1000/1000
Hot
Most Recent

Status epilepticus (SE) is a major medical condition that is associated with poor outcome in approximately 50% of cases, despite the use of conventional anticonvulsive treatments. The age of the patients, a previous history of epilepsy, SE refractoriness, and a primary cerebral insult as the cause of SE have been identified as independent predictors of poor outcom
Secondary brain insults refer to a cascade of cellular, biochemical, tissue, or blood vessel changes that occur over a period ranging from minutes to days after a primary brain injury caused by various conditions, such as traumatic brain injury, stroke, or subarachnoid hemorrhage, ultimately resulting in additional brain tissue damage [1][2][3][4][5]. The common denominator for all factors of secondary brain insults is cerebral blood flow (CBF) which can be approximated by monitoring cerebral perfusion pressure (CPP) [6]. Several factors can affect CBF: flow-metabolism coupling and cerebral autoregulation/mean arterial blood pressure, arterial partial pressure of carbon dioxide (PaCO2), arterial partial pressure of oxygen (PaO2), temperature, natremia and osmolality, glycemia, and hemoglobinemia. The occurrence of these irreversible secondary brain lesions has an impact on outcome, not only in terms of mortality, but also on longer-term functional neurological impairment [7][8][9][10][11][12].
In SE, an experimental study performed on baboons exposed to prolonged epileptic seizures showed physiological changes to be characterized by a two-step phenomenon. First, animals demonstrated adaptive adrenergic activation as a mechanism to protect the brain by increasing CBF and thus improving oxygen and glucose delivery during the acute phase of seizure activity. Second, the animals demonstrated an inadequate decrease in CBF and a deleterious reduction of oxygen and glucose delivery to the brain as the seizures continued, associated with several systemic factors that correspond to secondary brain insults, also reported in primary brain injury, from other causes: mild arterial hypotension, acidosis, hypoxia, hyperpyrexia, and severe hypoglycemia [13][14][15].
Among the 2209 articles responding to our search strategy, 56 were included in this systematic review (Figure 1). Study results covered experimental data showing the interplay between various physiologic derangements indistinguishable from the effects of ongoing seizure activity and from the effects of physiology on seizures, and the resulting secondary brain insults. The only clinical data were covered by a randomized controlled trial focusing on the effect of temperature.
Figure 2 illustrates the synthesis of the evidence of secondary brain insults in experimental status epilepticus.
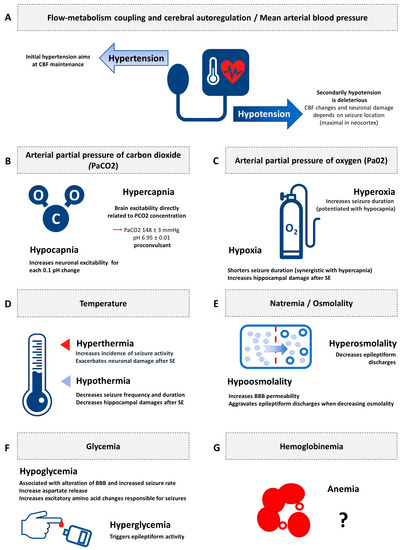
Figure 2. Synthesis of the evidence of secondary brain insults in experimental status epilepticus. CBF: cerebral blood flow; PaCO2: arterial partial pressure of carbon dioxide; PaO2: arterial partial pressure of oxygen; BBB: blood brain barrier. (A) Flow-metabolism coupling and cerebral autoregulation/Mean arterial blood pressure; (B) Arterial partial pressure of carbon dioxide (PaCO2); (C) Arterial partial pressure of oxygen (Pa02); (D) Temperature; (E) Natremia/Osmolality; (F) Glycemia; (G) Hemoglobinemia.
The natural course of SE is characterized by an initial phase of adrenergic activation [17][18], resulting in an increase in CBF, characterized by the association of arterial blood pressure hypertension and a decrease in cerebrovascular resistance. [19][20][21] The second phase consists of a progressive decrease in arterial blood pressure, and finally, hemodynamic failure, responsible for a deleterious drop in CBF [15][20][22].
Hypocapnia is routinely used as an activation test during electroencephalogram recording [23]. Several clinical studies reported an association between respiratory alkalosis and seizure activity [24][25][26][27][28], potentiated by tapering antiepileptic drugs [28]. Paradoxically, in a large retrospective study, a clinical seizure was elicited after 5 min of hyperventilation in only 2/433 (0.5%) of hospitalized patients monitored by long-term video-EEG in an epilepsy unit [24]. However, various experimental findings support the hypothesis of enhancing neuronal excitability after the induction of hypocapnia [23]. In a rodent model of epilepsy, hypocapnia increased ictal activity (population spike amplitude, spreading depression-like response, postsynaptic neurons electrical excitability), which varied for each decrease in pH of 0.1 [29]. Seizure duration was demonstrated to be inversely related to PaCO2 levels in another study of dogs receiving electroconvulsive therapy [30].
Studies to assess the pro- or anti-convulsant properties of hypercapnia yielded conflicting results. An increase in PaCO2 was associated with the stimulation of inhibitory interneurons which halted seizure activity in several experimental studies [31][32]. However, seizure activity has been associated with respiratory acidosis in human [17][33], and some experimental data support the hypothesis of carbon dioxide neurotoxicity. Woodbury et al. [34] and Withrow et al. [35] reported seizure activity in all rats exposed to an atmosphere containing 45% CO2 and 90% of adult rats exposed to 30% CO2 for 8 min, respectively. The relationship between the CO2 concentration in inspired air, plasma PaCO2, and brain excitability was demonstrated by Brodie et al. Indeed, rats exposed to an atmosphere containing 30% CO2 (PaCO2 148 ± 3 mmHg, pH 6.95 ± 0.01) exhibited intermittent, repetitive, clonic seizures, whereas rats exposed to concentrations of 50% CO2 (PaCO2 264 ± 48 mmHg, pH 6.72 ± 0.02) did not exhibit seizures but appeared to be anesthetized [34]. The effect of CO2 on brain excitability appears to be directly related to the concentration inhaled, thus the PaCO2 and the level of acidosis. The underlying mechanism of seizures in hypercapnia has yet to be completely understood. In an experimental setting, Katsura et al. induced cerebral ischemia by elevating the CO2 concentration in inhaled gas, reaching PaCO2 values of 300 mmHg. Protein kinase systems have been suggested to be involved in neurotransmission and affected by acidosis. This is one potential mechanism that may induce seizure activity during hypercapnia, but there are no data to support this hypothesis [36].
Hypoxia has been shown to shorten the duration of seizures produced during kainic acid-induced SE, but was associated with higher hippocampal neuronal toxicity, illustrating the potentiation of neuronal damage by hypoxia during long-lasting seizures [37]. Another study found a synergistic association between hypoxemia and hypercapnia to shorten seizure duration [38]. An interventional therapeutic approach was evaluated by Wasterlain et al., who tested the interest of mechanical ventilation and muscular blockade in 10 rats receiving repetitive convulsive electroshocks. They found that the correction of hypoxemia by supportive techniques markedly diminished mortality relative to a control group of hypoxemic rats [39]. However, the correction of hypoxia alone was not sufficient to avoid neuronal toxicity, since this damage can result from SE per se, with or without its association with tissue hypoxia [40]. Finally, preconditioning hypoxemia was the only situation found with a favorable outcome. Application of preconditioning hypoxia to rats with kainic acid-induced SE demonstrated adaptive properties, leading to better control of seizure activity [41] and neuroprotection by decreasing brain edema [42].
Hyperoxia-induced seizures are a well-known complication, in particular during hyperbaric oxygen administration [43][44]. However, demonstration of neuronal toxicity of hyperoxia in normobaric conditions of administration in the context of SE is still a subject of debate. In animals, as in humans, hyperoxia increases seizure duration, especially as it is associated with hypocapnia [30][38]. Given these findings, epileptic rats mechanically ventilated with 100% oxygen, surprisingly, did not demonstrate neuronal damage that was different from that of normoxic epileptic animals [45][46].
Fever is the most studied cause of secondary brain insult in SE [47]. Indeed, hyperthermia between 39.5 and 42 °C was associated with an increased incidence of seizure activity and exacerbated neuronal damage in studies of rats with self-sustaining SE [48][49][50]. Conversely, mouse models exposed to SE and subsequently treated showed a strengthening of the anesthetic’s antiepileptic activity after induction of hypothermia, characterized by a decrease in the frequency and latency to the onset of seizures and a decrease in recurrence after withdrawal of the anesthetic treatments [49][51][52][53]. Moreover, the induction of SE in rats under conditions of normothermia or moderate hypothermia (32–34 °C) was associated with a decreased number of apoptotic hippocampal neurons [54]. These experimental elements were confirmed by other studies, which made it possible to specify the involved pathophysiological mechanisms. Yu et al. showed that moderate hypothermia (32–34 °C) was associated with regulation of the expression of the GluR1 and GluR2 subunits of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors to glutamate, again reducing the volume of necrotic hippocampal neurons and apoptosis [55]. More recently, Phillips et al. showed a decrease in intracellular calcium entry via the activity of N-methyl-d-aspartate (NMDA) receptors in rats receiving hypothermia [56]. Finally, Wang et al. also made an important contribution to our understanding of the neurotoxicity associated with SE by demonstrating a decrease in brain edema induced by seizure activity and improved cognitive abilities in rats treated by hypothermia immediately after an episode of SE induced by kainic acid [54].
The regulation of changes in osmolarity is mediated by osmoreceptors localized in the hypothalamus and involves the supraoptic and paraventricular nuclei of the hypothalamus neurons, which synthesize the antidiuretic hormone that acts at the posterior lobe of the pituitary gland. Changes in osmolarity can be determined by direct measurement of plasma osmolality or calculated according to the following formula: osmolarity = (2 × natremia (mmol/L)) + glycemia (mmol/L) + uremia (mmol/L). Thus, any sudden changes in osmolarity related to the variation of one of its determinants (most often natremia) can affect intracellular hydration and, consequently, neuronal intracellular tonicity. The blood brain barrier (BBB) is permeable to water and small lipophilic molecules, but impervious to electrolytes and plasma proteins. Small changes in plasma osmolarity, therefore, cause significant movement of water on both sides of this barrier. Natremia is one of the main determinants of plasma osmolarity, modulating the movement of water between plasma and the cerebral parenchyma. Thus, hypo-osmolar hyponatremia leads to plasma hypo-osmolarity and the transfer of water from the plasma to brain cells, leading to an increase in intracranial pressure, the appearance of cerebral edema, and intracranial hypertension. Conversely, hypernatremia leads to plasma hyperosmolarity and the movement of water towards the plasma sector, with a decrease in cerebral edema and ICP. However, brain cells have a mechanism to regulate osmolarity, which makes it possible to limit the variations in their volume in either situation.
We found no study evaluating the direct association of natremia and seizures or SE in animals. However, several studies performed on hippocampal tissue slices tested the effects of osmolality changes on neuronal activity; they demonstrated the enhancement of epileptiform discharges with decreasing osmolality. Conversely, any increase in osmolality resulted in less neuronal epileptiform activity [57][58][59][60]. These in vitro findings were confirmed in animal studies. Indeed, adult rats receiving hypo-osmolar treatment by intraperitoneal administration of distilled water demonstrated high BBB permeability [61]. Similar alterations in BBB integrity were observed after epileptic seizures [62].
Moderate hypoglycemia was demonstrated to be associated with BBB alterations in an experimental study on Wistar rats subjected to electroconvulsive seizures [63]. The severity of BBB breakdown was linked to the number of electroconvulsive seizures. Such alterations occurred mostly in the thalamus, hypothalamus, amygdale nuclei, and frontoparietal and occipital cortices. Conversely, rats exposed to hyperglycemia and seizures exhibited no significant BBB dysfunction relative to normoglycemic rats [63][64]. Finally, further experimental findings on mice demonstrated a relationship between dysglycemia and seizure-induced cell death. In these animals, glycemic control might participate in diminishing neuronal injury [65].
Hypoglycemia has also been demonstrated to be responsible for clinical seizure activity. The involved pathophysiological mechanisms are related to aspartate release to the extracellular space of the brain, increasing the levels of excitatory amino acids responsible for seizure activity [66]. Paradoxically, in vitro glucose depletion contributes to the arrest of epileptiform activity [67]. The reason for this discrepancy between clinical and in vitro findings has yet to be elucidated.
Hyperglycemia also has proconvulsant properties. Higher glucose levels triggered epileptiform activity in an experimental study. Indeed, in vitro epileptiform activity correlated with glucose levels, and the lower epileptic threshold was associated with a higher glucose concentration, whereas a lower a glucose concentration was suppressive [64].
Arterial oxygen transport depends on oxygen (O2) and hemoglobinemia. Thus, hemoglobin plays an important role in increasing the cerebral tissue pressure in oxygen (PtiO2), which is the true marker of cerebral oxygenation. However, anemia decreases blood viscosity and is associated with an increase in CBF. Its role as a factor in secondary brain insults is therefore a subject of debate in traumatic brain injury. There has been no reported association of anemia and seizures or SE in animals.
Physiologic changes linked to ictal activity were investigated in few clinical studies. In patients with subarachnoid hemorrhage, nonconvulsive seizures were independently associated systemic inflammatory response syndrome characterized by higher tumor necrosis factor receptor and high-sensitivity C-reactive protein serum levels [68]. Moreover, SE is associated with systemic complications that may lead to multiple organ failure and death. Most of them can directly or indirectly be responsible for the secondary brain insults cited above. For instance, cardiac complications, which can be encountered in two-thirds of patients with SE, may be associated by arterial blood pressure hypotension. Similarly, respiratory failure, occurring in about one-third of cases during SE, may induce hypocapnia, hypercapnia, and hypoxemia [69].
However, apart from one randomized controlled trial that assessed the effect of targeted temperature management, no strategy specifically targeted to the management of secondary brain insults in SE has been investigated to date. In this trial, a group of patients with convulsive SE, requiring mechanical ventilation for refractory convulsive SE or any other reason, receiving therapeutic hypothermia treatment (between 32 and 34 °C) were compared to a similar group of patients receiving normothermic treatment. The primary endpoint was neuroprotection, assessed by a 90 day Glasgow Outcome Scale score of 5, corresponding to a good recovery, in other words, full recovery or minor symptoms that do not affect daily life. The secondary endpoints were concerned with evaluation of the antiepileptic properties of therapeutic hypothermia, namely total seizure duration and progression to non-convulsive SE, refractory SE, or super refractory SE [70]. The results were negative for the primary endpoint, as 49% of patients in the hypothermia group demonstrated a 90 day GOS of 5 versus 43% in the normothermia group. The results for the secondary endpoints showed a favorable trend of therapeutic hypothermia for an anticonvulsant effect. Progression to non-convulsive SE was the only variable that significantly decreased in the hypothermia group relative to the normothermia group [70]. Finally, the results of this randomized controlled trial did not support the use of therapeutic hypothermia as a neuroprotective strategy after convulsive status epilepticus but provided encouraging data regarding its anticonvulsant activity.
SE is associated with various physiologic changes and complications that can lead to secondary brain insults, as consequences and/or sequelae of ongoing seizure activity, but also as consequences and/or sequalae of the treatments of refractory or super refractory SE, as well as the underlying critical illness which further exacerbates and contributes to secondary brain insults. Further studies are required to evaluate the association of secondary brain insults and cause of death in status epilepticus, and to assess the neuroprotective benefit of controlling secondary brain insult after SE [71].