1000/1000
Hot
Most Recent

“Romantic love is a motivational state typically associated with a desire for long-term mating with a particular individual. It occurs across the lifespan and is associated with distinctive cognitive, emotional, behavioural, social, genetic, neural, and endocrine activity in both sexes. Throughout much of the life course, it serves mate choice, courtship, sex, and pair-bonding functions. It is a suite of adaptations and by-products that arose sometime during the recent evolutionary history of humans”.
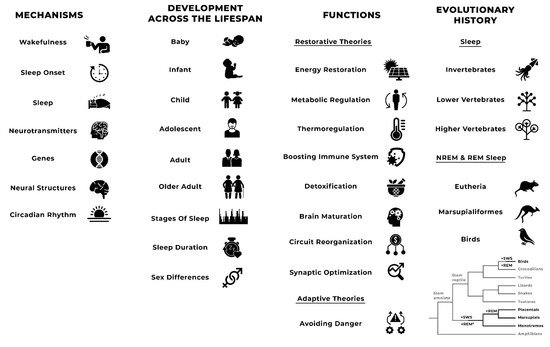
Sleep is common in the animal kingdom, although it takes various forms. Sleep in humans is defined “on the basis of both behaviour of the person while asleep and the related physiologic changes that occur to the waking brain’s electrical rhythm in sleep”. Behavioural characteristics of sleep include lack of mobility or slight mobility, closed eyes, a characteristic species-specific sleeping posture, reduced response to external stimulation, quiescence, increased reaction time, elevated arousal threshold, impaired cognitive function, and a reversible unconscious state. It includes non-rapid eye movement (NREM) sleep and rapid eye movement (REM) sleep. Non-rapid eye movement sleep is characterised by synchronised electroencephalographic activity, mildly reduced muscle tone, and slow rolling eye movements. Rapid eye movement sleep is characterised by theta or saw tooth waves and desynchronised electroencephalographic activity, moderately to severely reduced or absent muscle tone, and rapid eye movements. There are three stages of NREM sleep (i.e., N1, N2, N3) and one stage of REM sleep.
| Adolescents | Young Adults | Studies | |
|---|---|---|---|
| Sleep onset latency | - | Shorter | [37][38]; see also [35][36][72][73] |
| Sleep duration | Shorter | - | [72][74] *; see also [35][36][37][38][73] |
| WASO | - | Fewer | [37][38]; see also [35][36] |
| Sleep quality | - | Better | [37][38]; see also [72] and [35][36][37][73][74] |
| Restoring sleep | Increased | [37][38] |