidity and mortality rates [81]. In an attempt to establish if there is a possible role for the ECS in ovarian pathophysiology, we [14] studied the expression levels of different components of the ECS [20], and demonstrated expression of CB1, CB2, and the NAE-modulating enzymes NAPE-PLD and FAAH in normal human ovaries using immunohistochemistry [14]. Additionally, AEA concentrations in follicular fluid after ovarian stimulation by hormones (following an in vitro fertilisation protocol that caused an increase in follicle size) were directly correlated with follicle size, suggesting that AEA is indeed involved in the hormonal maturation of follicles and oocytes [14][20]. Furthermore, data exist to indicate that AEA, OEA, and PEA are all elevated in follicular fluids of ovarian cancer patients and women with ovarian cysts [22].
Bagavandoss and colleagues demonstrated CB1 and FAAH expression in ovarian surface epithelium, the site from which some ovarian cancers often arise, providing another clue for a possible involvement of the ECS in ovarian cancer [23]. Regarding the expression of CB1 in ovarian cancer, Messalli and coworkers [82] showed that CB1 expression was moderate in benign and borderline epithelial rat ovarian tumours, but was increased in invasive ovarian tumours, suggesting a correlation between the extent of expression of the ECS components and the prognosis for patients with more aggressive ovarian cancer [82]. The levels of lysophospholipids such as lysophosphatidylinositol (an endogenous GPR55 agonist) in blood and ascitic fluids were also found to be elevated in ovarian cancer patients compared to healthy controls, a finding associated with proliferation and the metastatic potential of ovarian cancer cells [83]. Hofman and colleagues [84] more recently found that elevated lysophosphatidylinositol levels in the ovarian cancer cell lines OVCAR-3, OVCAR-5, and COV-362 resulted in GPR55-dependent angiogenesis. Their conclusion was based on experiments where pharmacological inhibition and genetic deletion of GPR55 reduced the pro-angiogenic potential of lysophosphatidylinositol in these cell lines. Additionally, they demonstrated that the mitogen-activated protein kinase pathway triggered via GPR55 by phosphorylation of ERK1/2 and p38, which are signalling molecules known to be involved in proliferative and migratory responses, could be curtailed by chemical interventions [84]. This observation suggests that some ovarian cancers might be amenable to pharmaceutical intercession. In addition, other components of the endocannabinoid system are important here. For example, the 2-AG degrading enzyme MAGL has been shown to be upregulated in aggressive human ovary cancer cells [45], and it is also thought to be involved in oncogenic signalling and, hence, in increased migration, invasion, and survival of many other cancer cell types [85]. These data suggest that identification of an effective drug that targets the ECS to treat ovarian cancer may have applications in the treatment of other cancers too. The application of such therapies would need to be timely, because MAGL overexpression in non-aggressive cancer cells often results in tumours that subsequently exhibit an increased pathogenic phenotype [45]. Moreover, the application of an MAGL inhibitor led to a reversion of the enhanced pathogenicity [45]. Thus, the involvement of the ECS, and especially the 2-AG signalling pathways in ovarian cancer, may fuel expectations on new therapeutics to combat this and other types of cancer. Some preliminary evidence suggests that OEA and its structural analogues may also have a beneficial effect on inhibiting ovarian cancer growth, but these data need to be confirmed in vivo [41]. There is little evidence that plant-derived (phyto)cannabinoids have any effect on ovarian biology or ovarian cancer development or progression, a concept that came from a study where SKOV-3-derived tumours were grown on the chorioallantoic membrane of fertilised chicken eggs [86], and then were treated with CBD-containing nanoparticles. The data indicated that CBD caused a 1.35- to 1.50-fold reduction in tumour size depending on the type of CBD formulation used [86]. The authors indicated that these nanoparticle preparations might be useful in the treatment of peritoneal metastases of ovarian cancer, possibly with lower adverse drug effects [86]. Furthermore, the preparations also reduced SKOV-3 ovarian cancer cell numbers in vitro, to almost zero within 48 h, possibly making this a good candidate for a randomised clinical trial. Of course, many additional studies are required before any candidate CBD formulation can be used in such clinical trials.
Fallopian tube cancer is a relatively rare gynaecological cancer (Figure 2). It is often categorised as being part of ovarian cancer (especially as there is emerging evidence that most surface epithelial ovarian cancers maybe of fimbrial origin), but it is important to study it as a separate entity. Just like other parts of the female reproductive tract, the oviduct (Fallopian tube) expresses all the components of the ECS, with CB1 and FAAH expression intimately associated with proper oviductal function [19][25][26]. When dysfunctional, the risk of ectopic pregnancy is markedly increased [15][19][26][27][87]. There is little evidence on the effect of cannabinoids on human oviductal function, but in the murine oviduct [38], THC reduces fertility because of the increased number of ectopically implanting embryos. In the bovine oviduct, there is gradation of AEA, OEA, and PEA concentrations in the oviductal epithelial cells with low levels in the isthmus and significantly higher levels of OEA and PEA (but not AEA) in the ampulla at the same point of the oestrous cycle [88]. These levels significantly fluctuated during the oestrous cycle [88], as they do in the human oviduct (Fallopian tube) during the menstrual cycle and along its length [19], with OEA causing a reduction in epithelial cell cilia beat frequencies [87], an effect that is likely to prevent timely movement of fertilised oocytes and precipitate ectopic pregnancy [19]. Although possible relationships between the ECS, cannabinoids, and oviductal cancer currently do not exist (Table 1), the fact that dysregulation of the ECS in the fallopian tube is related to the development of ectopic pregnancy makes us speculate that there could be a role for the ECS in oviductal cancer, and that such a possibility deserves to be investigated.
Endometrial cancer, which is classified into type 1 and 2 [89], is the fourth most common cancer in women [90] and the most common gynaecological cancer. Various therapies exist depending on the disease grade and stage. Prognosis is poor, especially in those women with late presentation/detection [89]. Guida and coworkers [58] reported an upregulation of CB2 expression in endometrial cancer, whereby immunostaining was only successful in transformed malignant cells, while being completely absent in normal endometrial tissue. Furthermore, 2-AG levels were increased, but MAGL expression was decreased in comparison to controls, while AEA levels and FAAH expression were unaffected [58]. Similarly, Jove and colleagues [68] demonstrated that CB1 and CB2 were expressed at higher levels in stage III and IV endometrial carcinoma that has a poor prognosis. Unlike Guida and coworkers, the latter researchers found, by immunohistochemistry, an increase in CB1 expression, but no change in CB2 expression in stage 1 endometrial carcinoma tissue compared to normal endometrial tissue [68]. These observations were at odds with those of Risinger and coworkers, who found a decrease in CB1 receptor at the transcriptional level in stage 1 tissue [59]. These contradictory observations prompted us to investigate the ECS in endometrial cancer, using more than a single technique to interrogate CB1 and CB2 expression in endometrial cancer [55][56][57]. Our data indicated that CB1 and CB2 expression are decreased not only at the transcript level, but also at the protein level in both types 1 and 2 (stage 1) endometrial cancers (Table 1; Figure 3). We concluded that the discrepancy between these and previous studies was due to technical issues in the different methodologies used, including tissue sampling [56][57]. Furthermore, we examined the concentrations of plasma and tumour levels of AEA, OEA, and PEA in women with and without endometrial cancer, and showed that although the levels of all three N-acylethanolamines were increased in the tumours and in blood, only AEA and PEA were significantly higher in the plasma of such patients [91][53]. These data suggest that the differential catabolism of these three N-acylethanolamines might explain the different patterns of expression in endometrial cancer and plasma. We subsequently discovered that the apparent discrepancy between the tissue levels and plasma concentrations of OEA in the sample patient cohort was due to a decrease in the expression of FAAH in the tumour [66], without any change in the expression of NAPE-PLD (Table 1; Figure 3). The latter study also allowed us to define cut-off values for plasma AEA, OEA, and PEA concentrations (>1.36, >4.97, and 27.5 nM, respectively) that could be used in the prediction of endometrial cancer in symptomatic women [92], an observation that awaits confirmation in a larger, multicentre trial.
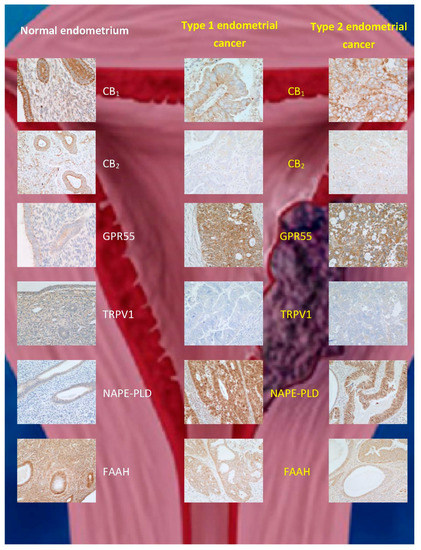
Figure 3. Immunohistochemical staining patterns for ECS proteins in normal endometrium and (type 1 and type 2) endometrial cancer. The data are taken from [58], where validation for the commercial antibodies and techniques used can be found. Note the reduction in CB1, CB2, TRPV1, and FAAH protein staining and increases for GPR55 and N-acylphosphatidylethanolamine-specific phospholipase D (NAPE-PLD) expression in both types of endometrial cancer when compared to that of normal tissue.
Cervical cancer is the second leading cause of malignancy-related deaths in women worldwide due to the lack of customisable and effective treatments (especially in low- and middle-income countries), with more than 250,000 deaths being reported annually [93]. A possible role of the ECS in the development of cervical cancer has been elucidated in recent years. Contassot and coworkers [48] reported a strong expression pattern of CB1 and CB2, as well as of TRPV1, in cervical carcinoma cell lines and biopsies. In addition, it was shown that AEA had a pro-apoptotic effect on cervical carcinoma cell lines (HeLa and Caski) [48], which were not inhibited, but were instead enhanced by CB1 and CB2 antagonists. On the other hand, the TRPV1 selective antagonist capsazepine protected the cell lines from AEA-induced apoptosis, indicating an important role of the TRPV1 channel in the pro-apoptotic action of AEA [94]. Additionally, it was demonstrated by Ramer and collaborators [95] that CBD decreased the invasiveness of cancer cells in a concentration-dependent manner. This effect was observed in the cervical cancer cell lines HeLa and C33A, as well as in the lung cancer cell line A549, and seemed to be mediated by the upregulation of TIMP-1 via CB1/CB2 and TRPV1. TIMP-1 is an inhibitor of matrix metalloproteinases, and as such, it prevents the movement of cells out of the tissue and, hence, a metastatic disease, as has been observed in a patient with ovarian cancer treated with CBD [96].
The activation of p38 and p42/44 mitogen-activated protein kinases was identified as an upstream event in TIMP-1 upregulation [95]. In agreement with these findings, it was reported that treatment of different cervical cancer cell lines (HeLa, SiHa, ME-180) with CBD led to a decrease in cell proliferation [97]. Furthermore, CBD induced cell death by the accumulation of cells in the sub-G0 phase (cell death phase) of the cell cycle, a finding that was most likely caspase-dependent because caspase-9 as well as caspase-3 were upregulated upon CBD treatment [97]. Hence, CBD may be an additional therapeutic tool for the treatment of cervical cancer, yet additional in vivo studies, similar to that performed on a single ovarian cancer patient [96], will be needed to clarify the impact of CBD on cervical cancer.
Vaginal cancer is uncommon, and the American Cancer Society estimated that >6000 women will be diagnosed with it in 2020. The estimated lifetime risk is 1 in 1100 (i.e., less than 0.1%). Of the 6000 USA women expected to be diagnosed with vaginal cancer in 2020, 1450 will die because they have this disease [98]. The role of the ECS in vaginal cancer has not been fully examined. We [47] have demonstrated that CB1 and FAAH are expressed in the normal vagina; however, there are no data on the expression of other components of the ECS (Table 1), nor on what their normal function might be. What happens to the expression of these factors or what effects cannabinoid and eCB ligands might have on the vagina or on cells of vaginal tumours is uncertain/unclear (Table 1). The internet is one source of information, and for the vagina, it is reported that some women experience a “vaginal high” when using cannabinoids, especially as a topical application [76]. The problem with these data is that only 40% of women experience this “psychological” effect [76]. Nevertheless, these statements have led some internet sites to suggest that different cannabis-containing preparations might be useful for the treatment of some of the symptoms associated with vaginal cancer [75][99]. Obviously, a lot more information is needed on the role of cannabinoids and eCBs in the human vagina, and especially in vaginal cancer.
Vulvar cancer is a less common gynaecological cancer [100]. The vulva is very similar to normal thin skin and is known to express CB1 and FAAH [47], but it is not known if it contains all the main components of the ECS (Table 1). The only existing evidence that cannabinoids have an effect on the vulva comes from a less-than-reliable internet source [78]. A C. sativa ethanolic extract and a purified CBD preparation had anti-inflammatory effects on keratinocytes and skin fibroblasts in vitro, suggesting that CBD was the main active ingredient that would be effective in wound injury [101].
This seems important because women with vulvar cancer often undergo radical surgery to remove their malignancy, which causes disfiguration of the female external genitalia, and causes significant long-term emotional and physical instability [102]. Indeed, the use of the CBD derivative VCE-004.3 on skin fibrosis and inflammation [103] demonstrated a CB2/PPARγ-dependent effect, and suggested that similar compounds might be beneficial for patients with vulvar cancer who have undergone surgery and need topical treatment for the pruritus; the latter is associated with skin fibrosis and inflammation, especially as VCE-004.3 appears to inhibit mast cell degranulation [103]. The toxicity profile of such topical administrations remains to be determined; however, ethanolic extracts of THC, CBD, and other cannabinoids appear in the blood shortly after administration; thus, some caution is advised, also in the light of the pleiotropic effects of these compounds [104]. Obviously, more detailed analysis of the role of the ECS and of plant-derived cannabinoids in the treatment of vulvar cancer is warranted.
The function of the female reproductive tract is to support the embryo and fetus during its development into an independent offspring (Figure 1). In order to do this, the coordinated actions of many interacting factors need to take place, of which the ECS is an integral part [28][35][39][105][106][107][108]. A key tissue in human reproduction is the fetoplacental unit. The entire ECS is present in the placenta [108] (see also Table 1), and modifications of its components result in obstetrical problems, such as miscarriage [109][65][46][110], babies that are small for gestational age [111][112], and pre-eclampsia [113]. In addition, dysregulated N-acylethanolamine levels may be responsible for preterm delivery [72][74]. The placenta can also undergo neoplastic changes into two clinically relevant conditions, hydatiform mole (a non-malignant transformation) and choriocarcinoma (a malignant transformation), which appear noteworthy. Currently, there are no data on the expression of the ECS in either of these tumours; there is, however, evidence that AEA and THC both affect a model for choriocarcinoma, like BeWo cells [114][115][116][117], and a model for normal trophoblast, like TCL-1 cells [118], where cell growth is affected mainly through a CB2-dependent mechanism [116][117][118]. These observations, coupled with evidence that THC decreases STAT3 signalling in mice with reduced fetus numbers and placental weights [111], support the view that cannabinoid use in human pregnancy is likely to affect the placenta in a similarly dangerous manner [119]. The increased use of CBD in pregnancy as an anti-emetic [120][121] is thus of great concern because the toxicity profile of CBD in pregnancy is not fully known [120][122][123], and especially as CBD can inactivate both placental CB1 and CB2 receptors in vitro [124].