1000/1000
Hot
Most Recent

Plant non coding RNA review paper highlight the current knowledge of plant microRNAs, siRNAs, and lncRNAs, focussing on their origin, biogenesis, mode of actions, and their fundamental roles in plant response to abiotic stresses.
While up to 90% of the eukaryotic genome is transcribed into RNA, only about 2% of transcribed RNAs generate protein products [1][2]. The remaining transcriptome comprises ncRNAs transcripts, which were previously considered transcriptional noise owing to the absence of protein-coding ability and poorly conserved sequences [1][3]. However, high-throughput sequencing analysis and experimental validation have confirmed the involvement of ncRNAs in a myriad of distinct gene regulatory levels, including epigenetic, transcriptional, and post-transcriptional [4].
Since their initial discovery, ncRNAs have been categorized into several distinct classes based on their origin, biogenesis, and mechanism of action. Housekeeping and regulatory ncRNAs are the major groups of ncRNA transcripts. The housekeeping ncRNAs primarily deals with cellular and ribosomal functions. They include transfer RNAs (tRNAs), small nuclear RNAs (snRNAs), ribosomal RNAs (rRNAs), and small nucleolar RNAs (snoRNAs) (Figure 1). On the other hand, the regulatory ncRNAs consist of microRNAs (miRNAs), short interfering RNAs (siRNAs), piwi-interacting RNAs (piRNAs), and long non-coding RNAs (lncRNAs) [5], (Figure 1).
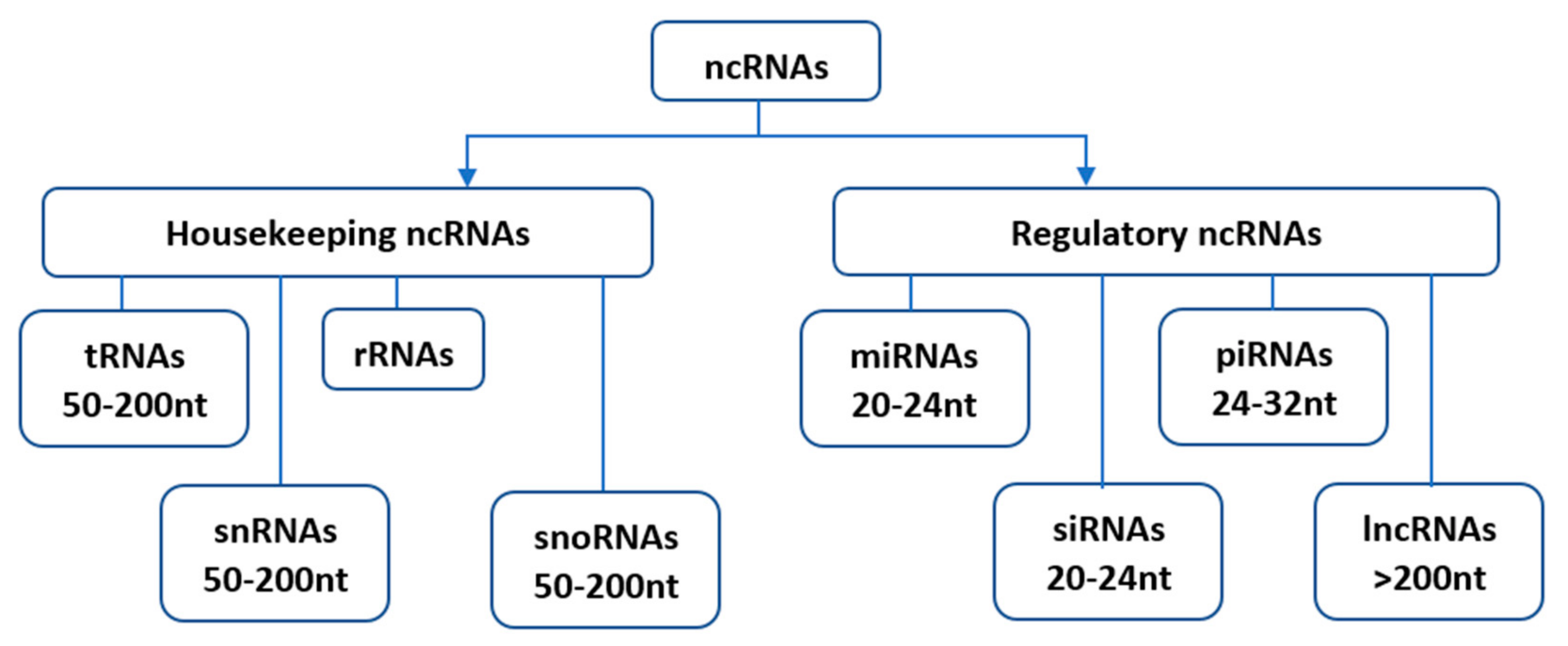
Figure 1. Classification of non-coding RNAs (ncRNAs). Housekeeping ncRNAs include; tRNAs- transfer RNAs, snRNAs-small nuclear RNAs, rRNAs-ribosomal RNAs, snoRNAs-small nucleolar RNAs. The regulatory ncRNAs consist of miRNAs-microRNAs, siRNAs-short interfering RNAs, piRNAs-piwi-interacting RNAs, and lncRNAs-long non-coding RNAs.
MicroRNAs are small but fundamental molecules which are predominantly 20–22 nt in length [6]. They are synthesized from miRNA genes (MIR genes), which are processed by RNA polymerase II (RNA pol II), RNase III Dicer-like protein 1 (DCL1) [7] (Figure 2). HASTY (HST) transports the resulting miRNA–miRNA* duplex to the cytoplasm where it is integrated into the RNA-induced silencing complex (RISC) containing ARGONAUTE 1 (AGO1) proteins that control gene silencing by mRNA cleavage and translational repression [8], (Figure 2).
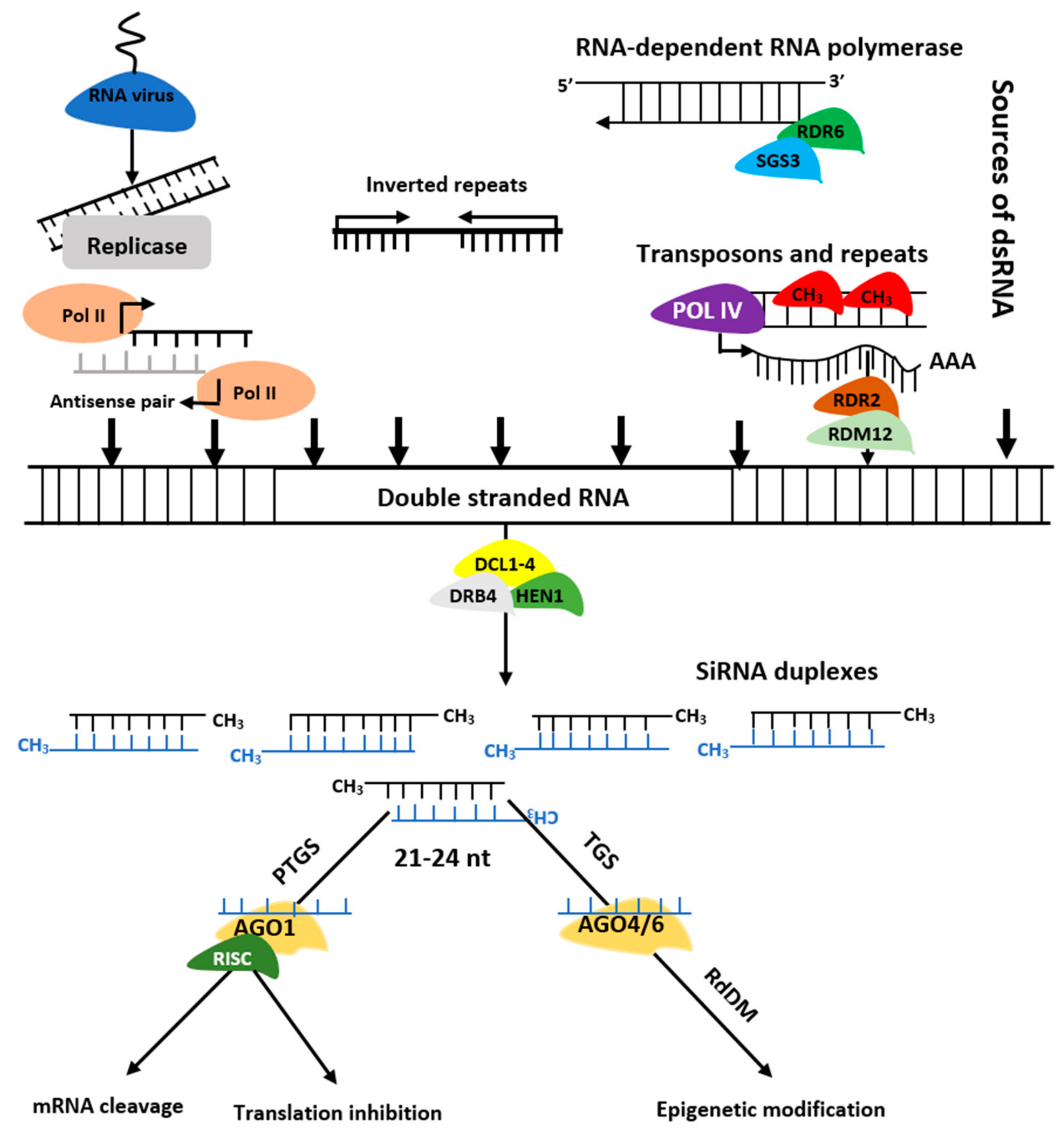
Figure 2. Biogenesis of plant miRNAs. In the nucleus, MIR genes are processed to miRNA/miRNA* duplexes through the action of Pol II, DCL1, SE, HYL1, HEN1, and HASTY. In the cytoplasm, the duplex is incorporated with RISC-AGO complex to which guides it towards the target, resulting in suppression of the translation and cleavage of mRNAs.
The siRNAs are grouped into trans-acting siRNAs (tasiRNA), heterochromatic siRNAs (hc-siRNA), and natural antisense siRNAs (nat-siRNAs) depending on the mode of action as well as biogenesis. The siRNAs biogenesis involves dsRNAs, DCL enzymes activities, and RISC containing AGO protein that controls target regulation at post-transcription or transcriptional level [9] (Figure 3). Biogenesis of ta-siRNA is dependent on SUPPRESSOR OF GENE SILENCING3 (SGS3), RNA-dependent RNA polymerases 6 (RDR6), and DCL4 [10] while that of nat-siRNAs depends on SGS3, RDR6, DCL1, DCL2, and a plant-specific RNA polymerase, NRPD1A [11]. Similarly, hc-siRNAs are generated by Pol IV, DCL3, RDR2, 5S rRNA genes, and NRPD1A [12].

Figure 3. Biogenesis of siRNAs. The double stranded RNA (dsRNAs) is transformed into siRNAs by DCL, HEN1, and DRB. The RISC-AGO complex then guides the selected strands of siRNA duplexes to post transcription gene silencing (PTGS) or transcription gene silencing (TGS).
Long non-coding RNAs are transcripts that exceed more than 200 base pair (bp) in length and cannot be translated into protein [13]. They are classified into cis-natural antisense (cis-NATs), trans-natural antisense transcripts (trans-NATs), and pseudogenes [14], sense or antisense (strand of origin) [15], divergent, or convergent (orientation of transcription), and as intronic or intergenic (location) [16] (Figure 4). They are transcribed by RNA pol II, III, IV, and V and control target regulation via multiple ways, including chromatin remodeling, transcriptional repression, mimicry, RNA splicing and transcriptional enhancer (Figure 4).
Figure 4. The biogenesis of long-non-coding RNAs (lncRNAs) and their gene regulation mechanisms in plants. (i) The transcripts of lncRNA (red box) are classified on the basis of their genomic location and in relation to the nearest gene (blue box): (A) sense lncRNAs are transcribed on the same strand as an exon; (B) antisense lncRNAs are transcribed on the opposite strand of an exon; (C) intronic lncRNAs are transcribed on the intron; (D) intergenic lncRNAs are located between two distinct genes; (E) enhancer lncRNAs emerge from an enhancer region of protein-coding genes. (ii) The gene regulation pathways induced in plants by lncRNAs. LncRNAs regulate gene expression either by: (1) interacting with transcriptional activator leading to gene activation; (2) interacting with transcriptional repressor thereby suppressing transcription; (3) controlling RNA splicing by interacting with splicing factor or binding premRNA splicing junction; (4) recruiting chromatin remodeling complex such as PRC to regulate gene expression in the promoter region; (5) LncRNA mimics miRNAs by occupying their target sites on the mRNA.
For miRNAs, the MIR genes are processed by DCL1, HYL1 and SE complex into mature miRNA–miRNA* duplex and transported by HASTY protein to the cytoplasm where it is integrated with RISC containing AGO1. The RISC-AGO1 complex binds to the complementary sites of sense sequence on its targeted RNA transcript and degrade it. In contrast, the antisense strand of miRNAs remains in the RISC [17]. In siRNAs, one strand which is integrated with RISC-AGO1 or AGO7 complex is guided to cleave the transcripts of the target gene at 10–11 nt upstream of the 5′ end of the antisense strand [18]. The EXONUCLEASE 4 (XRN4) enzyme then degrade both the 3′ and 5′ cleaved fragments [19]. Recent studies have reported the RNA binding and target slicer activities of AGO2, AGO4, and AGO10 in plants, indicating the complexity of the sRNA-induced gene silencing mechanism [20].
The RISC-AGO1 complex suppresses translation by binding to the 5′ untranslated region (UTR) or open reading frame (ORF) of the target gene, thereby restricting ribosome recruitment or movement [21]. This translation inhibition process is regulated by other factors such as ALTERED MERISTEM PROGRAM 1 (AMP1), VARICOSE (VCS), GW-repeat protein, and microtubule enzyme KATANIN (KTN1) [22]. Despite the involvement of both AGO1 and AGO10, the specific function of each AGO gene in this inhibition process is still not clear [22].
The DCL family has multiple copies in Arabidopsis, which contribute to the biogenesis of divergent lengths of sRNA. DCL1 is involved in the conversion of partially paired dsRNAs precursors into 21-nt mature miRNAs [23]. On the other hand, DCL2 and DCL44 initiate the generation of 20–22nt siRNAs from perfect complementary dsRNAs precursors [24]. DCL3 produces 24-nt siRNAs (hc-siRNA) that typically silence gene expression via the RdDM pathway [25]. These hc-siRNAs are transcribed at the heterochromatic regions where they stimulate cytosine methylation in the sequence contexts of CG, CHG, and CHH, in cis [26]. The DCL3-dependent miRNAs integrate with AGO4 forming a complex which repress gene silencing by cytosine and histone methylation [27]. However, the de novo hc-siRNA-induced RdDM process integrates the activities of RDR, DCL, AGO, and Pol IV and V which transcribe the double-stranded precursors and promote methylation at the target sites respectively. Therefore, DNA and lysine methylation at the ninth position of histone H3 (H3K9) induces systemic silencing, while the hc-siRNA/AGO4 RISC guides DNA and H3K9 methyltransferases to the target sequence for transcriptional gene silencing [26].
Plant lncRNAs function in cis and trans. The cis-acting lncRNAs function near their synthesis sites and operate directly on the local nucleotide sequences or chromosome regions on one or more contiguous genes. The trans-acting lncRNAs, on the other hand, disperse from the synthesis site and can function on several genes at great distances, including on different chromosomes [28]. Moreover, lncRNAs can also be transcribed as sRNAs precursors [29], miRNA target mimics where the lncRNAs competes with the target mRNA for miRNA binding [30], protein-protein interactions, modification, localization [31], and via epigenetic regulation mechanisms such as DNA methylation, histone modification, and chromatin remodeling [32].
Plants have developed various mechanisms to adjust their growth and development during environmental stress conditions. Multiple research findings have recently shown the differential expression of ncRNAs in such unfavourable conditions [33]. These ncRNAs either control gene expression in related cellular networks or function directly in response to stress [34]. miRNAs have become new targets for improving plant productivity and abiotic stress tolerance due to the broad functions of their targets [35]. The regulation of miRNAs appears to rely on their roles on the abiotic stress response. Stress-up-regulated miRNAs might down-regulate their target genes, which might be negative regulators of stress tolerance (e.g. stress-responsive gene repressors) [36]. Moreover, the down-regulation of miRNAs under stress might accumulate their target gene mRNAs, which might positively regulate stress tolerance [36]. Moreover, various siRNAs and lncRNAs have been reported in response to multiple abiotic stress [33]. All these ncRNAs have been reported to execute their functions in responding to abiotic stresses in different ways.