1000/1000
Hot
Most Recent

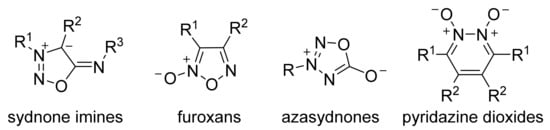
In a series of known structural classes capable of NO release, heterocyclic NO-donors are of special importance due to their increased hydrolytic stability and low toxicity. It is no wonder that synthetic and biochemical investigations of heterocyclic NO-donors have emerged significantly in recent years. In this review, we summarized recent advances in the synthesis, reactivity and biomedical applications of promising heterocyclic NO-donors (furoxans, sydnone imines, pyridazine dioxides, azasydnones).
Nitric oxide (NO) (also known as an endothelium-derived relaxing factor) is an endogenous inorganic soluble gas produced in mammalians from L-arginine and molecular oxygen by the enzyme nitric oxide synthase (NOS) [1][2]. NO is one of the most versatile molecules in animal and human biology with diverse roles in both physiology and pathophysiology. NO exhibits vasodilating properties with anti-smooth muscle cell activity [3], inhibits platelet adhesion and aggregation [4] and has other anti-inflammatory properties [5]. In addition, NO is capable of neurotransmittance and neuromodulation due to its involvement in cerebral blood flow auto- and chemoregulation [6] .
In 1998, Furchgott, Ignarro and Murad received a Nobel Prize in Physiology or Medicine “for their discoveries concerning nitric oxide as a signaling molecule in the cardiovascular system” [7][8][9]. Since then, researches on the biochemical roles of NO have grown rapidly. It was found that NO plays also an important role in a number of pathophysiological diseases, such as arthritis, atherosclerosis, cancer, diabetes and various degenerative neuronal diseases [10][11][12]. Different release patterns of NO by different NO donors can modulate angiogenesis differentially: it was shown that while the short term NO donors are primarily active to initiate the angiogenesis by inducing cellular migration and ring formation, long term NO donors define later stages of angiogenesis such as vessel maturation and neovascularization [13]. NO donor or overexpression of endothelial NO synthase fused to a green fluorescent protein (eNOS-GFP) has a protective effect against hypoxia-induced cellular deadhesion and greatly improves the redox balance by inhibiting the oxidative stress [14]. Ectopic release of NO stimulates the protection of the endothelium leaky and improves actin dynamics under hypoxia milieu in chick embryo extravascular models [15]. At the same time, overexpression of exogenous NO levels in chicken embryos may increase the cell migration and cell proliferation on the right-hand side of the heart resulting in the situs inversus which is referred to as a congenital condition comprising of the reversion of the major visceral organs from their normal positions [16]. Recently, regulation of nitrosative and oxidative stresses by NO and its influence on lung diseases and cardiogenesis were thoroughly reviewed [17][18]. No wonder, the discovery of such crucial and indispensable biochemical patterns of NO stimulated a search of prodrug candidates capable of NO release under physiological conditions [19][20][21][22][23]. Overall, the creation of efficient methodologies for the construction of novel NO-donor heterocyclic and acyclic systems became one of the rapidly developing fields in organic and medicinal chemistry.
Glyceryl trinitrate (GTN) is a well-known, approved and inexpensive NO-donor, which lowers blood pressure and increases heart rate. However, GTN suffers from various side effects, such as headache, difficult or labored breathing, dizziness, and also may induce nitrate tolerance upon continuous exposure [24]. A similar pharmacological profile matches other organic nitrate-based pharmacologically active substances (e.g., isosorbide dinitrate) [25], although their levels of NO release are quite different [26]. Aside from organic nitrates, other nitrogen–oxygen acyclic species were reported as NO-donors: C - or N -nitroso compounds, nitrosothiols, oximes, hydroxylamines, hydroxyurea and metal-nitrosyl complexes (sodium nitroprusside) [22]. However, in recent years, heterocyclic NO-donors emerged with special attention due to their hydrolytic stability, safer storage and absence of tolerance [22][23][27]. The progress made in the design, synthesis and biochemistry of heterocyclic NO-donors in the last decade unveiled an application potential of such organic nitrogen–oxygen molecular systems in medicinal chemistry and drug design.
Therefore, researchers summarized recent advances in the synthesis and reactivity of structurally diverse NO-donors incorporating nitrogen–oxygen-enriched heterocyclic scaffold: sydnone imines, furoxans, azasydnones and pyridazine dioxides ( Figure 1 ). These heterocyclic subclasses were chosen due to an increased number of researches on their synthesis, functionalization and properties. Main trends in synthetic methodologies for each type of heterocycle are presented. NO-releasing properties, pharmacological activity and other biomedical applications along with an analysis of structure– property relationships are also considered.
N 3-Substituted sydnone imines 1 can be synthesized according to a standard method [28][29] via nitrosation of the corresponding amino nitriles 2 followed by cyclization to the target mesoionic heterocycle. However, the cyclization is reversible and possible ring cleavage occurs through several degradation pathways depending on the solvent and pH of the reaction media. Therefore, to obtain stable sydnone imines suitable for storage and applications, they are usually converted to salts 3 or exo- N -substituted derivatives 4 [30].
The 4-nitrophenyl carbonate derivatives 9 may also be used as suitable acylating agents to incorporate carbohydrate motifs onto the sydnone imine scaffold. Using this method, a few examples of sydnone imine glycosyl carbamates 10 representing a new class of glycosidase-dependent NO donors were prepared [31].
An interaction of sydnone imine hydrochlorides with sulfonyl chlorides in basic media afforded N 6-sulfonyl iminosydnones 14 which have a great potential in bioorthogonal click-and-release methodology [32][33].
An interaction of sydnone imine hydrochlorides with substituted phosphinate or phosphonate chlorides in the presence of diisopropylethylamine (DIPEA) was shown to be a highly efficient method for the preparation of N 6-phosphorylated iminosydnones 18 in good and high yields[34] . Gram-scale quantities of N 6-phosphorous derivatives 18 can be easily synthesized enabling their wide utilization in organic synthesis.
The final chapter of the present review covers several aspects of the synthesis and biomedical applications of two rather neglected heterocyclic subclasses: pyridazine dioxides and azasydnones (1,2,3,4-oxatriazolium-5-olates). Historically, both these heterocyclic derivatives are known as exogenous NO-donors for more than 30 years [27]. However, substrate-specific and harsh methods for their synthesis significantly restricted in-depth investigation of their functional properties. Nevertheless, recent achievements of several research groups provided some solutions to this problem and modern convenient synthetic protocols to an assembly of both pyridazine dioxide and azasydnone frameworks along with their application potential are summarized in this section.
A more convenient approach toward the formation of the pyridazine dioxide scaffold is based on an oxidative cyclization of 1,4-dioximes 68. This method is more efficient and provides high yields of target heterocyclic NO-donors 69 including those comprised of a pyridazine dioxide motif fused with other heterocyclic rings (pyrazole [35], furazan [36], furoxan [37][38]). Initially, PhI(OAc) 2 [39] or Pb(OAc) 4 [40][41] were used as oxidants, but later N 2O 4 [35][36][38] or HNO 3 [37] were shown to provide higher yields and purity of pyridazine dioxides 69.
Due to pronounced NO-releasing properties, azasydnones possess promising antihypertensive activity and low toxicity [42][43]. In addition, (1,2,5-oxadiazolyl)azasydnones showed excellent antiplatelet activity in the case of ADP and adrenaline used as inducers completely suppressing the formation of the aggregates, which is rather a unique feature. Importantly, (1,2,5-oxadiazolyl)azasydnones possess a selective mechanism of inhibition of platelets aggregation mediated only by ADP and adrenaline, which are considered to be the main agents causing thrombus formation [44]. Additionally, recently, some energetic applications of azasydnones were reported [45][46][47][48].
Mechanistic investigations on NO-donating properties of azasydnones revealed their ability to release NO at pH > 6 either in vitro or enzymatically. This process includes nucleophilic cleavage of the azasydnone ring followed by stepwise degradation of formed nitrosamines. Degradation of (1,2,5-oxadiazolyl)azasydnones afforded benzoic acid as a main decomposition product due to the concomitant cleavage of the 1,2,5-oxadiazole ring. Since benzoic acid is also non-toxic for living organisms, (1,2,5-oxadiazolyl)azasydnones are considered advantageous in future drug design [44].
Synthesis and reactivity of heterocyclic NO-donors have become one of the urgent areas of research in organic and medicinal chemistry. In comparison to clinically used organic nitrates, heterocyclic scaffolds capable of NO release under physiological conditions are more advantageous due to hydrolytic stability and improved pharmacological profiles. Aside from studying biomedical applications of simple heterocyclic NO-donors, numerous efforts were directed towards the construction of hybrid pharmaceuticals incorporating NO-donor heterocyclic subunits as a key structural fragment to advance the pharmacological profile of the parent drug. To realize the hybridization approach, a number of modern synthetic methodologies involving preliminary functionalization of the heterocyclic ring with subsequent hybridization with an appropriate pharmacophore were utilized. This approach is very promising for the construction of novel multifunctional drugs to overcome the significant problem of multidrug resistance. In general, diversification of the heterocyclic NO-donor scaffold has become a reliable tool in modern organic chemistry; therefore, the creation of novel pharmacologically active lead hybrids with NO-donor properties should be expected in near future.
In this review, recent advances in the synthesis, reactivity and pharmacological activity of the main NO-donor heterocyclic subclasses are summarized. Although the structures of considered nitrogen–oxygen molecular systems are quite similar, synthetic methods for their preparation and functionalization differ significantly. The synthesis of sydnone imines and furoxans was extensively studied and nowadays main trends are directed toward selective functionalization of these heterocycles. Furthermore, incorporation of the sydnone imine or furoxan motif in the structure of known pharmaceutical or promising drug candidates was found to be fruitful in search of novel pharmacologically active and non-toxic substances. In addition, sydnone imines are highly valuable substrates for bioorthogonal chemical reactions, which are of major importance in the field of chemical biology. On the contrary, the chemistry of pyridazine dioxides and azasydnones is explored to a lesser extent since both these heterocyclic subunits were hard to construct. Nevertheless, recently developed novel and convenient methods for the assembly of pyridazine dioxides and azasydnones will encourage further investigations on the pharmacological activity of these compounds. Overall, each type of considered heterocyclic NO-donors has a strong potential in medicinal chemistry and drug design and we hope that the present review will stimulate future research in this field.