1000/1000
Hot
Most Recent

The strong impact that ionic signaling exerts on the immune system is not restricted to T cells. Cellular players from both the innate and adaptive immune compartments are likewise exposed to differential concentrations of ionic signals in the tissue microenvironment. While several pathways have been shown to be conserved across cell types, others will be more cell type specific, leaving room for additional salt effects to be observed in the future.
T cell responses are adapted to their respective cognate antigen and display a high degree of heterogeneity to meet the challenges of antigen elimination in varying tissue contexts. Upon an antigen encounter, conveyed through presentation by antigen-presenting cells, several polarizing signals educate naïve T cells to develop into mature effector and memory T cells with specialized functions in terms of cytokine production and migration [1]. The three classic signals are T cell receptor activation, costimulation and cytokines. Their differential integration in both quality and quantity gives rise to an increasing family of distinct T helper cell subsets. Th1 and Th2 cells emerged as early representatives of T helper cell subsets before the discovery of Th17 cells in 2007 sparked the quest for further identification and characterization of novel T helper cell subsets, such as Th22, Th-GMCSF and Th9 cells [2][3][4][5][6][7]. Polarizing cytokines in the context of antigen recognition and their downstream signaling pathways have been the primary focus for dissecting T helper cell diversification into their respective subsets. This approach has come with tremendous translational success since therapeutic blockade of polarizing cytokines abrogates pathogenic T helper cell functions in various disease activities.
One signal, which has previously come to attention as a novel immunoregulator, is sodium chloride. Despite its well-established role in human health and disease and the sophisticated control mechanisms exerted by the kidneys to maintain electrolyte homeostasis, its impact on the immune system has been largely ignored for a long time despite early evidence for osmotic regulation of cytokine synthesis [8].
Sodium chloride has been largely overlooked as an immunoregulatory factor in the past because its concentration in body fluids is tightly controlled and stably maintained by the kidneys [9]. While this is supported by blood monitoring, peripheral tissue sodium concentrations have been previously shown to dynamically adapt to dietary changes in sodium chloride intake and to display differential sodium concentrations depending on the type of tissue, age, sex and inflammatory state [10][11][12]. This evidence implies a heterogeneous landscape of sodium chloride concentrations throughout the human body ( Figure 1 ). Since sodium chloride dissociates into positively charged Na + and negatively charged Cl – ions in body tissues, the detection of sodium is complicated by its noncovalent binding to negatively charged tissue components, such as glycosaminoglycans and hyaluronic acid. Several qualitative and quantitative analysis strategies have been employed to assess salt concentrations in an organic matrix. Neutron activation analysis is a very sensitive quantitative technology. It is based on the neutron irradiation of the sample under consideration in a nuclear reactor leading to the production of radioactive isotopes emitting gamma quanta of characteristic energy. The intensity of the characteristic gamma radiation allows the determination of the activity (given in Bq) of the emitter [13][14]. The mass elements O, H, C and N in organic tissue are hardly activated by thermal neutron irradiation, whereas Na exhibits a high activation cross section, in other words, the absorption probability for thermal neutrons. The activation product Na-24 emits two clearly measurable gamma quanta for its identification and allows for unparalleled precise sodium quantification [13]. Alternative technologies, such as sodium ( 23Na) magnetic resonance (MR) imaging, also serve the purpose of sodium visualization and quantification in tissues. The intricacies and limitations of this method, which is gaining increasing popularity in clinical settings, are reviewed elsewhere [15]. Lastly, chemical methods, such as inductively coupled plasma-optical emission spectrometry, allow for sodium quantification in multi-step processes [16].
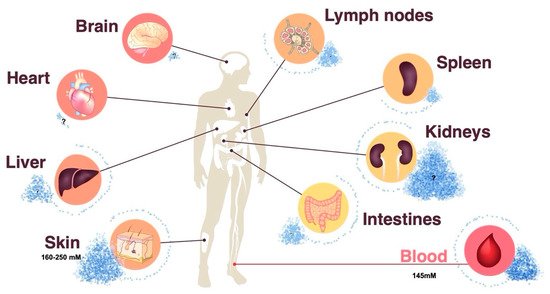
In particular, the skin has been demonstrated to serve as an interstitial sodium reservoir [17][18][19]. The mechanisms for differential sodium storage in peripheral tissues remain largely elusive. Therapeutic inhibition of glucose and sodium reabsorption from urine with inhibitors of sodium glucose cotransporter 2 (SGLT2) results in a reduction in cutaneous sodium concentrations. This effect has also been observed after diuretic treatment in patients with heart failure [10][20]. The enhanced sulfation of glycosaminoglycans in rats has been shown to serve as a negatively charged capacitor of positively charged sodium ions [19][21]. Dietary sodium chloride accumulation is associated with increased chondroitin synthase mRNA content in the skin. The extent of glycosaminoglycan (GAG) chain polymerization may therefore regulate sodium storage in the skin or other tissues [16][22]. Increased lymph flow following lymph capillary hyperplasia increases salt clearance from cutaneous stores. Interestingly, macrophages have been reported to participate in this process through their ability to produce vascular endothelial growth factor C (VEGFC) in response to salt [10][18].
Circulating immune cells are faced with a continuously changing sodium microenvironment throughout their system-wide immune surveillance under physiological conditions [23]. Under pathological conditions, the impact of sodium on immune cells is further accentuated. It has recently been shown that the lesional skin of patients suffering from atopic dermatitis, a Th2-mediated chronic inflammatory skin disease, displays up to 20-fold enriched sodium concentrations compared to those of healthy control skin [14]. Considering that the matched nonlesional skin of these patients displayed sodium concentrations resembling that of skin from healthy individuals excludes a dietary impact or other systemic reasons for this differential distribution of sodium in inflamed versus noninflamed skin within the same patient. Atopic dermatitis is characterized by an epidermal barrier defect that facilitates epidermal water loss. This water loss could potentially be a driving force for sodium enrichment in areas of increased barrier disruption. In addition, dysbiosis with Staphylococcus aureus is pathognomonic of atopic dermatitis [24]. S. aureus thrives in high salt concentrations at the expense of other species of the commensal skin microbiota [25]. Interestingly, sodium chloride has been shown to drive Th2 cell differentiation and to increase Th2-associated effector functions in memory T cells [14][26]. In particular, IL-4 and IL-13 production are increased upon increased sodium chloride exposure [14]. Together, these effects result in epidermal barrier defects, salt accumulation, dysbiosis and Th2 cell bias in a logical sequence of dysregulated events, ultimately culminating in full-blown eczematous atopic skin lesions [27]. The fibrotic skin in patients with systemic sclerosis [28] and lipedema [29] has also been associated with increased sodium concentrations. Psoriasis, another chronic inflammatory skin disease, in contrast, did not reveal increases in sodium concentrations in lesional or nonlesional skin upon analysis with a highly sensitive and specific neutron activation analysis [14]. This finding indicates that sodium is specifically associated with certain diseases but not a byproduct of chronic inflammation in general.
Interestingly, local infection has also been associated with interstitial sodium accumulation. A subcutaneous injection with Bacille Calmette–Guerin or Freund’s adjuvant in rodents has been found to result in enhanced tissue osmolalities [30]. In humans, superficial streptococcal skin infections have been found to be associated with elevated sodium levels as assessed by 23Na-MRI technology [31]. These elevated sodium levels resolved upon antibiotic treatment. Interestingly, this cutaneous sodium accumulation has been shown to promote a macrophage-driven host defense and to strengthen the antimicrobial barrier function [32]. Other organs beyond the skin have also been reported to display elevated sodium concentrations in pathological immune-mediated conditions. Disability and a progressive course in multiple sclerosis have, for example, been associated with sodium accumulation within brain lesions and within the white matter and cortical and deep gray matter [33]. Together, these findings illustrate that the level of sodium deposition is a critical factor for human health and disease through its impact on the immune system.
The polarizing cytokines for optimal human Th17 cells are still a matter of debate [34]. Sodium chloride has been demonstrated to exert very strong IL-17 induction in the pioneering work from the Kuchroo and Hafler groups in mice and humans, respectively [35][36]. The IL-17 increase in Th17 cells that are primed from naïve T cell precursors has been shown to require the presence of the Th17-polarizing cytokines IL-1β, IL-6, TGF-β and IL-21 [36]. Fully differentiated memory and effector T helper cells, on the other hand, upregulated the IL-17 expression independent of the polarizing cytokines in the presence of increased sodium chloride concentrations upon T cell receptor stimulation alone [37] ( Figure 2 ). This effect was not only restricted to the CD4 + T helper cell lineage, since CD8 + cytotoxic T cells were also shown to upregulate IL-17 upon restimulation in high sodium chloride conditions. IFN-γ and IL-4 expression, on the other hand, remained unaffected within the CD8 + T cell lineage [14].
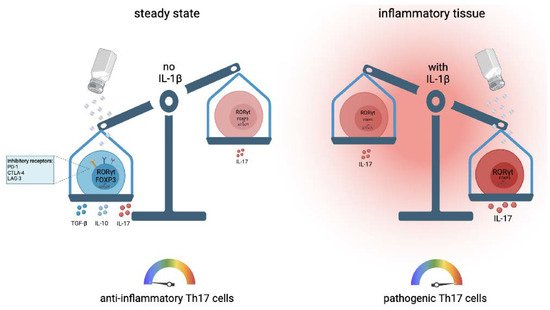
More recent work demonstrated that sodium chloride, instead, promoted the anti-inflammatory subset of Th17 cells [37] ( Figure 2 ). Sodium chloride upregulated expression of forkhead box P3 (FOXP3), the master regulator of regulatory T (Treg) cells, jointly with IL-17, along with multiple other anti-inflammatory T cell properties, including PD-1, CTLA-4 and Lag3 expression. This effect was also exerted via the NFAT5–SGK-1 signaling axis at high sodium chloride concentrations [37]. This anti-inflammatory Th17 cell phenotype also translated into immunosuppressive functions in vivo, as demonstrated by the reductions in clinical scores after the adoptive transfer of NaCl-stimulated Th17 cells into a mouse model of multiple sclerosis [37]. Together, these results demonstrate an anti-inflammatory impact of NaCl on Th17 cell functionality. This protective effect of NaCl in autoimmunity has recently been further supported with a high-salt diet in a spontaneous mouse model of central nervous system (CNS) autoimmunity [38].
Whether a high-salt diet, which is characteristic of the Western diet of the last 30 or more years, translates into an increased risk for autoimmunity or immunosuppression remains to be seen. In humans, a pilot study demonstrated that a moderate high-salt challenge increased Th17 cell levels via the modulation of the gut microbiota, leading to reduced Lactobacillus spp. abundance [39]. Another recent clinical study, however, could not identify any major changes in the immune cell subsets. In particular, Th17 cells were not found to be affected following 2 weeks of high-salt diet consumption [40]. Despite these discrepancies, dietary effects of NaCl on Th17 cell frequencies or functions cannot be entirely excluded with short-term intervention trials, meriting further investigation.
Quantitative evidence for increased sodium chloride concentrations in peripheral tissues in correlation with increased dietary intake is needed in the future to corroborate the causality between salt and immune effects in humans. Furthermore, the mechanistic networks involving the microbiota point to indirect effects of dietary salt on the immune system, suggesting more complex networks by which salt exerts its effects on human health and disease.
Treg cells are professional immunosuppressive T helper cells [41]. They display pronounced plasticity in proinflammatory cytokine microenvironments by the upregulation of IL-17 or IFN-γ, which compromises their anti-inflammatory function [42]. SGK-1, which is induced upon high NaCl exposure, has been shown to not only play a role in promoting Th17 cells (as discussed above), but to also play a critical role in the IL-23R-mediated inhibition of Treg cells [43]. High NaCl concentrations have been shown to impair Treg functions through the upregulation of IFN-γ in Treg cells via the SGK-1 node [44] ( Figure 3 ). In vivo, this translated into impaired Treg functions due to IFN-γ coproduction in a xenogeneic graft-versus-host disease model and in adoptive transfer models of experimental colitis [44]. Cumulatively, these findings suggest that Treg cells are co-opted as culprits in inflammation by losing their Treg identity through the NaCl-mediated upregulation of proinflammatory cytokine production.
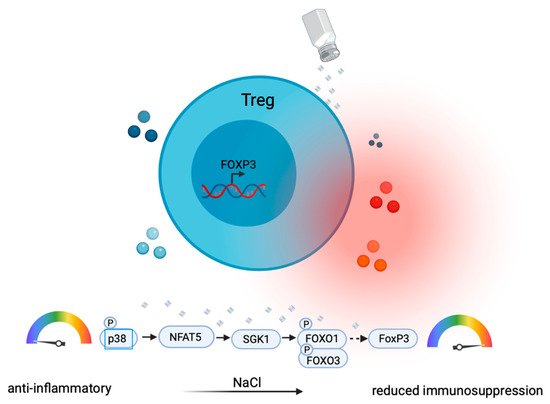
Another study did not observe increased IFN-γ expression but rather increased Th17-like differentiation in murine Treg cells upon NaCl treatment [45]. IL-17A + RAR-related orphan receptor gamma T (RORγt) + FOXP3 + induced Treg (iTreg) cells were not observed in vitro but were induced in vivo only, suggesting that additional inflammatory signals, such as IL-23 [43] or IL-1β [46], may promote the development of Th17-like Treg cells. Together, these results consistently suggest a proinflammatory conversion of Treg cells into a proinflammatory T cell subset through either IFN-γ or IL-17 induction ( Figure 3).
Interestingly, Th17-like Treg cells have previously been shown to adapt to high-salt conditions while maintaining their suppressive functions [45]. Another study supported the functional stability of Treg cells upon high NaCl exposure [47]. The development, differentiation, and functional activities of induced Treg (iTreg) cells were not compromised. Instead, the stability and function of iTreg cells were enhanced in vitro and in vivo. This result contrasted with that of the thymic Treg (tTreg) cells, and this effect has instead been attributed to plasticity in the response to NaCl [47]. These findings highlight the importance of the type of target cell for downstream sodium chloride induced effector functions.