Cadmium (Cd) is one of the most injurious heavy metals, affecting plant growth and development. Melatonin (N-acetyl-5-methoxytryptamine) was discovered in plants in 1995, and it is since known to act as a multifunctional molecule to alleviate abiotic and biotic stresses, especially Cd stress. Endogenously triggered or exogenously applied melatonin re-establishes the redox homeostasis by the improvement of the antioxidant defense system. It can also affect the Cd transportation and sequestration by regulating the transcripts of genes related to the major metal transport system, as well as the increase in glutathione (GSH) and phytochelatins (PCs).
1. Introduction
Heavy metal pollution is the most widespread contamination resulting from anthropogenic activities in the world
[1]. It has raised concerns about its various harmful risks to human health via the metal transfer along the food chain
[2]. Among the heavy metals, cadmium (Cd) is a toxic element and poses a hazardous impact to living organisms, such as renal tubular dysfunction and bone disease
[3]. In plants, Cd disturbs a range of important biochemical, morphological, physiological, and molecular processes, thus resulting in chlorosis and shunted growth
[4][5]. Cd stress deceases the chlorophyll content, net photosynthetic rate, stomatal conductance, intracellular CO
2 concentration, and transpiration rate
[4][5][6]. Cd stress induces the excess accumulation of reactive oxygen species (ROS), mainly due to the imbalance between ROS generation and scavenging
[7][8]. Increased concentrations of ROS further induce the lipid peroxidation and oxidative damage, destructing plant membranes, macromolecules, and organelles
[7][8]. Additionally, excessive bioaccumulation of Cd in plants inhibits Fe and Zn uptake, and disrupts the uptake and transport of K, Ca, Mg, P, and Mn
[9]. In response to Cd stress, plants have evolved the complex biochemical and molecular mechanisms that modulate ROS homeostasis and Cd compartmentation and chelation
[7][10][11][12]. Plant hormones (ethylene, salicylic acid (SA), abscisic acid (ABA), jasmonic acid (JA), auxin, brassinosteroids (BRs), and strigolactones (SLs)) and signaling molecules (nitric oxide (NO), carbon monoxide (CO), hydrogen sulfide (H
2S), and Ca
2+) are involved in plant response to Cd stress
[13][14]. Moreover, recent studies have reported that melatonin acts as a master regulator in plant Cd tolerance.
Melatonin (
N-acetyl-5-methoxytryptamine) was discovered in plants in 1995, and it is since known to act as a pleiotropic molecule to participate in multiple physiological processes, such as plant growth and development, and protection against abiotic and biotic stresses
[15][16]. In recent years, numerous studies have focused on the protective role of melatonin against Cd stress in plants
[17]. Application of exogenous melatonin increased photosynthetic pigments, and improved relative water content and stomatal conductance in mallow plants upon Cd stress
[18]. Many results showed that melatonin could re-establish redox homeostasis by certain enzymatic and non-enzymatic antioxidant defense systems to alleviate Cd-induced oxidative stress
[19][20]. In addition, melatonin decreased Cd accumulation via regulating the transcripts of several heavy metal transporter genes to restrict Cd influx, and promote Cd efflux and chelation
[19][21]. Moreover, NO and hydrogen peroxide (H
2O
2) signaling, microRNAs, heat shock factor HsfA1a and flavonoids may be involved in melatonin-mediated Cd tolerance in plants
[19][22][23][24][25].
2. Role of Melatonin in Plant Abiotic Stress Responses
2.1. Melatonin Biosynthesis and Catabolism
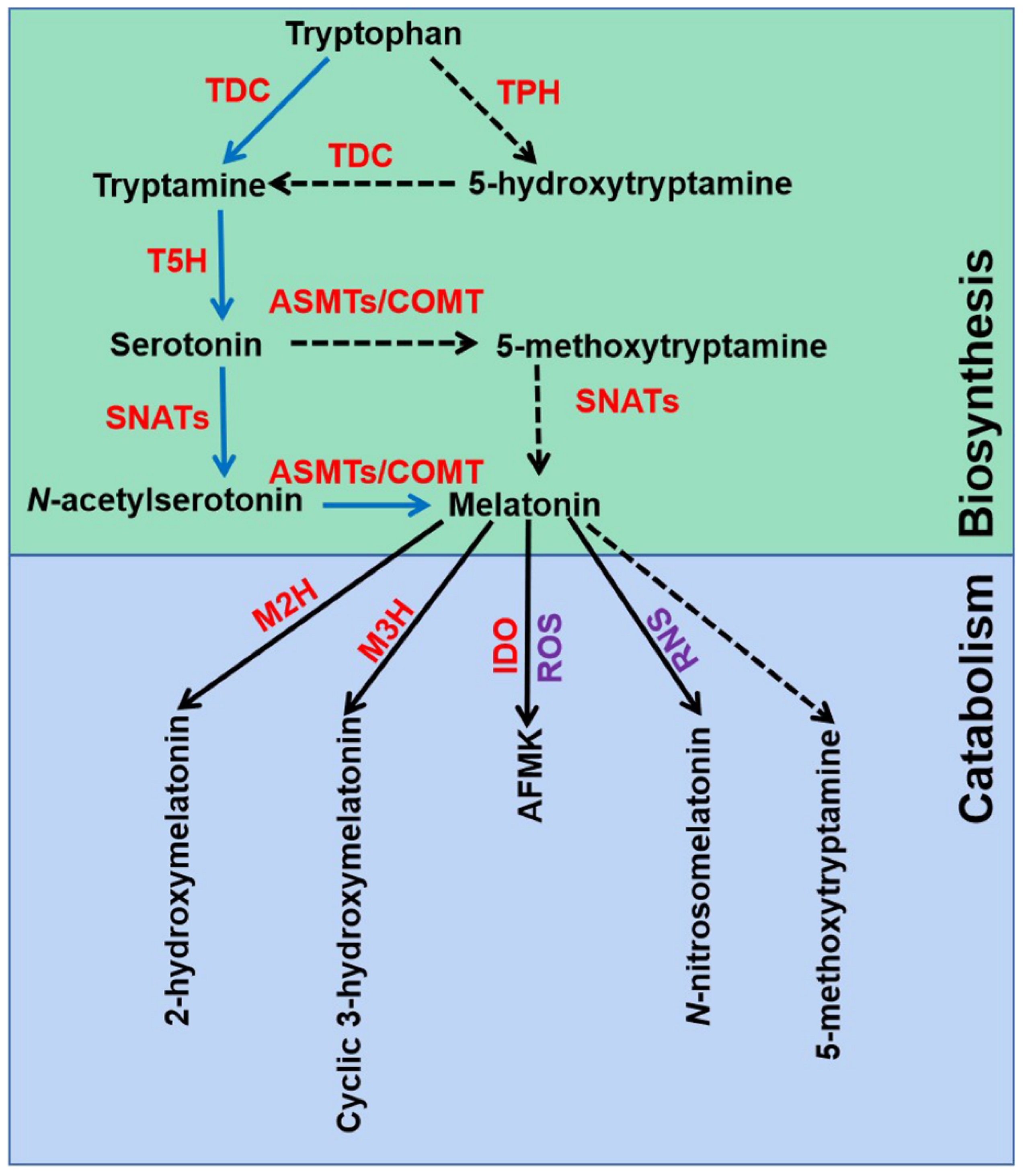
The melatonin metabolic pathway in plants contains two major parts: biosynthesis and catabolism (
Figure 1). Melatonin was discovered and confirmed by an isotope tracer study of St. John’s wort (
Hypericum perforatum L. cv. Anthos) seedlings
[15][26]. It was found that melatonin is synthesized via four continual enzymatic reactions from tryptophan, requiring at least six enzymes: tryptophan decarboxylase (TDC), tryptophan hydroxylase (TPH), tryptamine 5-hydroxylase (T5H),
N-acetylserotonin methyltransferase (ASMT), and serotonin
N-acetyltransferase (SNAT)
[17]. T5H-catalyzed hydroxylation of tryptamine is an important step of melatonin biosynthesis in rice (
Oryza sativa)
[27]. In animals, serotonin is initially acetylated to form
N-acetylserotonin, and then
O-methylated to form melatonin (named NM pathway)
[28]. It has also been found that serotonin is
O-methylated to form 5-methoxytryptamine, and then acetylated to form melatonin (named MN pathway)
[28]. Both NM and MN pathways exist in plants
[29].
Figure 1. Melatonin biosynthesis and metabolic pathways in plants. TDC, tryptophan decarboxylase; T5H, tryptamine 5-hydroxylase; TPH, tryptophan hydroxylase; SNATs, serotonin N-acetyltransferases; ASMTs, N-acetylserotonin-O-methyltransferases; COMT, caffeic acid O-methyltransferase; M2H, melatonin 2-hydroxylase; M3H, melatonin 3-hydroxylase; IDO, indoleamine 2,3-dioxygenase; AFMK, N1-acetyl-N2-formyl-5-methoxykynuramine; ROS, reactive oxygen species; RNS, reactive nitrogen species. The green box indicates melatonin biosynthesis pathways, and blue box indicates melatonin metabolic pathways.
Melatonin can be degraded by two distinct routes: non-enzymatic and enzymatic transformations
[17]. Transgenic tomato (
Solanum lycopersicum) plants expressing the gene encoding indoleamine 2,3-dioxygenase (IDO) in rice showed reduced melatonin levels
[30]. Thus, the pathway that melatonin converts to
N1-acetyl-
N2-formyl-5-methoxykynuramine (AFMK) exists in plants. Tan and Reiter speculated that AFMK is the product of melatonin interaction with ROS, which generated during photosynthesis
[28]. This might reflect the important role of melatonin in detoxifying ROS accumulation. In addition, melatonin hydroxylation metabolites, 2-hydroxymelatonin (2-OHMel) and cyclic 3-hydroxymelatonin (c3-OHMel), have been identified in plants. Their formation is attributed to melatonin 2-hydroxylase (M2H) and melatonin 3-hydroxylase (M3H), respectively
[31][32][33]. Singh et al. suggested that
N-nitrosomelatonin (NOmela) likely served as a nitric oxide (NO) carrier that participated in the redox signal transduction
[34]. Nevertheless, Mukherjee considered that NOmela served as an intracellular NO reserve in plants was questionable due to its sensitive and unstable characteristics
[35]. The processes of NOmela formation and transport are not fully understood and should be thoroughly investigated. In addition, whether 5-methoxytryptamine (5-MT) formed by melatonin deacetylation is of physiological importance remains to be investigated in plants.
2.2. Melatonin Acts as a Master Regulator in Plant Abiotic Stress
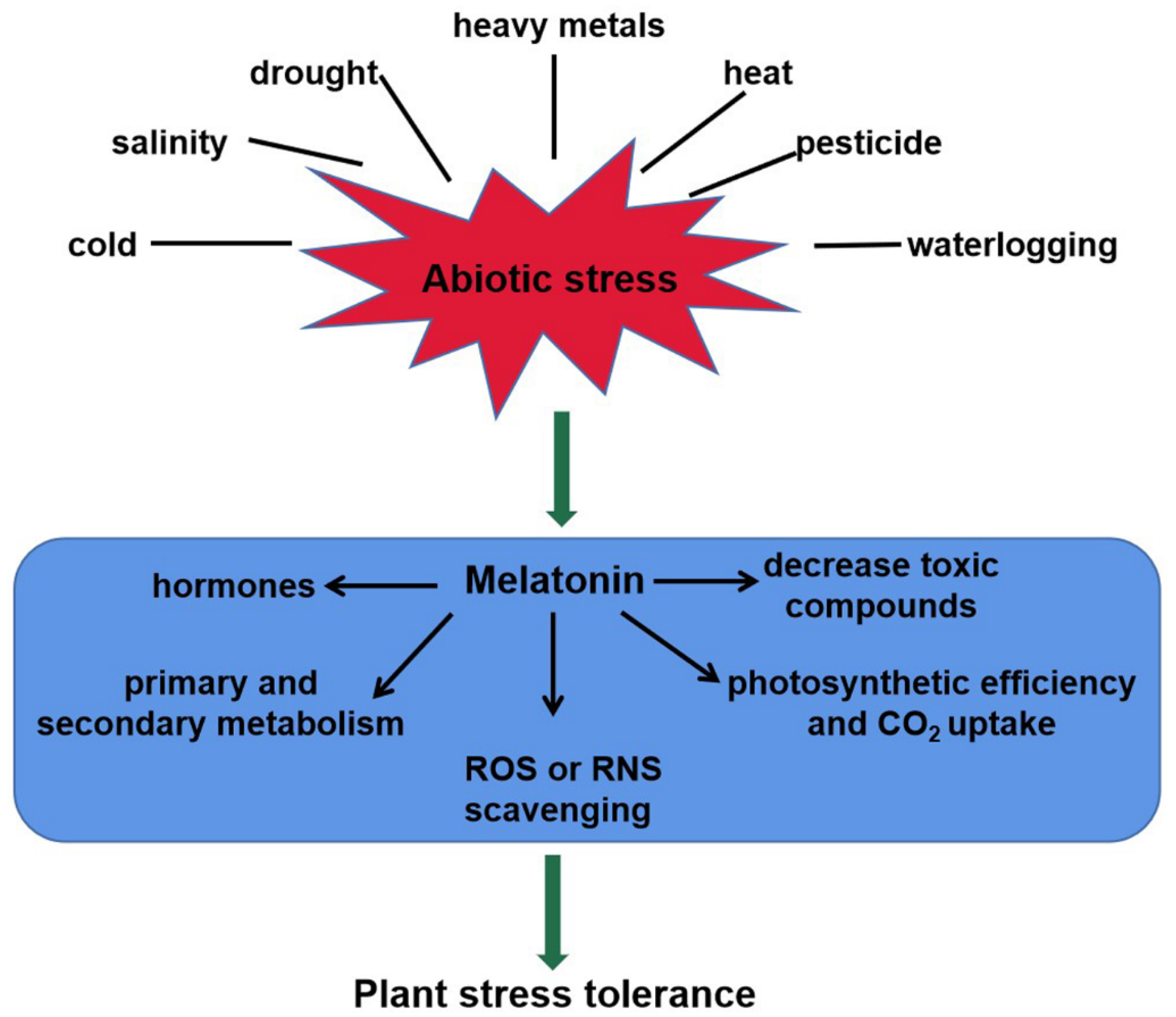
As a master regulator, melatonin plays important roles in plant tolerance to abiotic stresses, such as heavy metals, drought, salinity, cold, heat, waterlogging, and pesticides
[19][36][37][38][39][40][41]. This review shows schematically the melatonin-mediated responses to abiotic stresses in plants (
Figure 2). Melatonin levels are strongly induced by the above unfavorable conditions. For instance, endogenous melatonin level in
Arabidopsis wild-type plants was increased in response to salt stress
[36]. Loss-of-function mutation
atsnat in the
AtSNAT gene showed lower endogenous melatonin content and sensitivity to salinity stress
[36]. Cold stress induced melatonin accumulation by upregulating the relative expression of
ClASMT in watermelon plants
[38]. In tomato seedlings, Cd stress induced
COMT1 expression, and thereby improved the accumulation of melatonin
[22]. Transcription factor heat shock factor A1a (HsfA1a) bound to the
COMT1 gene promoter and activated the transcription of
COMT1 gene under Cd stress
[22]. However, the post-translational regulation of melatonin biosynthesis genes and modification of related proteins still remains largely unknown and should be elucidated in future.
Figure 2. The roles of melatonin in plant tolerance to abiotic stress. Melatonin content of plants increases significantly in responses to abiotic stresses, such as heavy metals, salinity, drought, heat, cold, waterlogging, and pesticides. It confers plant tolerance via multiple mechanisms, including ROS or RNS scavenging, toxic compounds decrease, photosynthetic efficiency increase, interaction with hormones, and secondary metabolite biosynthesis. ROS, reactive oxygen species; RNS, reactive nitrogen species.
Melatonin confers plant tolerance via multiple mechanisms, including photosynthetic efficiency increase, ROS or RNS scavenging, toxic compounds decrease, interaction with hormones, and secondary metabolite biosynthesis (
Figure 2). Melatonin stimulated stomatal conductance and improved photosynthesis, thus enhancing tolerance to water-deficient stress in grape cuttings
[42]. Another fact is that the photosynthetic efficiency was maximized by higher rates of CO
2 assimilation and stomatal conductance after application of melatonin
[43]. Several stresses can induce ROS or RNS accumulation, causing oxidative damage to plants
[44]. In this case, melatonin re-establishes the redox balance via activating enzymatic antioxidant defense systems, as well as the ascorbate–glutathione (AsA-GSH) cycle
[45]. In plants, the Salt-Overly Sensitive (SOS) pathway mediates ionic homeostasis and contributes to salinity tolerance
[46]. This pathway comprises three crucial genes, Salt-Overly Sensitive1 (
SOS1), Salt-Overly Sensitive2 (
SOS2) and Salt-Overly Sensitive3 (
SOS3), which function together to initiate transport of Na
+ out of the cell, or activating other transporters, thus leading to the sequestration of Na
+ in the vacuole
[47]. Melatonin reduced ion toxicity and improved salinity tolerance via the SOS pathway
[36]. ABA and H
2O
2/NO signaling transduction pathways were also modulated for plant tolerance in response to abiotic stress
[36][37][45][48]. In addition, melatonin could increase primary and secondary metabolites including amino acids, organic acids and sugars, and thus improving plant cold tolerance
[49].
3. Melatonin Improves Cd Tolerance in Plants
It has been found that Cd affects the ecosystem, causing stress and toxicity in plants. Melatonin acts as a key role in protecting plants from Cd stress.
Table 1 summarizes that Cd treatment up-regulates the transcripts of melatonin biosynthesis genes, such as
TDC,
T5H,
SNAT,
ASMT, and
COMT in
Arabidopsis thaliana,
Oryza sativa L.,
Solanum lycopersicum,
Triticum aestivum L.,
Nicotiana tabacum L., and
Agaricus campestris [48][50][51][52][53][54][55][56]. Therefore, melatonin contents are significantly increased. Notably, four
M2H genes, involved in melatonin degradation, were also induced
[54]. Byeon et al. suggested that both melatonin degradation and melatonin synthesis occurred in parallel, and 2-hydroxymelatonin of melatonin metabolite also acted as a signaling molecule in plant stress tolerance
[54]. As melatonin catabolism is complicated, other pathways and the role of their metabolites should be investigated in plants under Cd stress.
Table 1. Summary table explaining the effect of Cd on genes related to melatonin metabolic pathway.
|
Plant Species
|
Cd Stress and Duration
|
Impact on Genes Related to Melatonin Metabolic Pathway
|
References
|
|
Solanum lycopersicum
|
100 μM Cd2+ for 15 d
|
TDC, T5H, COMT genes (leaves)
|
[22]
|
|
Oryza sativa L.
|
500 μM Cd2+ for 3 d
|
TDC1, TDC3, SNAT1, SNAT2, ASMT, COMT, M2H, M3H genes (seedlings)
|
[23]
|
|
Triticum aestivum L.
|
200 μM Cd2+ for 1 d
|
ASMT, COMT, TDC genes (root and shoot)
|
[51]
|
|
Nicotiana tabacum L.
|
10 mg/kg Cd2+ for 1, 4, and 7 d
|
SNAT1 gene (leaves)
|
[52]
|
|
Agaricus campestris
|
2, 5, or 8 μM Cd2+ for 5 d
|
TDC, T5H, SNAT, ASMT, COMT genes
|
[53]
|
|
Oryza sativa L.
|
200 μM Cd2+ for 6, 12, 24, 72 h
|
SNAT, ASMT, COMT, TDC, T5H genes (leaves)
|
[54][56]
|
|
Arabidopsis thaliana
|
300 μM Cd2+ for 2, 3, 4 d
|
SNAT, COMT genes (leaves)
|
[55]
|
TDC1, tryptophan decarboxylase1; T5H, tryptamine 5-hydroxylase; COMT, caffeic acid O-methyltransferase; SNAT1, serotonin N-acetyltransferase1; SNAT2, serotonin N-acetyltransferase2; ASMT, N-acetylserotonin-O-methyltransferase; M2H, melatonin 2-hydroxylase; M3H, melatonin 3-hydroxylase.
Most studies showed that melatonin alleviated Cd-induced seedling growth inhibition, including the biomass (fresh weight and dry weight) and root length
[19]. Melatonin improved the photosynthesis rate (Pn), transpiration rate (E), intracellular CO
2 concentration and stomatal conductance (Gs) upon Cd stress in
Nicotiana tabacum L.
[6]. That melatonin enhanced stomatal opening and conductance capacity ultimately favored the photosynthesis in plants. Melatonin also prevented the degradation of the chlorophyll and carotenoid molecules in Chinese cabbage seedlings
[57]. Similarly, application of melatonin improved chlorophyll and the maximum quantum efficiency of photosystem II (Fv/Fm) levels of wheat plants
[20]. In chloroplasts, superoxide anion (O
2·−) in photosystem I (PSI) is generated by two molecules of O
2 with two electrons from photosystem II (PSII), and disproportionated to H
2O
2 catalyzed with superoxide dismutase (SOD)
[58]. The better potential in melatonin treated plants under Cd stress can aid in chlorophyll protection, improve photosynthesis, and mediate redox homeostasis from oxidative damage.
4. A Possible Role for H2S in Melatonin-Mediated Tolerance against Cd Stress
Acting as a signaling molecule, NO interacts with other molecules (H
2O
2, CO, and H
2S) to mediate plant growth and development, as well as abiotic stress responses
[59]. Among the molecules, H
2S is also involved in almost all physiological plant processes
[60][59]. To date, there is considerable research on the role of NO in melatonin-modulated plant abiotic stress tolerance. However, the functions of H
2S have been largely unknown. It will become a research hotspot to contribute to precise analysis of the collaboration between H
2S and melatonin, and provide deeper insight into melatonin-mitigated signaling mechanisms.
4.1. H2S Action in Plant Tolerance against Cd Stress
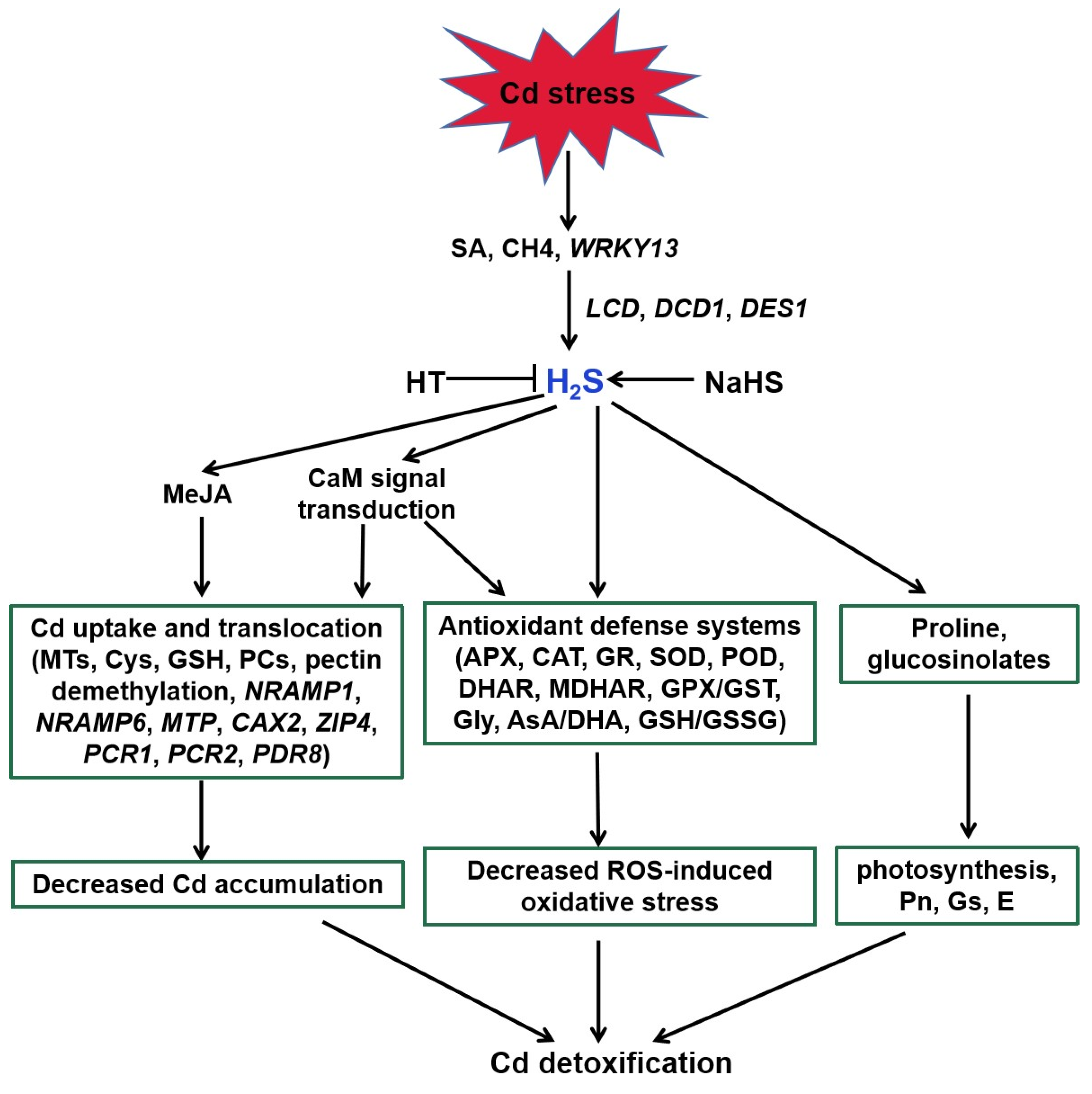
H
2S acts as a signaling molecule in modifying various metabolic processes in plants, especially Cd stress (
Figure 3,
[60]). Endogenous H
2S production was induced via expression of
LCD,
DCD, and
DES1 under Cd stress
[61][62][63]. SA, methane (CH
4), and WRKY DNA-binding protein 13 (
WRKY13) transcription factor were suggested to be involved in the above process
[64][65][66]. H
2S regulated the activities of key enzymes and AsA-GSH cycle involved in ROS homeostasis to alleviate Cd-induced oxidative stress
[67][68][69][70][71][72][73][74]. For example, H
2S enhanced the activities of antioxidant enzymes, such as POD, CAT, APX, and SOD, and thereby decreased ROS accumulation
[74]. Similarly, it also obviously increased AsA and GSH and the redox status (AsA/DHA and GSH/GSSG) levels to improve rice Cd resistance
[68][70].
Figure 3. Function of H2S in plant responses to Cd stress. SA, CH4, and WRKY13 are involved in Cd-induced H2S generation. H2S enhances the antioxidant defense systems to decrease the ROS accumulation, regulates the transcripts of genes related to Cd uptake and translocation to reduce the Cd accumulation, and increases proline and glucosinolates in response to Cd stress in plants. MeJA and Ca participate in the above regulatory pathways. SA, salicylic acid; CH4, methane; HT, hypotaurine; LCD, L-cysteine desulfhydrase; DCD, D-cysteine desulfhydrase; DES1, L-cysteine desulfhydrase 1; MeJA, methyl jasmonate; CaM, calmodulin; NRAMP1, natural resistance-associated macrophage protein1; NRAMP6, natural resistance-associated macrophage protein6; MTP, metal tolerance protein; CAX2, vacuolar cation/proton exchanger2; ZIP4, zinc-iron permease4; PCR1, plant cadmium resistance1; PCR2, plant cadmium resistance2; PDR8, pleiotropic drug resistance8.
Increasing evidence demonstrates that H
2S also regulates Cd uptake and translocation in plants
[64][71][73][75]. H
2S enhanced the expression of genes encoding metallothionein (MTs) and phytochelatin (PCS) in
Arabidopsis roots
[71]. Therefore, H
2S increased the metal chelators synthesis, contributing to Cd detoxification by binding the trace metal. In addition to enhancing the above genes expression, the protective effect of H
2S was attributed to a decrease in Cd accumulation associated with the expression of Cd transporter genes, such as
PCR1,
PCR2, and
PDR8 [64]. Exogenous application of NaHS weakened the expression of
NRAMP1 and
NRAMP6 genes, and intensified the expression of Cd homeostasis-related genes (
CAX2 and
ZIP4) to enhance Cd tolerance in foxtail millet
[76].
A number of studies address that H
2S can interact with other signaling molecules, such as SA, proline, MeJA, Ca, and NO during the responses of plants to Cd stress (
Figure 3 and
Figure 4;
[65][76][77]). H
2S acted as a downstream molecule of SA-transmitted signals to regulate Cd tolerance in
Arabidopsis [65]. The endogenous production of proline and MeJA enhanced by H
2S donor NaHS responded significantly to Cd stress in foxtail millet
[76][77]. H
2S also improved CaM gene expression and controlled the combination of Ca
2+ and CaM, which act as signal transducers
[78].
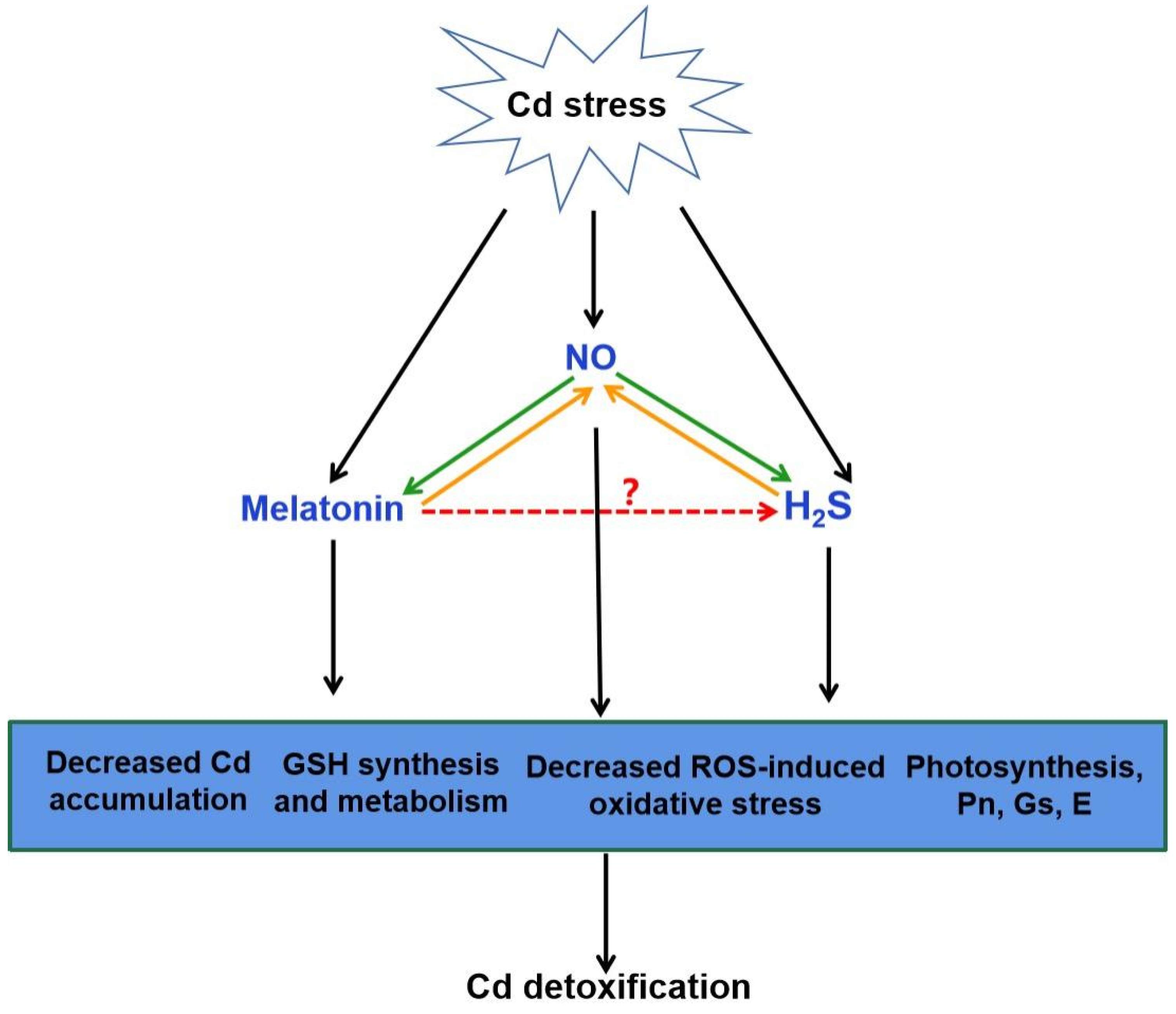
Figure 4. The possible role of H2S in melatonin-mediated Cd detoxification. NO generation can be induced by Cd stress. Increasing evidence showed that melatonin and H2S act as the downstream of NO in the responses to Cd stress, respectively (green arrow). It is also suggested that NO acts as a downstream of melatonin or H2S to improve Cd tolerance (orange arrow). The combination of melatonin, NO and H2S might be responsible for melatonin-triggered signal transduction in plant Cd tolerance via the decreased Cd accumulation, GSH synthesis and metabolism, decreased ROS-induced oxidative stress and improved photosynthesis. Red arrow, yet largely unknown. Cd, cadmium; NO, nitric oxide; H2S, hydrogen sulfide; GSH, glutathione; ROS, reactive oxygen species; Pn, photosynthesis rate; Gs, stomatal conductance; E, transpiration.
There exists a complicated and synergistic relationship between H
2S and NO in response to Cd stress in plants (
Figure 4;
[69][72][79][80]). Exogenous NO and H
2S application increased the Cd tolerance in plants
[69][79][81]. Subsequent pharmacological experiments proved that H
2S donor NaHS triggered NO production, which might act as a signal for alleviation of Cd-induced oxidative damage in alfalfa seedling roots
[79]. Nevertheless, H
2S production activated by NO is essential in Cd stress response of bermudagrass
[69]. As a second messenger, Ca acted both upstream and downstream of NO signal, and crosstalk of Ca and NO regulated the cysteine and H
2S to mitigate Cd toxicity in Vigna radiata
[81]. Moreover, application of sodium nitroprusside (SNP), the donor of NO, increased H
2S generation, and thus enhanced Cd stress tolerance in wheat
[72]. However, this protective effect was reversed by hypotaurine (HT), the scavenger of H
2S
[72]. These results suggested that H
2S and NO can function in a coordinated way under certain signaling cascades in plants under Cd stress.
4.2. Crosstalk of Melatonin and H2S in Plants
The interaction between melatonin and H
2S plays a beneficial role in abiotic stress response
[82]. Exogenous melatonin regulated the endogenous H
2S homeostasis by modulating the L-DES activity in salt-stressed tomato cotyledons
[83]. Moreover, an endogenous H
2S-dependent pathway was involved in melatonin-mediated salt stress tolerance in tomato seedling roots
[84]. Synergistic effects of melatonin and H
2S regulated K
+/Na
+ homeostasis, and reduced excessive accumulation of ROS by enhancing the activity of antioxidant enzymes. Inhibition of H
2S by HT reversed the melatonin-modulated heat tolerance by inhibiting photosynthesis, carbohydrate metabolism, and the activity of antioxidant enzymes in wheat
[85]. Recent investigation has revealed that melatonin-induced pepper tolerance to iron deficiency and salt stress was dependent on H
2S and NO
[72]. It was further confirmed that H
2S and NO jointly participated in melatonin-mitigated salt tolerance in cucumber
[86]. Thus, these results postulate that H
2S might act as a downstream signaling molecule of melatonin. Combined with the roles of H
2S and melatonin in alleviating Cd stress, it is easy to speculate that H
2S might be involved in melatonin-mediated Cd tolerance in plants (
Figure 4).
GSH plays a critical role in plant Cd tolerance. It is synthesized from glutamate, cysteine and glycine by
γ-glutamyl cysteine synthetase (
γ-ECS, encoded by
GSH1/
ECS gene) and glutathione synthetase (GS, encoded by
GSH2/
GS gene)
[87]. The catalysis of GSH1 is the rate-limiting step of GSH biosynthesis
[88]. Cd stress induced the transcripts of
GSH1 and
GSH2 in
Arabidopsis, as well as
ECS and
GS in
Medicago sativa [68][89][90][91]. It was suggested that H
2S could be quickly incorporated into cysteine and subsequently into GSH
[92]. Application of NaHS re-established (h)GSH homeostasis by further strengthening the up-regulation of
ECS and
GS genes
[68]. Similar results were also found in strawberry and cucumber plants
[93][94]. Interestingly, exogenous melatonin also increased the GSH content by inducing the transcript levels of
SlGSH1 in tomato
[95]. Hence, there might be a certain connection between H
2S and melatonin in regulating the GSH homeostasis at the transcriptional regulatory pathway. This will provide an interesting direction for further research on the complex interactions between melatonin and H
2S in improving Cd tolerance in plants.