1000/1000
Hot
Most Recent

Chronic hepatitis B virus (HBV) infection is one of the most common factors associated with hepatocellular carcinoma (HCC), which is the sixth most prevalent cancer among all cancers worldwide. The mechanisms underlying HBV-induced malignant transformation remain unclear, but some studies have suggested that the hepatitis B virus core (HBc) protein has a potential function in the pathogenesis of hepatocellular carcinoma in addition to the HBV X protein. This review focuses its discussion on the involvement of HBc in the development of hepatocellular carcinoma.
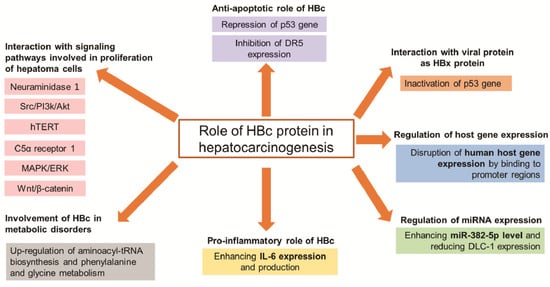
| Group | Target | Mechanism of HBc to Promote Hepatocarcinogenesis |
Reference |
|---|---|---|---|
| Signaling pathways | Neuraminidase 1 | Promote NEU1 expression inducing proliferation and migration of hepatoma cells | [18] |
| Src/PI3k/Akt | Activate Src/PI3k/Akt pathway inducing tumor formation of hepatoma cells | [14] | |
| hTERT | Upregulate the c-Ets2-dependent expression of hTERT inducing hepatoma cell proliferation | [17] | |
| C5α receptor 1 | Upregulate C5AR1 via NF-κB pathway to facilitate the growth and migration of hepatoma cells | [19] | |
| MAPK/ERK and Wnt/β-catenin | Bind to gene promoters of these pathways, thus participating in the progression of HCC | [20] | |
| Anti-apoptosis | p53 | Prevent hepatoma cells from anti-Fas antibody-induced apoptosis through the p53-dependent Fas/FasL signaling pathway | [21] |
| Repress the p53 gene through the transcription factor E2F1 binding site in the p53 promoter | [22] | ||
| DR5 | Prevent hepatocytes from TRAIL-induced apoptosis through inhibiting DR5 expression | [23] | |
| Metabolic disorders | Cell metabolism | Upregulate aminoacyl-tRNA biosynthesis and phenylalanine and glycine metabolism inducing development of HCC | [24] |
| Immune system |
IL-6 | Enhance IL-6 expression and production that involved in pathogenesis of HBV | [25] |
| Epigenetic | miR-382-5p | Promote HCC metastasis through enhancing miR-382-5p level and reducing DLC-1 expression | [26] |
| Genetic | Promoter regions |
Disrupt human host gene expression by binding to promoter regions, which modulate normal functions of liver cells | [20] |
| Interaction with viral protein as HBx protein | p53 | Inactivate the p53 gene, thus participating in HCC progress | [22] |
NEU1: neuraminidase 1; hTERT: human telomerase reverse transcriptase; C5AR1: C5α receptor 1; MAPK/ERK: mitogen-activated protein kinase/extracellular signal-regulated kinase; DR5: death receptor 5; MKK7: mitogen-activated protein kinase kinase 7; IL-6: interleukine-6; HBV: hepatitis B virus; HCC: hepatocellular carcinoma; DLC-1: deleted in liver cancer.
Resistance of HBV-infected hepatocytes to apoptosis is considered one of the major causes in the progression of chronic hepatitis to cirrhosis and ultimately to HCC [23][73]. Apoptosis of HBV-infected hepatocytes is mainly mediated by signaling belonging to the tumor necrosis factor (TNF) protein family, including TNF-α, Fas ligand (FasL), and TNF-related apoptosis-inducing ligand (TRAIL) [74][75]. TNF-α and FasL are considered as death receptor ligands.
FasL induces apoptosis of hepatocytes in both normally functioning liver and in various forms of liver disease [76] and the Fas/FasL system plays an important role in hepatocyte death during HBV infection. In response to DNA damage, the p53 tumor suppressor protein induces either apoptosis or cell cycle arrest at the G1-S. Previous studies reported that Fas transcriptional expression is regulated by p53 protein in hepatoma cells, and the cross-talk between the p53 and Fas-FasL pathways in modulating apoptosis is clinically important [77][78]. HCC may progress through the deactivation of the p53 gene [79][80].
A study demonstrated that the expression of HBx protein in infected cells inhibits the induction of apoptosis by direct interaction with the tumor suppressor p53 [81]. A similar mechanism is observed with HBc protein. Liu et al. therefore found that HBc mediated resistance of human hepatoma cells to agonistic anti-Fas antibody-induced apoptosis. They then identified that HBc significantly downregulated the expression of p53, total Fas and membrane-bound Fas at the RNA and protein levels and reduced FasL at the transcriptional level. In contrast, HBc increased the expression of soluble forms of Fas (sFas) by facilitation of Fas alternative splicing. Mechanistically, HBc-mediated Fas alternative mRNA splicing was associated with the upregulation of polypyrimidine tract-binding protein 1 and the downregulation of Fas-activated serine/threonine kinase. HBc may prevent hepatocytes from apoptosis induced by Fas/FasL system by the dual effects of reducing the expression of the proapoptotic form of Fas and enhancing the expression of the antiapoptotic form of the receptor, which may contribute to the survival and persistence of infected hepatocytes toward the development of chronic HBV infection. HBc is a survival factor capable of protecting cells, such as hepatoma cells from anti-Fas antibody-induced apoptosis through the p53-dependent Fas/FasL signaling pathway [21].
The E2F family of transcription factors plays an essential role in mediating cell cycle progression, particularly those involved in G1-S progression [82], and it has been implicated in the regulation of growth inhibition, differentiation, apoptosis, and oncogenic transformation. Previous studies have shown that E2F1 functions as both an oncogene and a tumor suppressor gene [83][84]. A study revealed that HBc protein is a transcriptional repressor of the human p53 gene. Indeed, an electrophoretic mobility shift assay demonstrated that the binding of HBc to E2F1 reduced the DNA-binding ability of E2F1 at the p53 promoter [22].
Unlike TNF and FasL, TRAIL preferentially induces apoptosis of tumor cells and virus-infected cells, but does not induce apoptosis of normal cells [75][85][86]. Following high-risk human papillomavirus (HPV) infection, viral proteins use different strategies to modulate apoptosis. In particular, the E5 protein of HPV can disrupt TRAIL-mediated apoptosis, which suggests that it may prevent apoptosis of cells at early stages of viral infection [87].
The TRAIL was recently reported to be implicated in hepatocyte death during HBV infection. Interestingly, two HBV proteins, HBx and truncated middle hepatitis B surface protein (MHBs(t)), were found to sensitize hepatocytes to TRAIL-induced apoptosis. Other data showed that HBx enhances TRAIL-induced apoptosis through Bax upregulation, whereas MHBs(t) does this through ERK2 activation [75][88].
In contrast, Liu et al. found that HBc protein had an opposite role in TRAIL-induced hepatocyte apoptosis. Indeed, HBc would be a strong inhibitor of TRAIL-induced apoptosis by blocking death receptor 5 (DR5) expression [89] inducing a decrease of TRAIL-induced apoptosis of human hepatoma cells. The DR5 gene promoter has no typical TATA-box, but has two Sp1 sites responsible for the basal transcription activity of the DR5 gene [90]. Transcription factors such as NF-kB and p53 can regulate the DR5 promoter activity. In hepatoma cell lines expressing the core protein, HBc protein induces a significant reduction in DR5 expression that represses the DR5 promoter activity. Consequently, HBc prevents hepatocytes from TRAIL-induced apoptosis through inhibiting DR5 expression [23]. In practice, if the pro-apoptotic proteins, such as HBx, are majority, HBV-infected hepatocytes may die as a consequence, and fulminant hepatitis may develop. In contrast, if the anti-apoptotic viral proteins, such as HBc predominate, the infected hepatocytes may not undergo apoptosis and chronic HBV infection may ensue.
HBc protein would prevent hepatocyte from FasL-induced apoptosis by altering the membrane and soluble Fas level, while it would also prevent sensitized TNF-α-induced apoptosis by disrupting the interaction between mitogen-activated protein kinase kinase 7 (MKK7) and receptor of activated protein kinase C 1 (RACK1). RACK1 is described as a scaffold protein that facilitates the phosphorylation of MKK7 by its upstream activators.
One study with ectopic expression of HBc in HepG2 cells and primary hepatocyte cultures reported that HBc abolishes the interaction between MKK7 and RACK1 by competing with MKK7 for binding to RACK1, thereby downregulating TNF-induced phosphorylation of MKK7 and the activation of JNK, an important regulator of TNF-α signaling. Specific knockdown of MKK7 increases the sensitivity of hepatocytes to TNF-induced apoptosis, while overexpression of RACK1 counteracts the proapoptotic activity of HBc. The expression of HBc makes hepatocytes susceptible to TNF-induced apoptosis by disrupting the interaction between MKK7 and RACK1 [91] and this finding suggests a direct role of the core protein in driving liver pathogenesis in chronically infected patients.
The progression of cancer seems to involve major disorders in cell metabolism [92]. Metabolic disorders are shared by both transformed cells and those infected with viruses, suggesting that metabolic reprogramming is an important hallmark of viral oncogenesis. Viruses handle metabolic pathways and associated-signaling cascades to provide sufficient resources for the production of new virions. Among viruses, chronic Hepatitis C virus (HCV) infection is more associated with metabolic alterations than HBV infection. Indeed, patients with chronic HCV often develop secondary metabolic disorders, such as insulin resistance and steatosis [93].
One study reported that the link between metabolic disorders and HCC could be attributed to the effects of HBV infection and in particular the HBV-encoded proteins [94], such as HBx protein [95]. Recently, multi-omics analyses of HBc transfected cells revealed that HBc protein promotes the expression of multiple metabolic enzymes and the secretion of metabolites from hepatoma cells modifying the metabolic characteristics of HCC cells, and contributes to HBV-related metabolic dysregulation through the modulation of glycolysis and amino acid metabolism. For instance, glycolysis and amino acid metabolism are significantly upregulated by HBc. Max-like protein X (MLX) would be an important protein in glycolysis and lipid biosynthesis in tumorigenesis. Besides, MLX might be recruited and enriched by HBc in the nucleus to regulate glycolysis pathways. Moreover, PGK1 is also upregulated by HBc. A recent study highlighted that PGK1 acted as a protein kinase in coordinating glycolysis and the tricarboxylic acid cycle, which is instrumental in cancer metabolism and tumorigenesis [96]. Therefore, Xie et al. concluded that nine pathways were considered closely related to the development of HCC, including aminoacyl-tRNA biosynthesis and phenylalanine and glycine metabolism [24].
Dysregulated cholesterol homeostasis is a characteristic of numerous diseases, including liver fibrosis, and even many cancers. A recent study showed that ethanol and HBV together synergistically enhance cholesterol biosynthesis and decrease cholesterol utilization and its uptake in vivo and in vitro. Thus, HBV is involved in the dysregulation of cholesterol homeostasis and increases hepatic cholesterol deposition in alcoholic fatty liver via the hepatitis B core protein [97]. These changes may contribute to the progression of various coexisting diseases.
Interleukin (IL)-6 is one of the most significant cytokines involved in hepatic inflammation and hepatocarcinogenesis in patients with liver diseases [98][99][100]. Its role has been described in HCV [101] and HBV infections [100]. Higher serum IL-6 level was an independent risk factor for HCC development in female hepatitis C patients. In the case of HBV infection, high serum IL-6 level was also associated with HCC risk [98] and aspartate aminotransferase [102], and considered as a prognostic indicator in HCC [103]. Previous studies described that human hepatoma cells and hepatic cells secrete IL-6 after activating NF-κB pathway and a MyD88-dependent signaling pathway, whose activation is regulated by protein phosphatase type 2 C alpha in the presence of HBx protein [104][105][106]. An intracellular HBcAg expression model (transfected hepatocyte-like cells) showed that the expression of HBc in hepatocytes enhances IL-6 expression and production (checked by qPCR and ELISA, respectively), which was mediated through activating p38 mitogen-activated protein kinase, extracellular signal-related kinase and NF-κB pathways. Cytoplasmic HBcAg seems to be a viral antigen for immune-mediated liver damage, and HBV-infected parenchymal cells may produce proinflammatory cytokines that are involved in pathogenesis of hepatitis B [25].
Accumulated epigenetic alterations including histone modification, DNA methylation and non-coding RNA (micro RNA or miRNA, lncRNA) were described to have profound significance in HBV-related carcinogenesis [107]. Through its partially complementary sequence to the 3′-UTR of target mRNAs, miRNAs result in gene silencing via translational repression and/or mRNA degradation and are therefore involved in regulating almost all known physiological and pathological processes [108]. miRNAs play a key role in host-virus interactions [109] and their dysregulation is involved in liver fibrosis and a number of human cancers such as HCC. MiR-122 is the most abundant miRNA in the liver, representing 70% of the total miRNA in hepatocytes [110]. MiR-122 is downregulated in patients with HBV-related HCC, while it is upregulated in patients with HBV chronic infection [111].
HBc protein promotes the hepatocarcinogenesis process through the regulation of some miRNAs. The deleted in liver cancer (DLC-1) gene encodes a Rho-GTPase activating protein and is an important negative regulator for cell motility. Previous studies have demonstrated that DLC-1 functioned as a tumor suppressor gene and downregulation or even loss of DLC-1 expression often occurred in HCC [112]. The DLC-1 gene is potentially targeted by several differentially expressed miRNAs in HBc-introduced cells according to the miRNA-target gene network analysis. Thus, miR-382-5p seems to be significantly upregulated in HBc-overexpressing HCC cells. The HBc protein promotes HCC metastasis through enhancing the miR-382-5p level and reducing DLC-1 expression. The miR-382-5p/DLC-1 axis is essential for HBc-promoted HCC metastasis. These data further showed that, similar to HBx, HBc protein might also play multiple roles in different stages of HCC development [26]. A similar mechanism was observed with HCV. Indeed, miRNAs miR-141 and miR-200a are accentuated in HCV-infected human primary hepatocytes and can target DLC-1 mRNA reducing its expression and then induce HCC development [112].
A part of miRNA is closely related to the stage of liver disease. Indeed, studies have shown that the miRNA circulating in serum or plasma could serve as the role of biomarker for the diagnosis and prognosis of HBV-related diseases [113].
HBV targets host genes that are involved in cell survival to escape immune surveillance and facilitate malignant transformation. HBc protein may bind specifically to certain human gene promoters, through either its C-terminal functional domain or its N-terminal assembling domain. Previous research has generated the genome-wide profile of HBc in HBV-infected hepatocytes using chromatin immunoprecipitation microarray studies [20]. This study showed that HBc could bind to 64 gene promoters of the MAPK pathways and 41 gene promoters of the Wnt/β-catenin signaling pathways, whereas these two pathways are known to be critically involved in the development of HBV-related hepatocellular carcinoma. Moreover, the authors suggested that HBc tended to target the regulatory regions of genes with molecular function and malignant transformation in the liver cell repertoire. A previous study highlights that the accumulation of slight effects from HBc binding to many gene promoters may produce quite large effects on host cellular functions, possibly increasing a cell’s susceptibility to carcinogens [114].
Therefore, HBc has the ability to bind gene promoters in the human genome to modulate normal functions of liver cells infected with HBV and HBc could disrupt the expression of nearly 3100 human host genes by binding them to promoter regions [20].
HBc protein seems to be able to interact with HBx, but the impact of this interaction on hepatocarcinogenesis seems to be controversial in some cases. Kwon et al., in their study of cultured HepG2 cells, showed that HBc and HBx proteins could synergistically repress both the promoter activity and the expression of the human p53 tumor suppressor gene [22]. The inactivation of the p53 gene participates in HCC progress and the synergistic action of these two proteins has an impact on malignant transformation.
In contrast, HBc and HBx proteins could reduce the expression of two Id proteins (Id1 and Id3) whereas Id proteins are supposed to be elevated in many tumor types and would correspond with the poor prognosis of HCC patients. Thus, HBc is capable of restraining the BMP/Smad signaling pathway and HBx is able to interact with both Id proteins for facilitating their degradation through proteasome-dependent manners. Therefore, it would be interesting to explore how HBV, one of the pathogenic factors of HCC, influences Id proteins [115].
Then, HBx protein can transactivate the expression of all HBV proteins through these two enhancers and can therefore increase the expression of the HBc in vitro and in vivo by transactivating the C promoter. The regulation of HBx level may be important in HCC development. In cultured human hepatoma cells, Kim et al. demonstrated that the level of HBx protein was significantly reduced by the co-expression of HBc, whereas the level of HBx mRNA was unaffected. The inhibitory effect of HBc is specific to HBx, and it did not affect other HBV proteins. It seems that HBx activates the synthesis of HBc during the early stage of viral replication and that HBc in turn functions as an effective downregulator of HBx. The regulation of the HBx level could involve the activation of the proteasome-mediated degradation of HBx. To date, no direct physical interaction between HBc and HBx has been demonstrated. Nevertheless, mutational analysis indicated that the C-terminal half of HBc is responsible for its inhibitory effect and that HBc protein controls the HBx level via a form of inhibitory feedback mechanism [116]. This study highlights a novel aspect of HBc function in the HBV life cycle and possibly in the development of HCC through control of the HBx level. To our knowledge, the molecular mechanism was not identified.
All of these elements emphasize a close link between HBc and HBx proteins, with a number of potential impacts on liver tumorigenesis.
Due to the high morbidity and mortality of HCC worldwide, for a number of years, many investigations on HCC carcinogenesis have been conducted that seek to elucidate the molecular mechanisms facilitating the design of better strategies to treat HCC. Evidence supports that the HBc protein has an oncogenic role in HBV-related HCC through several mechanisms, thereby controlling cancer cell proliferation and enabling malignant transformation. These include the signaling pathways involved in migration/proliferation of hepatoma cells; the resistance of cells to apoptosis; cell metabolic disorders; enhancing IL-6 expression and production; epigenetic alterations (miRNA); and other genetic processes.
With regard to garnering new insights into the biological roles of HBc in regulating HBV-related hepatocarcinogenesis, further exploration of the molecular mechanisms related to the dysfunction of hepatoma cells mediated by HBc may help us find novel therapeutic strategies for HBV-related HCC.