Drimia (synonym Urginea) plants are bulbous plants belonging to the family Asparagaceae (formerly the family Hyacinthaceae) and are distinctive, powerful medicinal plants. Just some species are indigenous to South Africa and have been traditionally utilized for centuries to cure various diseases and/or ailments. They have been recognized among the most famous and used medicinal plants in South Africa. Traditionally, the plants are used for various illnesses such as dropsy, respiratory disease, bone and joint complications, skin disorders, epilepsy and cancer. A number of studies have reported biological properties such as antiviral, antibacterial, antioxidant and anti-inflammatory, immunomodulatory, and anticancer activities. Their bulbs are a popular treatment for colds, measles, pneumonia, coughs, fever and headaches.
1. Introduction
Members of the genus
Drimia Jacq ex Willd have been utilized since ancient times for various ailments such as dropsy, respiratory ailments, bone and joint complications, skin disorders, epilepsy and cancer.
Drimia, also known as
Urginea (Hyacinthaceae = Asparagaceae) plant, is a distinctive, highly poisonous, deep-red-color bulb native to the surrounding savanna areas of South Africa
[1][2], Africa, Asia and Europe
[3]. It is a pear-shaped, onion-like, scaly bulb that may grow up to 30 cm in diameter and is found below the surface of the soil
[2][3].
Drimia species are well known in traditional herbal medicine and used for the treatment of venereal diseases, such as stomach pain, abdominal pains, backache and hypertension, and as a blood purifier and an abortifacient
[2][4][5].
Drimia species and other traditional medicinal plants play an important role in the search for new bioactive compounds. Watt and Breyer-Brandwijk
[6] and Hutchings et al.
[7] believe that bulbous plants are used to reduce inflammation and possess some antimicrobial activities. In addition, South African traditional healers and herbalists utilize
Drimia to treat numerous illnesses such as, but not limited to, diseases or problems related to respiratory dysfunction and problems affecting the bones, joints and skin. According to Watt and Breyer-Brandwijk
[6] and van Wyk and Gericke
[8], traditional healers use the plant to treat venereal infections, abdominal pain, hypertension and numbness and its water extract to purify blood. The bulb is the most used part of
Drimia, as it can be applied in emetics, heart tonics, expectorants, diuretics and ointments
[9]. It can increase sex drive in males and help females who are infertile and ease dysmenorrhea
[9].
When heated, the bulb scales can treat gout and rheumatic diseases and, when in the form of powder, be used to fight viruses such as influenza and treat chronic diseases such as asthma and bronchitis
[10]. Historically,
Drimia species bulbs have been utilized for the treatment of respiratory conditions and bone, joint and skin complications. In addition, the people of Asia, Egypt and Europe have used
D. maritima to cure diseases and ailments since 1500 BC. This
Drimia species is famously known for its healing properties, and it is widely distributed in the Mediterranean area, where it has been commercialized
[1]. Traditionally, it has been utilized to treat various ailments such as bladder-related problems, headache, infertility and cardiac edema
[11][12]. The underground parts can be administered orally and externally for ritual washing and cleansing
[13]. It also has been used for the treatment of retained placenta and related diseases in cattle
[14]. Although this plant is used for most ailments in traditional medicine, traditional practitioners are well informed about the toxic nature of the plant.
2. Botanical Characteristics and Geographical Distribution of Drimia
Drimia is classified as part of the family Asparagaceae (formerly the family Hyacinthaceae). Hyacinthaceae is grouped into 1000 species, with 35 genera currently recognized. Furthermore, the genus
Drimia belongs to the subfamily Scilloideae, famously known for their bulbous plants. In addition,
Drimia consists of nearly 100 species. There is an ongoing debate about the classification of the genera
Drimia. The current consensus from taxonomic research indicates that
Urginea is a synonym for
Drimia species. In the Greek language, “
Drimia” translates to drimus, meaning “acrid” or “pungent”.
Drimia spp. are deciduous, apomorphic, short-lived flowering plants growing from perennial bulbs. Their seeds are usually black and winged
[15][16].
According to de Wet
[17],
Drimia is considered the most important and best-known species among all “slangkop” species. In addition, it is one of the six most common poisonous plants in southern Africa that are toxic to livestock and humans
[18].
Drimia can grow in various soil types, including lime, clay and stone soils, as well as alluvial soils near rivers or lakes
[1]. It has huge, pear-shaped, onion-like, reddish-brown bulbs that are 30 cm in diameter, usually dormant, just below the ground surface. The bulb is covered by a variety of thin black or dark-purple scales that have a bitter taste when applied to the tip of the tongue
[19][20][21]. Older bulbs are broken into much smaller bulbs.
The single flowering stem is normally 30 cm long, but it can grow up to 60 cm and has several small flowers that appear before the leaves in spring. The flowers appear in early spring (September to November), but flowering can begin as early as mid-July in warmer regions. Indigenous bulbs are noticeably wine red with a woody base plant and flashy roots attached
[13]. Various species of the genera
Drimia have been sketched:
D. maritima [22],
D. elata [23] and
D. altissima [24]. Raimondo et al.
[25] illustrated different parts of
Drimia sanguinea under the Red List of South African Plants (SANBI, Pretoria, South Africa)
[25].
Drimia spp. are widely distributed throughout Africa
[1], Madagascar, the Mediterranean area, southeast Asia and India
[16]. Studies have recorded that
Drimia spp. grows in various South African provinces such as the North West, Mpumalanga, Free State, Gauteng and Northern Cape. It is also found in countries such as Zimbabwe, Namibia and Botswana. The geographical distribution of
D. sanguinea is depicted by SANBI, Pretoria, South Africa
[25]. The geographical locations of
D. intricata [26] and
D. sigmoidea are in southern Africa, while other
Drimia spp. are widely spread in the Mediterranean, Spain, Sardinia, Corsica, and Egypt
[2].
They are widely known for their medicinal properties, as indicated in the oldest written record from 1500 BC. Until now, the genera
Drimia have been well investigated for bioactive compounds and commercialized
[1].
3. Biological Activities of Drimia spp.
Cardiovascular effects are the oldest behavior reported among the biological activities of the genus
Drimia. Previous in vivo or studies in clinical settings of
Drimia species concentrated on this property.
D. robusta colonies, both naturally grown and in vitro, demonstrated strong antibacterial and antifungal activity against several pathogens
[27], whereby the leaves displayed more strength than the bulb.
3.1. Antimicrobial Activity
To date, antibiotic therapy is still regarded as an essential treatment of secondary infections
[28], despite the rise in microbial resistance, hence the evolution of chronic diseases
[29][30]. A number of studies have reported antimicrobial properties such as antibacterial, antifungal and antiviral properties of the genera
Drimia in both in vitro and in vivo models. In addition, reports of the antimicrobial activities of some famous
Drimia spp. are well documented; however, the antimicrobial activities of other species are limited. Of the 40 medicinal plants tested against 11 strains of bacteria,
D. indica was graded as successful against bacterial strains. There was also a record of potent activity of
D. indica against other bacterial strains, especially
Bacillus megaterium and
Neisseria gonorrhoeae.
D. sanguinea bulbs displayed significant anti-
Staphylococcus aureus activity. It was found that
D. altissima had no activity against
Listeria monocytogenes. No significant antiviral activity from the extract of
D. maritima was reported. Among the active compounds with antimicrobial properties are the homoisoflavanone compound from
D. delagoensis and scillarenin from
D. maritima [31]. Baskaran et al.
[27] reported that
D. robusta showed significant antibacterial activity. Furthermore, the utmost concentration (19.68 μg mg
−1 DW) of proscillaridin A was reported in the roots of ex vitro plants
[27].
A study conducted on
D. indica displayed antibacterial and antifungal effects. The minimum inhibitory concentrations (MICs) ranged from 8.2 to 10.6 mg for antibacterial effects and 1.36 to 1.38 mg for antifungal effects. Various bioactive compounds such as salicylic acid, quercetin, coumarins, kaempferol, luteolin and apigenin were isolated from
D. maritima [32]. Pandey and Gupta
[33] extracted the metabolites of
Urginea indica (
D. indica) from the roots, stems and leaves using polar (aqueous, methanol), dipolar (acetone) and nonpolar (chloroform) solvents. The plant extracts were tested for antimicrobial activity against
Bacillus cereus,
Staphylococcus aureus,
Staphylococcus epidermidis,
Escherichia coli,
Proteus vulgaris and
Pseudomonas aeruginosa and against two fungi,
Aspergillus niger and
Candida albicans. They reported that root methanol extracts exhibited the highest activity against
B. cereus, while acetone extracts inhibited
P. aeruginosa. Fungi
A. niger and
C. albicans were inhibited by root acetone extract. Furthermore, the phytochemical analysis showed major compounds such as alkaloids, tannins, quinones, saponins, flavonoids, glycosides, phytosterols and resins
[33].
In another study, the activity of
U. maritima bulb extract was used to control foodborne pathogens, including
Listeria monocytogenes,
Escherichia coli,
Bacillus subtilis,
Bacillus cereus,
Klebsiella pneumoniae and
Staphylococcus aureus. The bulb extract was also tested against pathogenic
Colletotrichum graminicola,
Sclerotium rolfsii,
Fusarium oxysporum and
Penicillium digitatum. The results showed that
U. maritima bulb extract had the highest antifungal effect
P. digitatum (EC
50 = 69.01 ± 2.29 µg/well) and
C. graminicola (EC
50 = 86.89 ± 1.17 μg/well). The highest antibacterial activity was detected against
S. aureus (66.81 ± 1.06%) and
B. subtilis (57.94 ± 0.92%)
[34]. An in vitro propagation study and antibacterial assessments were conducted using
E. autumnalis and
D. robusta plants. Several bacterial pathogens, including
B. subtilis,
Micrococcus luteus,
Enterococcus faecalis,
Klebsiella pneumoniae,
S. aureus,
P. aeruginosa and
E. coli, were used to determine the antibacterial effectiveness of the plant extracts.
D. robusta bulb extract showed excellent antibacterial properties against
E. faecalis,
S. aureus and
M. luteus. Moreover,
D. robusta was determined as a noble effective bioresource
[35]. Crude extracts of
U. indica were proven to have good antifungal properties, with high inhibitory effects of 14.06 ± 0.06 mm and 13.26 ± 0.26 mm against
C. albicans and
A. niger, respectively
[36].
It was discovered that chitinase, which is a hydrolytic enzyme that disintegrates glycosidic bonds in chitin, possessed antifungal properties in the
Urginea indica (Indian squill) bulbs. The protein was purified, and in vitro results showed inhibitory effects against pathogenic
Fusarium oxysporum and
Rhizoctonia solani [37]. Matotoka and coworkers
[38] conducted a study to determine the effectiveness of herbal concoctions against HIV reverse-transcriptase and cyclooxygenase activities.
D. elata Jacq. (Sekanama) was one of the plants investigated. The results proved that concoctions made in combination with
D. elata extracts exhibited the highest HIV reverse-transcriptase effects (IC
50 = 2.90 μg/mL), better than current anti-HIV drugs (lamivudine, zidovudine, lopinavir and ritonavir)
[38]. In a previous study, Semenya et al.
[39] conducted an ethnobotanical survey on indigenous knowledge about plants used to cure sexually transmitted infections by Bapedi traditional healers. It was determined that the
D. elata bulb is used for the treatment of gonorrhea and HIV/AIDS
[39]. Several studies have reported various biocompounds such as scillarenin, tannins, cardiac glycosides and bufadienolides isolated from the genera
Drimia (
Urginea) exhibiting good antiviral activities
[39][40][41][42][43][44].
3.2. Anti-Inflammatory Activity
Aqueous extract of
D. sanguinea has shown a good range of toxic activity against fungi associated with the deterioration of food commodities and herbal drugs with antiaflatoxigenic activity
[45]. The bulb of
D. sanguinea also possesses an effective antioxidant property
[46]. Moreover, a considerable number of antimicrobial compounds have been isolated from extracts of this plant. Several plants have been identified to have anti-inflammatory properties, and most were found to be safe, effective, nontoxic and less toxic anti-inflammatory
[47] and antioxidant
[48][49]. Current research studies focusing on medicinal plants have been created on the backbone of indigenous knowledge. For example, traditionally,
Urginea maritima (
D. maritima) has been utilized for the treatment of cardiac disorders and fungal infections and as a diuretic agent. Hence, Kazemi Rad and colleagues
[50] investigated the relaxation effect of
U. maritima on rat tracheal smooth muscles. It was suggested that the incubated tissues compressing
U. maritima extract showed significantly higher relaxant outcomes compared to the nonincubated tissues. The bronchodilatory effect of the plant extract promotes the beta-2 adrenoceptor and prevents the muscarinic receptor, potassium opening and calcium channels
[50]. The anti-inflammatory activity of
D. nagarjunae extracts from the leaves and bulbs of the plant using in vitro protein denaturation techniques was investigated further. Nonpolar to polar compounds were extracted by using various solvents, including hexane, chloroform, ethyl acetate, methanol and water. The plant extracts exhibited strong anti-inflammatory activity of 82.97% ± 1.16 at 100 µg/mL
[51]. The cardiac glycoside compound showed a potent acute and chronic inflammatory effect and reduced inflammatory symptoms in vivo and animal models
[52]. In an assessment of the anti-inflammatory activity of
U. indica extracts isolated from aqueous and ethanolic solvents, it was shown that these extracts aid in relieving joint inflammation using various models
[53].
3.3. Antioxidant Activity
Recently, a study evaluated the effects of
D. maritima flowers and bulbs (essential oils) using various techniques, including DPPH, ABTS+ and total antioxidant capacity. It found that their essential oils possessed excellent antioxidant activity better than Trolox and vitamin E. The nitric oxide chelation scavenging activity of ethanol extracts from bulbs and flowers exhibited IC
50 values of 5.05 and 5.12 μg/mL, respectively. Analytical GC-MS results showed high levels of eugenol and carvacrol, which account for 41.23% and 27.29%, respectively.
D. maritima essential oils also demonstrated strong antimicrobial activities
[53]. In a 2010 study, Mammadov et al.
[54] evaluated the antioxidant activities of
U. maritima extracts produced from leaves and tubers using different solvents such as methanol, benzene, ethanol and acetone. A β-karotene-linoleic acid system and DPPH assay (free radical scavenging) were utilized to determine the total antioxidant activity of the
U. maritima extracts, and according to the results, ethanol extracts showed high antioxidant activity of 72.67%, while the benzine extract activity was 31.12%. In addition, methanol extract demonstrated free radical scavenging activity of 66.89%
[54]. Mahato et al.
[55] found methanolic extract of
U. indica bulbs possessed remarkably antioxidant activity using DPPH assay with an IC
50 value of 51.87 µg/mL, which was greater than gallic acid with an IC
50 value of 39.91 µg/mL. In addition, phytochemical screening revealed several biocompounds with alkaloids as the highest quantity, followed by flavonoids, phenols and saponins. The authors recommended
U. indica (traditional wild onion) as an alternative for the management of numerous chronic diseases
[55].
In a study on the evaluation of the antioxidant and free radical scavenging activity of various
Drimia sp. (
D. govindappae, D. coromandeliana, D. indica, D. polyantha, D. nagarjunae, D. razii and
D. raogibikei) bulb extracts, results demonstrated that
D. coromandeliana displayed the most antioxidant and free radical scavenging activities
[56]. In a recent study, free radical scavenging techniques such as DPPH, superoxide anion, hydroxyl radical and ABTS were utilized to determine the antioxidant activities of
D. maritima. The ethyl acetate extract demonstrated good scavenging activity and reduced power by employing DPPH and ABTS tests. Furthermore, aqueous extract displayed the highest activity against superoxide anions, hydroxyl radicals and lipid peroxidation
[57].
3.4. Anticancer Activity
A previous study conducted on the bulbs of South African
D. altissima isolated novel bufadienolides and drimianins A–G (1–7). Furthermore, a screening assay utilizing bufadienolide showed anticancer activity against human cancer cell lines in the NCI-60 screen
[58][59]. On the other hand, novel flavonoid C-apioglucoside, 6-C-[-apio-α-D-furanosyl-(1→6)-β-glucopyranosyl]-4′, 5, 7-trihydroxyflavone (altissimin) was recently discovered from the chemical characterization of
D. altissima. An in vitro bioassay showed antiproliferative potency against HeLa cervical cancer cells
[59]. In a study conducted by Bevara et al.
[60], the effect of C-glycosyl flavone was tested on human normal epithelial, breast, hepatic and colon cancer cell lines. The results indicate cytotoxicity potency of C-glycosyl flavone with respect to the induction of apoptosis, cell cycle arrest and inhibition of angiogenesis via CDK6
[60].
Proscillaridin A and cardiac glycosides are among the compounds that were isolated from
Drimia spp., exhibiting cytotoxic potency and/or antiproliferative activity against human breast carcinoma
[61]. In a follow-up study,
D. robusta extracts were prepared from whole plants and exhibited anticancer effects against human cell lines such as breast MCF7, melanoma UACC62 and renal TK10
[62]. A recent study on
U. maritima bulb extract also showed anticancer properties by preventing cell cycle arrest and inducing apoptosis in breast cancer cell lines
[63]. In an animal study,
U. indica methanolic extract revealed anticancer inhibitory potential in Swiss albino mice
[64]. A number of researchers have determined that
Drimia species have been utilized for a broad spectrum of applications, including ailments, respiratory conditions, bone and skin disorders, joint complications, cancer and epilepsy. An in-depth in vivo and in vitro investigation showed beneficial antibacterial, antiviral, antifungal, anti-inflammatory, antioxidant and insecticidal effects
[1], as shown in
Table 1.
Table 1. Biological investigation of Drimia plants.
| Drimia Species |
Biological Activity |
Part Used |
Extraction Form |
Chemical Composition |
Ref. |
| D. sanguinea |
Antibacterial activity, antifungal, antioxidant, anticytotoxicity |
Bulbs |
Methanol extract, petroleum ether, extract |
Pentanoic acid, n-hexadecanoic acid, 1-nonadecene, hexadecanoic acid, ethyl ester, di-isooctyl phthalate, α-sitosterol |
[65] |
| D. indica |
Antifungal activity |
Bulbs |
Crude extract, methanol extract |
O-glycosyl flavanone,
O-glycosyl flavone and C-glycosyl flavone |
[32] |
| D. indica |
Anthelmintic activity |
Bulbs |
Aqueous extract, crude extract |
Not specified |
[32] |
| D. indica |
Antitumor activity |
Bulbs |
Crude extract |
a C-glycosyl flavone (5,7-dihydroxy-2-[40-hydroxy-30-(methoxymethyl) phenyl]-6-C-βglucopyranosyl flavone |
[66][67] |
| D. indica |
Antibacterial activity |
Bulbs |
Aqueous extract, ethanol extract, methanol extract |
Not specified |
[68][69][70] |
| D. indica |
Antioxidant activity |
Bulbs |
Methanol extract, chloroform extract |
Flavonoids, phenolic and proanthocyanidins |
[71][72][73] |
| D. coromandeliana, D. govindappae, D. indica, D. nagarjunae, D. polyantha, D. raogibikei and D. razii |
Antioxidant activity |
Bulbs |
Hydrochloric acid extract and methanol extract |
Total phenolics and proanthocyanidins |
[57] |
| D. indica |
Antidiabetic activity |
Bulbs |
Ethanol extract |
Not specified |
[74] |
| D. indica |
Anti-inflammatory |
Bulbs |
Alcoholic extract |
Not specified |
[75] |
| D. robusta |
Antibacterial, anti-inflammatory, antihypertensive and anticancer activities |
Leaf Bulb |
Alcoholic extract |
Cardiac glycosides, bufadienolides |
[76] |
| D. maritime |
Antibacterial, anti-inflammatory and anticancer activities |
Bulbs |
Ethanol extract |
Cardiac glycosides, bufadienolides sclerosis and triterpenoids. |
[77] |
| D. robusta |
Antibacterial and anticandidal activities |
Bulbs and leaves |
Petroleum ether, dichloromethane, ethanol and water extracts |
Phenolic compounds |
[78] |
| D. maritima |
Antimalarial activity and cytotoxicity |
Bulb |
Aqueous extract |
Not specified |
[79] |
| D. maritima |
Asthma effect |
Bulb |
Squill oxymel (a traditional form of Drimia maritima), simple oxymel |
Not specified |
[80] |
| D. maritima |
Anticancer effects |
Whole plant |
Methanol extract |
Cardiac glycoside |
[81] |
| D. maritima |
Analgesic effects |
Squill bulb |
Proscillaridin A, taxifolin and scilliroside |
Not specified |
[82] |
| D. maritima |
Antioxidant activity and antihemolytic effect |
Flowers |
Ethanolic, chloroform and ethyl acetate extract |
Total phenolic, flavonoid and tannin |
[57] |
| D. robusta |
Antibacterial activity |
Bulb |
Ethanolic extract |
Cardiac glycosides (2-deoxy sugars), bufadienolides |
[83] |
| D. maritima |
Antioxidant activities |
Leaves and tubers. |
Ethanol, methanol, acetone extracts |
Phenolic compounds |
[57] |
| D. macrocentra and U. riparia |
Anticancer activity |
Bulbs |
Extracts |
Bufadienolides, rubellin and riparianin |
[84] |
| D. maritima |
Acaricidal activity |
Leaves and bulbs |
Methanol, ethanol, acetone and butanol |
Bufadienolides derivatives |
[85] |
| D. numidica |
Antioxidant activity |
Flowers, scales, leaves, bulbs and roots |
Methanolic |
Bufadienolides and total phenolic content |
[86] |
| D. nagarjunae |
Anticancer activity |
Bulbs and leaves |
Ethyl acetate and chloroform |
Acetic acid, (D,L)-malic acid, hexadecanoic acid, ethyl[4-t-Butyl-2,6-bis(1-methoxy-1-methylethyl)phenyl]phosphinate, octadecanoic acid, 2-hydroxy-1-(hydroxymethyl)ethyl ester |
[51] |
| D. maritima |
Decreasing dyspareunia and increasing sexual satisfaction |
Squill oil |
N/A |
Flavonoids |
[87] |
| D. maritima |
Acaricidal activity |
Leaves and bulbs |
Methanol, ethanol, acetone and butanol |
Bufadienolides |
[88] |
4. Phytochemicals of Drimia spp.
Due to the therapeutic effects of
D. sanguinea bulb, researchers have focused on the phytochemicals of
Drimia species, and the leaves and roots have also been examined
[1]. The principal constituents isolated from this genus are cardiac glycosides. In addition, in these plants, phenolic compounds, phytosterols and other phytochemical constituents have been identified
[89][90]. The major phytochemicals that are commonly present in
Drimia spp. are alkaloids, tannins, quinones, saponins, flavonoids, glycosides, phytosterols, resins, salicylic acid, quercetin, coumarins, kaempferol, luteolin and apigenin
[32][33]. In another study, GC-MS analysis was utilized to identify compounds in
U. indica crude extract. The results showed 36 compounds were identified, namely 9,12,15-octadecatrienoic acid, stigmasterol, squalene, hypocholesterolemic
n-hexadecanoic acid, diuretic phytol, pyrogallol 10.40%, 9,12-octadecadienoic acid and octadecanoic acid. In addition, a number of alkaloids, flavonoid glycosides, saponins, proteins and carbohydrates were recognized
[41].
4.1. Cardiac Glycosides
The positive impact on the function of the heart and blood vessels (cardioactive effect) caused by
Drimia species has sparked interest in the identification of compounds since the early 1800s
[1]. In 1933, Arthur Stoll was the first person to extract scillaren A (cardiac glycoside) from
D. maritima, which was a novel discovery in cardiac therapy
[91]. Cardiac glycosides are organic steroidal compounds consisting of C-24 or C-23 and biological properties such as inotropic and chronotropic effects
[92]. Furthermore, the cardiac glucoside structure is composed of tetracyclic 10, a 13-dimethyl-cyclopentanoperhydrophenanthrene nucleus and its steroidal nucleus, which is known by the cell receptors
[93][94].
A comparative study was conducted by El-Seedi et al.
[82], whereby 61 Egyptian medicinal plants from 29 families were investigated. The study suggested that cardiac glycoside from
U. maritima was accountable for the cytotoxic activities
[82].
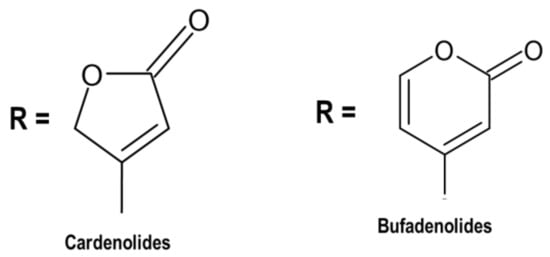
Cardenolides and bufadienolides (
Figure 1) are chemical compounds belonging to cardiac glycosides, depending on the lactone ring comprising five or six carbon atoms
[95][96]. The sugar moiety of cardenolides and bufadienolides affects pharmacological actions. In this regard, the sugar chain consists of one to three sugars, which are connected to Position 3 of the steroidal core
[97]. Morsy
[98] reported that flowering plants (angiosperms) are an abundant source of cardiac glycosides. Cardenolides are widely abundant compared to bufadienolides, and there is little possibility to obtain bufadienolide from animals and plants. Proscillaridin A (endogenous bufadienolides) has been obtained from mammalian plasma and other body fluids. However,
Drimia plants are rich in bufadienolide.
Figure 1. Two classes of cardiac glycosides occurring in nature
[95].
Follow-up studies were conducted based on the initial isolation of scillaren from
D. maritima plant in 1933
[91], and researchers focused on bufadienolide from
Drimia plants. Nuclear magnetic resonance spectroscopy (NMRS) for the identification of bufadienolides and high-performance liquid chromatography (HPLC coupled with a detector) were also utilized to identify the compounds in a mixture
[98].
Analytical techniques were also used for the detection of major constituents, including proscillaridin A, scillaren A and scillirosiden from different varieties of
D. maritima. In addition to bufadienolide compounds, several cardenolides were isolated from methanolic extract of
D. fugax, and the structure of new cardenolide was elucidated using high-field NMR spectroscopy. In a recent study, phytochemical investigations showed cardiac glycosides of Indian
Drimia exhibited significant antioxidant properties, which serves as an ideal candidate for the isolation of bufadienolides. Bufadienolides, namely scillaren A, were isolated from
D. coromandeliana and
D. razii [21]. Proscillaridin A (cardiac glycoside) derived from
U. maritima exhibited antimicrobial
[27] and anticancer properties
[62]. Several studies have utilized this compound for the treatment of congestive heart failure and cardiac arrhythmia
[27][62][99].
An earlier study used fast atom bombardment mass spectrometry (FAB-MS) and nuclear magnetic resonance (NMR) to detect 10 unknown biocompounds (Compounds 6, 14, 17, 19, 22–26 and 32) from the bulbs of
U. maritima. Compounds 6, 14 and 17 belonged to bufadienolides, which lacked sugars; Compound 32 was lignan glycoside, which is unusual in
U. maritima; and Compounds 19 and 22—26 were unfamiliar bufadienolide glycosides
[100]. Cardiac glycosides are characterized as sugar residue with an unsaturated lactone ring (five or six atoms) and a steroidal residue. These secondary compounds are produced in plants, insects and animals. In the past, plant or animal extracts comprising cardiac glycosides were used for diuretics, emetics, as poison on arrows and darts and for suicide or murder
[101].
4.2. Phenolic Compounds
A number of phytochemical compounds have been isolated and identified from
Drimia species, including flavonoids, which are phenolic metabolites
[19]. The TLC technique was used to detect the flavonoid compounds from cardiotonic glycosides
[102]. Freeform pelargonidin-3-monoglucoside and cyanidin-3-monoglucoside and ρ-cumaric acid acylated with caffeic acid were obtained from Spanish
D. maritima (red bulbs). Other phenolic compounds were quercetin-3-monoglucoside, taxifolin 4′-glucoside and
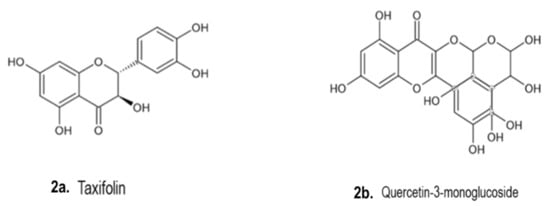
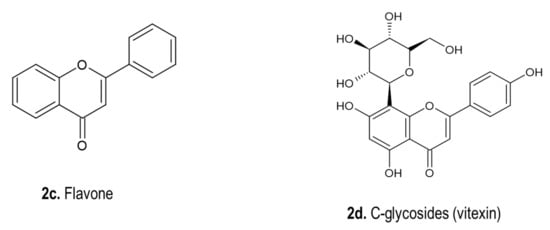
C-glycosyl flavones.
Figure 2 illustrates some chemical structures of phenolic compounds such as taxifolin (C
15H
12O
7), quercetin-3-monoglucoside (C
21H
19O
12) and flavonoid C-glycosyl (C
22H
22O
11) isolated from
Drimia spp. A homoisoflavonoid compound was isolated from
D. delagoensis. Three flavonoid glycosides were identified in
D. indica bulbs. Caffeic acid from
D. maritima, 4-hydroxy-3-methoxybenzoic acid from
D. delagoensis and phloroglucinol derivatives from
D. sanguinea were also identified as other phenolic constituents
[1]. Langat et al.
[59] discovered a new flavonoid called C-apioglucoside, 6-
C-[-apio-α-D-furanosyl-(1→6)-β-glucopyranosyl]-4′, 5, 7-trihydroxyflavone, which was isolated from
D. altissima plants. Moreover, the compound exhibited good antiproliferative potential
[59]. Various phytochemicals were isolated and identified from the bulbs of
U. maritima, and the main findings revealed high concentrations of polyphenols and flavonoids. Furthermore, HPLC-ESI/TOF-MS analysis detected ferulic acid, vanillic acid and 4-hydroxybenzoic acid as the main phenolic compounds. The biocompounds exhibited some beneficial insecticidal properties and restrictive activity on the acetylcholinesterase enzyme system in the rice weevil
Sitophilus oryzae (L.) (Coleoptera: Curculionidae)
[34].
Figure 2. Chemical structures of phenolic compounds isolated from
Drimia spp.
[1]. (
a). Taxifolin (Type of flavonoid, C
15H
12O
7); (
b). Quercetin-3-monoglucoside (C
21H
19O
12); (
c). Flavone (C
15H
10O
2) and (
d) Vitexin (Type of flavone glycosides, C
21H
20O
10).
In a follow-up study, various biocompounds such as tannins, phenols and flavonoids were extracted from
U. indica bulb extracts and identified
[55]. A number of secondary compounds were produced from the fresh plant material of red sea squill (
U. maritima). The plant extract was prepared with aqueous acetone (90:10,
v/
v). Reverse-phase HPLC (RP-HPLC) coupled with DAD and MSn detection was used to identify several compounds, including cardiac glycosides, phenolic acids and flavonoids. Dihydroquercetin, which is a potent flavonoid, was detected in high concentrations
[103]. In the endless pursuit of novel biocompounds, Sultana et al.
[104] discovered three unknown flavonoid glycosides, namely 5,4′-dihydroxy-3-
O-α-L-rhamnopyranosyl-6-
C-glucopyranosyl-7-
O-(6′′-para-coumaroyl-β-D-glucopyranosyl) flavone (2)5,6-dimethyoxy-3′,4′′-dioxymethylene-7-
O-(6′′-β-D-glucopyranosyl-β-D-glucopyranosyl) flavanone (1), and 5,4′-dihydroxy-3-
O-(2′′′′′-β-glucopyranosyl-α-L-rhamnopyranosyl)-6-
C-glucopyranosyl-7-
O-(6′′-para-coumaroyl-β-D-glucopyranosyl) flavone (3) from
U. indica bulbs (Indian squill)
[104].
4.3. Phytosterols
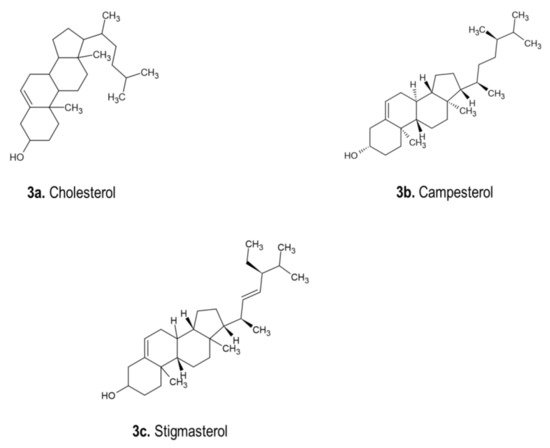
A study showed that beta- and gamma-sitosterol were obtained from
D. indica bulbs. Furthermore, the leaves, bulbs and roots of different
D. indica cytotypes were studied, and phytosterols was the dominating sterol, followed by stigmasterol. Campesterol was only obtained from triploides. Stigmasterol was also isolated from
D. sanguinea bulbs
[1]. The chemical structures of phytosterols, including cholesterol, campesterol and stigmasterol isolated from
Drimia spp., are displayed in
Figure 3. Phytosterols, β-sitosterol and stigmasterol were detected in plant parts of
U. indica. The highest total sterol content was noticeable in the leaf with 23.46 mg/gdw, and the lowest was observed in the bulb with 18.18 mg/gdw
[105]. However, Raj and Kameshwari
[44] utilized liquid chromatography–mass spectroscopy (LC-MS) and nuclear magnetic resonance (NMR) to determine the biocompounds in
U. wightii extract. Several secondary compounds, including hexadecanoic acid methyl ester, 1,3,7,11,15-tetramethyl-2-hexadecenol and stigmasterol, were identified and established to possess antioxidant effects
[44]. Phytosterols are generally referred to as plant sterols that are similar to cholesterol in structure with distant sidechain configurations. They are triterpenes with a four-ring steroid nucleus, the 3β-hydroxyl group and, frequently, a 5,6-double bond. In addition, the purpose of phytosterols is to balance the phospholipid bilayers in cell membranes. They have various applications in cosmetics, nutrition and therapeutic purposes. Other critical properties include anticancer properties
[106].
Figure 3. Chemical structures of phytosterols isolated from
Drimia spp.
[1]. (
a). Cholesterol (C
27H
46O); (
b). Campesterol (C
28H
48O); (
c). Stigmasterol (C
29H
48O).
4.4. Miscellaneous Compounds
Two alkaloids were isolated from
D. altissima [107] and were potent against
Phytophthora capsici, a tomato pest. The results were possible but not accurate because of the similarities between
D. altissima and some amaryllidaceous, as there are still a lack of herbarium data and species. From
D. altissima,
Eudesmane sesquiterpenoids have been identified. Calcium oxalate needles have been released from
D. altissima flowering bulbs
[108]. Idioblasts from
Drimia have been reported as causative factors of surface irritation
[109].
Histamine was not found in
Drimia plant. However, calcium oxalate raphides were found in
D. maritima, particularly mucilaginous idioblasts. Other compounds such as sinstrin were obtained from
D. maritima plant, which was utilized for renal clearance. Dihydro-benzofuran-typeneolignan glucoside and free amino acids such as L-azatidine-2-carboxilic acid were also isolated from
D. maritima, together with trace amounts of volatile oils. Approximately 29 kDa glycoprotein, which possessed some antifungal and antitumor effectiveness, was found by gel filtration and reverse-phase HPLC from
D. indica bulbs. Lectin-like protein and asteroidal sapogenin are other miscellaneous compounds isolated from the bulbs of
D. robusta and
D. sanguinea, respectively
[1]. Medicinal plants produces a variety of bioactive compounds with different concentrations, thus poking important issues in regards to quality, safety and efficacy
[110].