Metabolic syndrome (MetS) is a clustering of biochemical and physical conditions associated with an increased risk for developing type 2 diabetes (T2D) and cardiovascular diseases. Excess abdominal fat is most closely associated with metabolic risk factors and is most likely the initiating factor of risk factor clustering in MetS. These risk factors, namely, impaired blood glucose, dyslipidaemia, and raised blood pressure, are symptoms of metabolic chaos inside the body.
1. Introduction
Metabolic syndrome (MetS) is a clustering of biochemical and physical conditions associated with an increased risk for developing type 2 diabetes (T2D) and cardiovascular diseases [1]. Excess abdominal fat is most closely associated with metabolic risk factors and is most likely the initiating factor of risk factor clustering in MetS [2]. These risk factors, namely, impaired blood glucose, dyslipidaemia, and raised blood pressure, are symptoms of metabolic chaos inside the body. A high carbohydrate diet, inadequate hydration, a poor sleeping pattern, and overnutrition were described as strong modifiable risk factors for MetS [3]. Hence, MetS became an ideal target of lifestyle-focused interventions.
Group-based interventions have been documented to manage various metabolic conditions, especially among those with T2D and obesity [4][5]. Although evidence suggests that group interventions have a therapeutic benefit beyond providing patients with information and education, studies on its effectiveness need further exploration [6]. For example, while group-based programs effectively improved clinical, lifestyle, and psychosocial outcomes among patients with T2D [4], the interventions themselves have been poorly documented [7]. The designs and effectiveness of these interventions also tend to vary considerably among the studies [5]. However, interventions underpinned with a theoretical framework, education, and social support tend to be more efficacious [8].
In addition to that, peer support has become a common aspect of group-based interventions. While not all group-based interventions use the peer support framework, it has been shown to have beneficial effects on intervention outcomes. Peer support in lifestyle interventions has provided a space for healthcare providers or trained peer leaders to deliver extensive health education and self-management instruction while also allowing for increased adoption and productivity [9]. Peer support combines the benefits of receiving and providing social support, making it superiorly beneficial for adults with similar chronic diseases who need a lifestyle change [9].
2. Four Strategies
Non-communicable diseases (NCDs) disproportionately affect people in low- and middle-income countries. More than 85% of global NCD deaths occur in low-resource settings
[10]. The exorbitant costs of NCDs, including prolonged, expensive treatment, pose a significant economic burden worldwide, especially in developing countries
[11]. Therefore, cost-effective innovations to manage and prevent NCDs, including MetS, are required. This review highlighted 13 studies of variable designs exploring group-based lifestyle interventions in MetS. Most of the studies employed existing diabetes self-management programs and published national guidelines and were primarily focused on clinical endpoints.
Despite the need to scale up lifestyle-based interventions for NCDs, very few interventions met the benchmark
[12]. The inclusion of an appropriate behavioural theory in intervention development among MetS patients was beneficial
[13]. Based on our review, several psychological theories have been used in program development, including the Social Cognitive Theory, Transtheoretical Model of Change, the Health Belief Model, and self-efficacy theory. Hence, it can be concluded that there is yet to be conclusive evidence to point out the most suitable theory or model for the clustering of risk factors or MetS as a whole
[14]. The selection of behavioural theory in an intervention should be made based on the objective of the intervention and the factor to be intervened.
There is a need for a systematic evaluation of behavioural approaches in lifestyle intervention to help researchers learn about fundamental elements that may improve or halt behavioural change among adults with MetS
[15]. This understanding can assist researchers in designing a better framework to intervene in different populations throughout the world. As the information and knowledge of peer-based intervention on NCDs, particularly MetS, is still in infancy, research is needed on all dimensions, including designing, implementing, and evaluating different peer support models to meet the needs of diverse populations in various settings
[14][16]. This review suggests a set of research directions for the prevention and management of MetS (
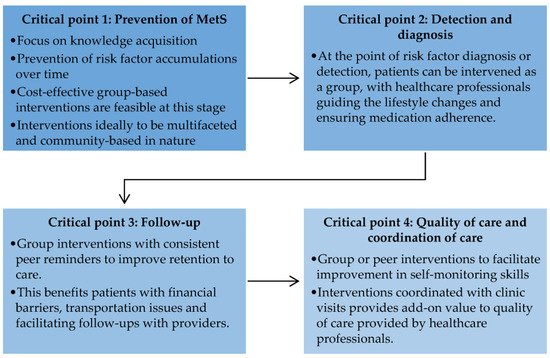
Figure 1).
Figure 1. Critical points for future research in the prevention and management of metabolic syndrome.
2.1. Critical Point 1: Prevention of Metabolic Syndrome
Poor knowledge of preventive measures is a crucial contributor to the rising incidence of MetS. MetS itself is a non-consensus disease, as it is perceived as a clustering of metabolic risk factors
[17]. Hence, the effort to prevent MetS is overshadowed by intervention involving pre-diabetes and overweight and obese adults, albeit related
[17]. However, the early prevention of MetS could reduce the accumulating effect of more risk factors, which may become harder to manage with time
[15][18]. For this purpose, future studies could adopt group-based interactive interventional designs, such as those involving peer support, as these designs are cost effective and feasible
[18].
2.2. Critical Point 2: Detection and Diagnosis
Although trained health professionals as peer leaders have been leveraged to address the risk factors for MetS in developed countries, this was shown to be inefficient
[19][20]. Group-based intervention may increase awareness through education and behaviour change through inter-peer communication. Such systems can be tailored to provide information regarding MetS frequently to remind all about healthy lifestyle practices and medication adherence under the supervision and guidance of healthcare professionals. This has been demonstrated in interventions with decision support for hypertension
[21] and T2D
[22] in developing countries.
2.3. Critical Point 3: Follow-Up
Group-based intervention improves retention to care, as inter-peer communications often deal with consistent reminders about lifestyle behaviour change, daily monitoring, medication, and upcoming interactive sessions
[14]. Peer reminders, for example, can play a role in promoting sustained lifestyle modification among adults with MetS. From a broader perspective, peer-based intervention can also support retention to care by assisting patients with financial barriers and transportation issues and by facilitating follow-ups with providers
[23]. Research on prospective peer support intervention following clinic visits should elucidate the efficacy of peer support in promoting sustained lifestyle changes.
2.4. Critical Point 4: Quality of Care and Coordination of Care
The peer-based program can support patient education, training, work planning, decision support, and treatment adherence in addition to routine clinic visits
[9][24]. This added value to the quality of care provided by healthcare professionals. Peer-based programs, such as weekend classes, allow peer leaders to inform peers on MetS and improve self-monitoring skills following official clinic visits
[25]. While the evidence of peer-based intervention points out improvement in MetS control, the outcomes of coordinated care between healthcare professionals and peers are still unknown. The details of coordinated care provided by peer-based intervention in addition to clinic visits are essential to ensure the continuity of care and informed decision making in MetS.
Triglycerides and high-density lipoprotein cholesterol (HDL-C) showed the most negligible improvements. This is not surprising since these lipid alterations are accompanied by the predominance of small yet dense low-density lipoprotein cholesterol (LDL-C) and are usually under recognised in MetS. These three lipid alterations constitute the so-called lipid triad or atherogenic lipoprotein phenotype, which is the main feature of MetS
[26]. Small LDL-C particles are closely associated with atherosclerosis formation and progression
[27] and are strong predictors of future cardiovascular and cerebrovascular diseases in individuals with MetS
[28]. Therefore, early recognition of these lipid alterations in MetS can contribute to proper management and treatment to reduce cardiometabolic risk
[29].
Strategic future studies are crucial to elucidate the optimal interventional strategies for MetS to be aligned with the target set by clinical standards. The urgency for high-quality evidence stems from the need to inform many important decisions regarding the diagnosis, prevention, and treatment of MetS, which facilitates peer support alongside clinical practice, highlighting the need to incorporate implementation research, monitoring, and evaluation in peer-based research. It may assist stakeholders and policymakers in evaluating innovations and strategies that merit incorporation into the existing health system and adding further investment.