1000/1000
Hot
Most Recent

The current review comprehensively covers major research to evaluate the effectiveness of PGPR in alleviating crop water stress and to find effective PGPR to help crops in maintaining water status under drought conditions. The aim of the present review is to provide insights into the role of phytohormones, plant metabolites, exopolysaccharides (EPS), and 1-aminocyclopropane-1-caroboylic acid (ACC) deaminase activity in stress tolerance of plants in response to PGPR inoculation. This review identifies the challenges of drought stress and involvement of PGPR in the mitigation of drought stress in plants for sustainable production.
Desertification, drought, and land degradation are major challenges to sustainable crop production throughout the world especially in developed countries. Water scarcity mainly due to low annual precipitation is very damaging for plant growth, and ultimately sustainable crop production. However, there is an inordinate need to use these areas even with marginal productivity due to damage to basic farmlands. Therefore, there is more interest in producing crops using low or marginal yields of soil (e.g., sandy soil) [1]. However, sandy soil has high temperatures and suffers severe drought. Stress losses can range from 50% to 80%, depending on the stress period and type of plant species [2]. Drought stress in desert areas affects plant water potential, restricts the normal plant performance,[3], and alters the plant physiological and morphological characteristics [4][5]. Drought stress-induced plant growth was studied in wheat [6], barley [7], rice, and corn[8]. Moisture content and plant biomass are common growth factors impacted by drought stress in these plants [9]. Besides, drought stress stimulus negatively impacts the nutrient uptake and translocation as the soil nutrients are transferred to the roots via water.
Consequently, drought stress reduces the absorption of nutrient and mass-flux of water-soluble nutrients, for example, calcium, nitrate, sulfate, silicon, and magnesium[10]. Drought stress enhances formation of free radicals that damage plant defence system resulting in an increase in reactive oxygen species (ROS), such as superoxide radicals, hydroxyl radicals, and hydrogen peroxide induces oxidative stress. ROS can cause tissue damage, to membrane corrosion, proteins and nucleic acids by causing their lipid peroxidation [11][12][13].
Water stress is responsible for high economic losses in arid and semi-arid regions. It disturbs plant–water relations at cellular and whole plant levels, resulting in specific and non-specific responses [14]. Plant reaction to water stress is a complex process that tends to include polyamine formation and a collection of novel proteins with relatively unknown functions. Drought decreases the photosynthesis supply of carbon dioxide, which may contribute to ROS production from misguided electrons in the camera system[15][16]. It also creates free radicals during abiotic tension. ROS, such as radical superoxide (O2−), radical hydroxyl (OH), and hydrogen peroxide, enhance the damaging effect of lipid peroxidation throughout the membrane[17]. Plants have an antioxidant defensive system which prevents cellular membranes and DNA from ROS-induced oxidative damage by converting ROS into non-toxic forms such as water and oxygen[18][19].
Inoculation of plants with growth-promoting microorganisms can improve water retention strategies and drought tolerance of plants grown in arid or semi-arid regions [20]. These useful microbes inhabit the rhizosphere/endogenous rhizosphere of the plant through various direct-indirect mechanisms and promote plant growth (Figure 1). The rhizosphere is a thin layer of soil surrounding the roots of the plant and is a very critical and active area of root activity and metabolism[21][22][23]. A significant number of microorganisms coexist in the rhizosphere, such as bacteria, fungi, protozoa, and algae, but mostly different types of bacteria. Plants release organic compounds through exudate to select the bacteria that contribute most to the plant’s health under stressful conditions [24]. The beneficial relationships of plant-microbes in the rhizosphere are the key determinants in water conservation, soil productivity, and plant health. Plant growth-promoting rhizobacteria (PGPR) affect growth, yield, and nutrient uptake through a series of mechanisms. Some strains (e.g., Azospirillum brasilense, Aeromonas punctata, Bacillus megaterium, Pseudomonas fluorescens, Serratia marcescens) directly modulate plant physiology by stimulating the production of plant hormones, while others upturn minerals and nitrogen in the soil as a means of increasing growth under water-deficient conditions[25][26][27][28].
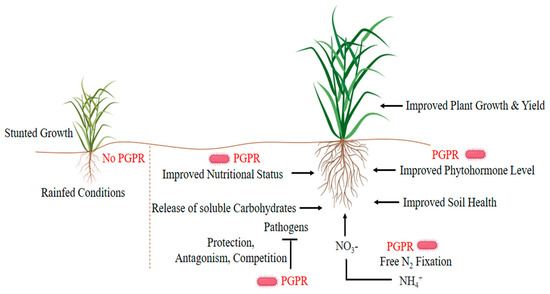
Figure 1. Plant growth-promoting strategies of plant growth-promoting rhizobacteria (PGPR) under drought stress.
A species may have a complementary set of survival strategies enabling it to survive under small and unpredictable distribution of rain[29]. Desert plants may have no water for many years. Plants exhibit different responses when sensing abiotic stimuli, which are related to specific stress-tolerance mechanisms[30][31][32]. A series of epidermis waxes protect plants from excessive moisture loss and provide protection against various pathogenic antagonistic activities [33]. In addition, osmoprotectants like proline accumulation aid in sustaining the plant’s water potential, and promotes the plant’s extraction of water from the soil[34]. Changes in primary metabolism are considered to be the most obvious of all metabolic reactions and comprise changes in the level of sugar/sugar alcohol, amino acid, and tricarboxylic acid cycle intermediates, exhibiting a common tendency for ecological stress reactions. However, changes in secondary metabolism are exact to particular stress and are precise to the type of plant species[35][36].
Some of the metabolic compounds that are associated with abiotic stresses and act as protectants include the sorbitol, polyols, mannitol, sucrose, fructan, proline, and ectoine[37]. Other small molecules such as carotenoids, ascorbic acid, tocopherols and anthocyanins also protect plants from being subjected to oxidative injury and protect plants by eliminating stress-induced ROS in plants. The production of phytoalexins and initiation of phenylpropanoid pathways and lignin biosynthesis are related to plant defence mechanisms[38][39][40]. Plant molecules such as salicylic acid, jasmonic acid, methyl salicylate, and methyl jasmonate are formed under stress. They can also act as signalling molecules that trigger defences against various biotic and abiotic stresses in crop plants [41]. In recent years, metabolomics has been used for various purposes, such as (1) assessing the effect of various stresses in plants; (2) pursuing the contribution of specific compounds in a specific biosynthetic or secondary deprivation pathway and (3) organizing various plant samples[42]. Stability, defence, and signalling of metabolites can be used to measure the degree of plant lenience to diverse abiotic stresses [43][44]. Extensive research is carried out to develop policies against drought stress by growing drought-tolerant crops, improving crop calendars and resource management practices [45].