1000/1000
Hot
Most Recent

Neurofilament light chain (NfL) is a neuron specific structural protein which can be detected in the blood (serum and plasma). Elevate blood levels may serve as an important surrogate neuro-axonal injury in a variety of neurological conditions, already showing promising associations with outcomes of interest.
Neurofilaments are neuronal-specific heteropolymers conventionally considered to consist of a triplet of light (NfL), medium (NfM) and heavy (NfH) chains according to their molecular mass [1]. More recent discoveries show that α-Internexin in the central nervous system [2] and peripherin in the peripheral nervous system [3] can also be included in neurofilament heteropolymers. These five proteins co-assemble into the 10 nM intermediate filaments in different combinations and concentrations depending on the type of neuron, location in the axon and stage of development [4].
Each of the neurofilament proteins consists of an amino-terminal domain that is thought to regulate the formation of oligomers [5], a central helical rod domain, and a variable carboxy-terminal domain. The chain-specific C-terminal domains are the main determinants of differences in molecular mass and phosphorylation between subunits. Following synthesis and assembly in the neuron cell body, tetramers of neurofilament proteins are transported bidirectionally along axons by the microtubular apparatus prior to forming a continuously overlapping array that runs parallel to axons. Once formed, in the healthy state, they are remarkably stable for months to years [6].
In mature myelinated axons, neurofilaments are the single most abundant protein [7]. They perform key roles as part of the neuroaxonal scaffold to resist external pressures, determine axonal diameter, indirectly moderate conduction velocity, and act as an attachment for organelles and other proteins [4]. Beyond their primary structural role in axons, mounting evidence indicates that a unique pool of synaptic neurofilament proteins serves dynamic functions beyond static structural support [8]. Changes in neurofilament phosphorylation may be involved in long term potentiation that underpins memory [9] and NMDA receptor stability is dependent on a synaptic scaffold of neurofilament proteins.
Damage to central nervous system (CNS) neurons and physiologic turnover causes neurofilament release. This translates to elevated levels in the cerebrospinal fluid (CSF) and eventually blood, where the concentration reflects the rate of release from neurons (Figure 1, where we focus on NfL). Physiologic degradation of neurofilaments within neurons is proposed to be a combination of ubiquitin-mediated proteasomal and apophagocytotic pathways [10]. Based on the trafficking of other proteins degraded in the CNS, it is likely that partially degraded fragments of neurofilaments drain directly into CSF and blood via multiple routes. These include direct drainage into CSF and blood via arachnoid granulations as well as lymphatic drainage into the subarachnoid spaces and perivascular spaces [11][12]. Several studies have demonstrated strong correlations between blood and CSF NfL, with r values typically ranging from 0.7 to 8 (e.g., [13]). However, our understanding of the kinetics of neurofilament release, distribution and metabolism is incomplete.
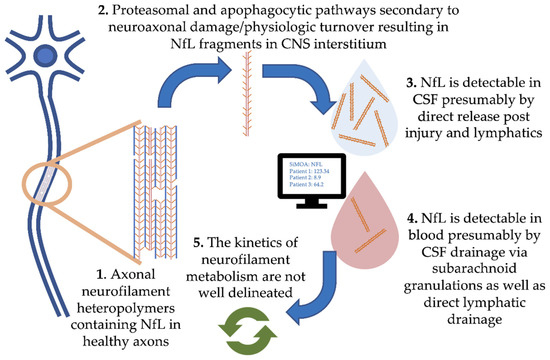
Figure 1. Pathophysiology of neurofilament light chain in blood and cerebrospinal fluid (CSF).
Blood–brain barrier permeability itself may be a confounder; neurofilament quotient in blood compared to CSF could be selectively increased following periods of inflammation such as that seen in MS relapse, positively skewing blood NfL levels. Two recent studies on this topic in MS patients present conflicting results [14][15].
Once NfL enters the blood, the half-life is a key consideration with implications on the frequency of disease activity monitoring. In a longitudinal study of NfL levels before and after intrathecal catheter insertion, NfL in both CSF and serum peaked at 1-month post-surgery, returning to baseline after 6 to 9 months [16]. In longitudinally sampled MS patients around the time of relapse, levels increasing 5 months before, peaking at clinical onset, and recovery within 4–5 months [17]. Therefore, quarterly measurement is likely sufficient, a frequency that our group is currently investigating in longitudinal prospective studies of serum NfL.
Age is the principal physiologic covariate of NfL levels. Levels in healthy controls increase by 2.2% per year [18][19]. Furthermore, an inflection point is observable above the age of 60, after which both sNfL levels, as well as the inter-individual variability in levels, increase greatly [20]. It is speculated that these changes are attributable to both aging itself as well as the accumulation of subclinical comorbidities. Other factors outside of neurological disease itself that may alter neurofilament levels include BMI [21] as well as vascular risk factors [22]
Although the primary focus of this review is the pathophysiologic relevance of NfL concentrations as they relate to neurological diseases such as MS, the vital role of neurofilaments is underlined by various human mutations that interfere with their function and homeostasis. Mutations of gigaxonin, a key component in the ubiquitin-dependent intermediate filament degradation, results in the pathological aggregation of neurofilaments in neurons and a severe neurodegenerative condition called giant axonal neuropathy [23]. Mutations in the neurofilament light chain gene itself result in axonal forms of hereditary motor sensory polyneuropathy [24] and variants of the heavy neurofilament subunit are associated with the development of amyotrophic lateral sclerosis [25].
Of the family of neurofilament proteins, neurofilament light chain (NfL) has gained the most interest as a candidate marker of outcomes in neurological diseases. This was not without contention. While the neurofilament light chain is the most abundant and soluble of the neurofilament proteins, phosphorylated neurofilament heavy chain (NfH) was initially thought to be more resistant to protease activity [26][27][28]. NfL was thought to be unstable in vitro [29] and initial research focused on NfH quantified by enzyme-linked immunosorbent assay (ELISA) or electrochemiluminescence (ECL) as a biomarker of axonal damage in MS [30][31]. However, in 2013, a comparative study of NfL and NfH found both proteins showed equivalent stability after several days at room temperature and through freeze–thaw cycles [32]. Moreover, although some of the differences observed may correspond to analytical methodologies, this study found that NfL levels were higher than NfH and NfL was a better discriminator of MS patients from controls.
Initial studies looking at NfL in association with neurological disease outcomes focused on CSF measurements. Although CSF is “closer” to the CNS pathologies (e.g., MS) and NfL concentration is approximately 500-fold higher, the inconvenient and invasive lumbar puncture required severely limits its clinical utility as a frequent serial biomarker. Concentrations in the blood however were too low to be reliably measured with conventional immunoassays such as ELISA or ECL assays. It was not until recently, with the development of the Single-Molecule Assay (SiMoA), that analytical methods become sufficiently sensitive to measure the single-digit picogram per milliliter levels present in blood [33]. This SiMoA technology, similar to other immunoassays, is based on fluorescent microbeads coated with high-affinity capture antibodies that bind NfL followed secondly by a fluorescently labelled detector antibody [34]. The increased analytical sensitivity of the SiMoA assay is due to its unique method of detection. Assay beads with captured NfL and fluorescent detector antibody, are loaded onto an assay disk containing >200,000 microwells capable of holding only a single bead. At high analyte concentration, the total fluorescence can be captured in the traditional manner (analog) and correlated to the analyte concentration. At low analyte concentrations, rather than detecting total fluorescence, a digital image is captured that enumerates individual fluorescent microwells in a binary fashion, effectively lowering the limit of quantitation to the femtomolar range. Although there are several neurofilament assays in development based on other technologies, including widely used chemiluminescent-based assay [35], the SiMoA platform has enabled most of the most recent insights.
Measurement of a convenient objective blood marker of neuronal injury in patients is an appealing prospect for neurologists. Analogous to the cardiologist’s troponin, neurofilament light chain can be detected in the blood at elevated levels in a variety of neurological disease state in the central and peripheral nervous system causing neuroaxonal injury and can be followed longitudinally. These include neurodegenerative conditions, stroke, traumatic brain injury, peripheral demyelination and multiple sclerosis [36][37]. Although not generally considered a marker of diagnostic utility, levels in amyotrophic lateral sclerosis (ALS) are so discrepantly elevated that they may be useful in this setting [38]. In many of these conditions [39], the current rate of neuronal loss is predictive of future losses, and as such NfL may be of prognostic utility. NfL has been most intensively studied in patients suffering from Multiple Sclerosis. Here there is a great unmet need for improved prediction and disease monitoring largely due to availability of a range disease modifying treatments which when employed appropriately can greatly reduce the morbidity associated with this condition. Many are optimistic that NfL could represent the first of its kind in neurology: a broadly-applicable protein biomarker that objectively reflects underlying pathology which can be harnessed to improve patient outcomes. Important future work includes the establishment of age-adjusted normative datasets and cutoff values so that physicians can better interpret individual patient results.