1000/1000
Hot
Most Recent

Breast cancer has an extremely high incidence in women, and its morbidity and mortality rank first among female tumors. With the increasing development of medicine today, the clinical application of neoadjuvant chemotherapy has brought new hope to the treatment of breast cancer. Based on the relevant research on the existing drug resistance mechanism, the current treatment plan for reversing the resistance of breast cancer to neoadjuvant chemotherapy is explored, and the potential drug targets are analyzed, aiming to provide a new idea and strategy to reverse the resistance of neoadjuvant chemotherapy drugs in breast cancer.
Breast cancer is currently a cancer with an extremely high incidence in women, and its mortality rank first among female tumors [1]. According to the data of GLOBOCAN in 2018, about 2.1 million patients are diagnosed with breast cancer, and the death toll is 630,000 [2]. Today, with changes in the environment and lifestyle, the incidence of breast cancer is also increasing [3]. New statistics show that breast cancer still ranks first among female cancers. Therefore, overcoming breast cancer has increasingly become a problem of global concern.
In clinical treatment of breast cancer, surgery is usually combined with chemotherapy. With the development of biology and immunology, the approach to breast cancer treatment is constantly updated. In recent years, breast cancer has been considered a systemic disease, and neoadjuvant chemotherapy has also been included as an important part of the treatment of breast cancer.
Neoadjuvant chemotherapy refers to systemic chemotherapy before the implementation of local treatment methods (such as surgery or radiotherapy). It is mainly suitable for patients with mid-stage and locally advanced breast cancer. The concept of neoadjuvant chemotherapy was formally proposed by Rosen et al. in 1979. It aims to transform inoperable breast cancer into operable breast cancer, convert breast cancer that requires breast removal into breast-sparing breast cancer and provide drug basis in the follow-up treatment to improve the prognosis of patients [4].
At present, there is no uniform standard for neoadjuvant chemotherapy for breast cancer. In the early stage, a unified neoadjuvant chemotherapy regimen is generally given to all patients, but today’s neoadjuvant chemotherapy regimens tend to be personalized, as shown in Figure 1, generally based on curative effect prediction markers and molecular subtypes to give personalized treatment.
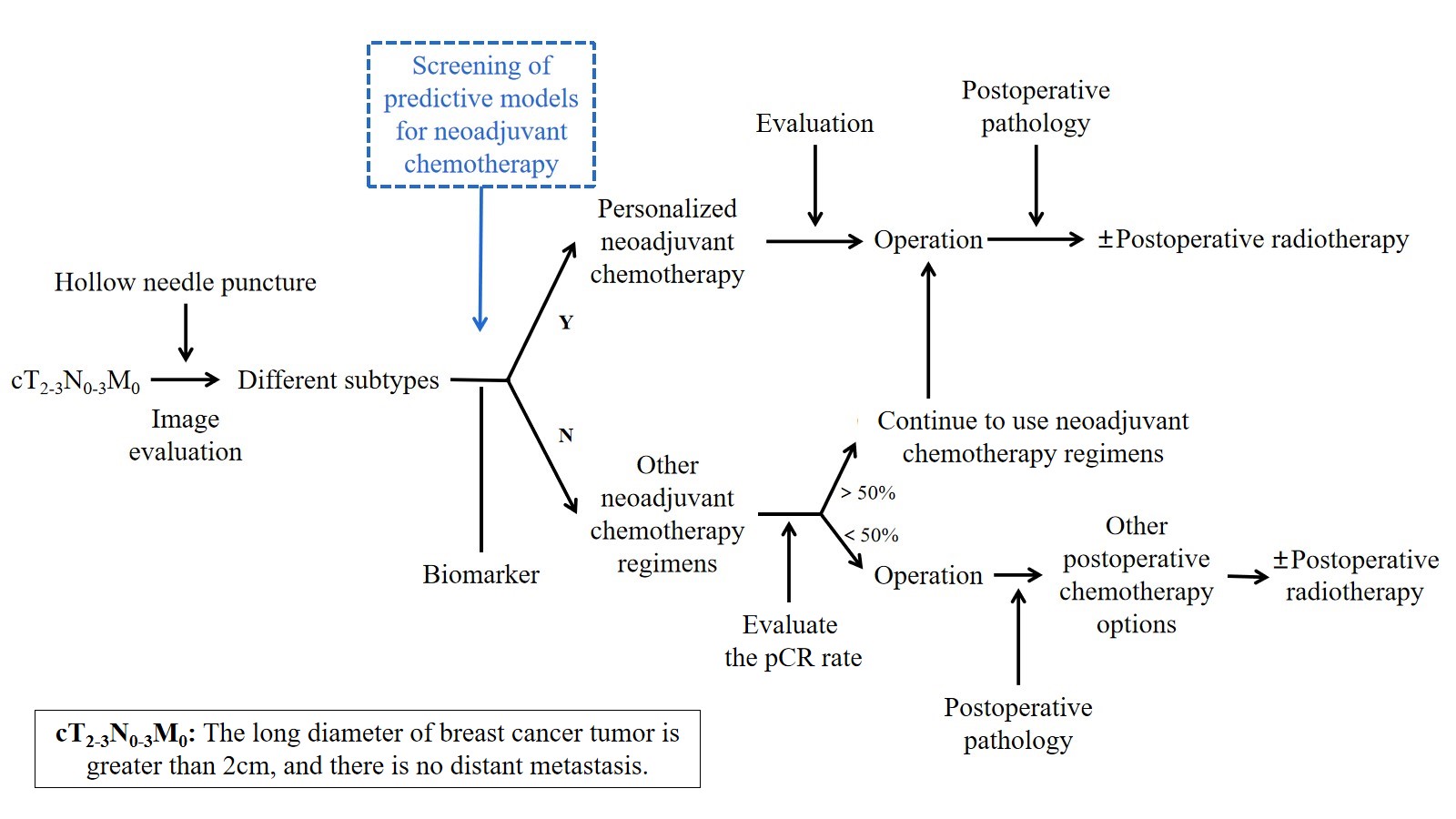
Figure 1. Personalized neoadjuvant chemotherapy.
In the use of chemotherapy drugs, anthracyclines, such as doxorubicin and epirubicin, are generally used in combination with drugs such as cyclophosphamide, epirubicin, and fluorouracil. The emergence of taxanes and their significant anti-tumor activity against advanced breast cancer have further improved the efficacy of neoadjuvant chemotherapy. In clinical use, no matter single drug or combination drugs, taxane drugs all show good anti-tumor activity. For breast cancer subtypes with HER2 overexpression, trastuzumab is generally added to the neoadjuvant chemotherapy regimen, and satisfactory results have been obtained in clinical use [5].
Although the efficacy of neoadjuvant chemotherapy has been confirmed, clinical trial data show that the effects of neoadjuvant chemotherapy for different breast cancer patients are very different, and it is easy to develop drug resistance, which is not conducive to subsequent treatment [6]. Drug resistance to neoadjuvant chemotherapy is one of the main reasons for its treatment failure, and it is one of the most challenging problems in the treatment of breast cancer today.
Neoadjuvant chemotherapy drug resistance is an important issue in clinical breast cancer treatment. At present, many studies are combining new drugs or new therapies with traditional neoadjuvant chemotherapy drugs, which can significantly reverse drug resistance caused by neoadjuvant chemotherapy drugs alone. The following chapter describes some drugs that have the potential to reverse neoadjuvant chemotherapy resistance. Although these drugs have not yet been clinically applied in breast cancer, it is believed that with research advances, these novel regimens can play an important role in the treatment of breast cancer.
According to the existing research on drug resistance mechanisms, many treatment options have brought hope to reverse the resistance of breast cancer to neoadjuvant chemotherapy. One of the more commonly used methods to reverse drug resistance is the combined use of multiple chemotherapy drugs.
On the one hand, the combined use of multiple drugs that work through different molecular mechanisms can greatly improve the problem of drug resistance caused by alterations in a single mechanism, thereby ensuring the efficacy of the drug. For example, in the clinical application of neoadjuvant chemotherapy drugs, anthracyclines that act on DNA and taxanes that act on proteins are used in combination, and drugs such as cyclophosphamide and epirubicin are used at the same time. These drugs act on different targets and can greatly improve the problem of drug resistance caused by a single mechanism, thereby ensuring the efficacy of the drug. Among neoadjuvant chemotherapy drugs for breast cancer, both trastuzumab and pertuzumab specifically act on the HER2 target, and their combined application can significantly enhance the sensitivity of breast cancer cells to neoadjuvant chemotherapy drugs. However, this method could inevitably enhance drug toxicity. In clinical treatment, it is also necessary to pay attention to the interaction between different drugs, detect the plasma drug concentration in time, and change the dose to obtain the best therapeutic effect.
On the other hand, while using neoadjuvant chemotherapy drugs, increasing the use of drugs that can block the drug resistance mechanism of tumors can restore the sensitivity of drug-resistant cells to drugs and enhance the efficacy of drugs. Most of these drugs are P-gp inhibitors, but due to high toxicity and pharmacokinetic effects, they have not been used in clinical practice [7]. The current research direction is to develop more efficient and low-toxic chemical drug reversal agents. Dual specificity phosphatase6 (DUSP6) inhibitors and histone deacetylase inhibitors (HDACI) have been identified as agents with the potential to reverse tumor drug resistance warranting follow-up research associated with drug development [8][9].
With the modern development of Chinese medicine, herbs and extractions from traditional Chinese medicine as drugs for treating tumors have gradually demonstrated their unique curative effects in the clinic [10][11][12][13]. More and more studies have shown that traditional Chinese medicine plays an important role in the prevention and treatment of breast cancer, contributing to the reversal of drug resistance of breast cancer. Moreover, compared with the above-mentioned chemical drug reversal agents, traditional Chinese medicine has the characteristics of safer, multi-component, multi-stage, and multi-targeted action. This makes traditional Chinese medicine monomers and extracts gradually attract more researchers’ attention as tumor drug reversal agents.
Many traditional Chinese medicine preparations have been developed and used in clinical practice, such as Elemene Injection and Shenqi Fuzheng Injection. The main active ingredient of Elemene Injection is a mixture of β-, γ-, and σ-elemene, which is an anti-cancer active ingredient extracted from Curcuma wenyujin Y.H.Chen. The combination of Elemene Injection and neoadjuvant chemotherapy drugs can reverse drug resistance of breast cancer cells, and its mechanism of action is mainly to inhibit the expression of P-gp, and reverse drug resistance of breast cancer cells through exosome and EMT inhibition [14][15][16][17]. Shenqi Fuzheng Injection is an injection made with Codonopsis and Astragalus as the main components. As early as 1999, Shenqi Fuzheng Injection has been formally approved by CFDA for adjuvant anti-tumor therapy [18]. Regarding its mechanism of reversing tumor drug resistance, the main mechanism is to induce cell cycle arrest and promote cell apoptosis [19]. However, it has not been used in neoadjuvant chemotherapy for breast cancer, and its mechanism of action should be further explored in the hope that it can be used in clinical practice in the future.
In addition to the above-mentioned traditional Chinese medicine compound prescriptions, there are many monomer drugs extracted from traditional Chinese medicines that have been proven to have the effect of reversing tumor resistance. Honokiol and magnolol, which are extracted from the plant Magnolia officinalis Rehd. et Wils., have a variety of pharmacological effects, such as inducing long-lasting central muscle relaxation and central nervous system inhibition, as well as anti-inflammatory, antibacterial, anti-ulcer, anti-tumor etc. Hyo-Kyung Han et al. [20] evaluated the inhibitory effects of honokiol and magnolol on P-pg activity, and found that honokiol can inhibit P-gp activity through a competitive mechanism, and both compounds can inhibit the expression of P-gp and help to improve the resistance of breast cancer to neoadjuvant chemotherapy drugs. Curcumin is derived from Curcuma longa L. In related studies, the combined treatment of curcumin and the neoadjuvant drug doxorubicin used in breast cancer can reduce the excessive efflux of doxorubicin caused by the overexpression of ABCB4, thereby enhancing the efficacy of doxorubicin [21]. Saikosaponin D is derived from Bupleurum chinense DC. It has been shown to have anti-inflammatory, antibacterial and anti-tumor effects. Relevant studies have shown that Saikosaponin D can enhance the sensitivity of breast cancer multidrug resistant cells MCF-7/ADR cells to chemotherapeutic drugs by reducing the expression of MDR1 and P-pg, which may effectively reverse the effects of neoadjuvant chemotherapy drug resistance [22]. Berberine is an alkaloid, extracted and purified from Coptis chinensis Franch. It has a variety of pharmacological effects, such as antibacterial, antihypertensive, antiarrhythmic and antitumor [23]. For reversing the drug resistance of breast cancer cells, different doses of berberine mediate different mechanisms. When using a low dose (5 µM) ofberberine, the AMPK/HIF-1α signaling pathway is inhibited and the expression of P-gp decreases, thereby increasing the sensitivity of breast cancer cells to chemotherapy drugs. The high dose (40 µM) of berberine can regulate the AMPK/HIF-1α signaling pathway, which in turn activates the expression of p53 and directly induces breast cancer cell apoptosis [24]. Resistance to paclitaxel drugs is a major obstacle in neoadjuvant chemotherapy for breast cancer, and studies have shown that gambogic acid can enhance the sensitivity of breast cancer cells to paclitaxel [25]. Gambogic acid is derived from Garcinia hanburyi Hook. f., it has anti-proliferative effects in triple-negative breast cancer cells [26]. It can also induce breast cancer cell apoptosis by inhibiting SHH signaling pathway, and has potential to be used as a combination drug in the neoadjuvant treatment of breast cancer. Ligustrazine is derived from the root of Ligusticum chuanxiong Hort. Various studies have shown that ligustrazine can block the G0/G1 phase of the cell cycle, thereby inhibiting DNA synthesis, and inducing breast cancer cell apoptosis, thereby reversing breast cancer cell resistance to neoadjuvant chemotherapy drugs [27].
Due to the complex composition of traditional Chinese medicine agents, numerous targets, and unclear mechanism of action, its follow-up research should also conduct efficacy and safety evaluations to accelerate the transformation to the clinic.
In recent years, the development of genetic engineering technology has made it possible to reverse the resistance of neoadjuvant chemotherapy drugs at the genetic level. Among them, nucleic acid-based technologies, such as siRNA, antisense oligonucleotide (ASO), mRNA, etc., have shown great potential in regulating the expression of tumor-related genes [28].
Mutations in the MEN1 gene encoding menin can cause tumors in multiple endocrine organs. For example, the occurrence and development of breast cancer are closely related to menin. Current studies have shown that inhibitors of the menin/MLL1 complex are effective in some cancers, but they are not effective in the treatment of breast cancer [29]. Recent studies have found that ASO targeting menin mRNA has a greater advantage than siRNA, and has a better curative effect in the treatment of triple-negative breast cancer [30]. More importantly, in vitro menin silencing can have a synergistic effect with the taxane drug docetaxel in neoadjuvant chemotherapy. Moreover, menin-ASO can be used in combination with neoadjuvant chemotherapy drugs that induce DNA damage or PARP inhibitors that inhibit DNA damage. It can promote cell apoptosis, which provides new ideas for neoadjuvant chemotherapy for breast cancer.
The upregulation of some pro-apoptotic genes, such as p53, can also reverse resistance of neoadjuvant chemotherapy drugs. In breast cancer cells, the tumor suppressor gene p53 is often mutated to cause abnormal expression, thereby activating the promoter of the MDR1 gene and increasing its expression. At present, the p53 gene using adenovirus as a vector, and the antisense gene of the mutant p53 gene can be used to introduce the wild-type p53 gene can be potentially used to reverse the resistance to neoadjuvant chemotherapy drugs in breast cancer [31][32].
MicroRNA (miRNA) is an endogenous 18–23-nucleotide small noncoding RNA, which can bind to the 3′-untranslated region (3’-UTR) of specific target messenger RNAs (mRNAs). Abnormal expression of miRNA, such as the oncomiRNA, microRNA-21, is overexpressed in breast cancer, which may lead to the occurrence of breast cancer, thus the regulation of miRNA is also a potential target for the treatment of breast cancer [33]. Anti-microRNA oligonucleotides (AMOs) are synthetic oligonucleotides that can complementally bind to the corresponding target miRNA, thereby inhibiting the expression of miRNA [34]. At present, most AMOs are designed for high-expressed miRNAs in breast cancer cells. Recent studies have shown that inhibiting low-expressed miRNAs in breast cancer cells, such as mir-148a of the mir-148/152 family, can also effectively inhibit the proliferation of breast cancer cells to treat breast cancer [35]. These studies show that the combination of AMO for the suppression of related miRNA expression and neoadjuvant chemotherapy has great potential to enhance the efficacy of neoadjuvant chemotherapy drugs and bring new hope for the treatment of breast cancer.
Genetic engineering mainly blocks the expression of drug-resistant genes, or acts synergistically with neoadjuvant chemotherapy drugs to promote breast cancer cell apoptosis. The development and clinical application of genetic engineering technology can effectively reverse the resistance of neoadjuvant chemotherapy drugs and improve the effect of clinical treatment of breast cancer. However, the drug resistance mechanism of breast cancer is complex, including multiple pathways and factors. For different mechanisms, the research direction should be focused on more approaches and more targets to achieve more therapeutic effects.
The current use of the immune system to reverse resistance to neoadjuvant chemotherapy drugs is mainly associated with the application of antibodies and the application of many cytokines in the body’s own immune system.
Antibodies include mainly P-gp antibodies and APO-1 monoclonal antibodies. P-gp antibodies can recognize the epitope of the P-gp membrane, competitively inhibit the pumping function of P-gp, reduce neoadjuvant chemotherapy drug efflux, enable drug accumulation in the cell, and reverse resistance to neoadjuvant chemotherapy drugs [36]. In addition, antibodies can mediate immune responses and participate in the reversal of drug resistance. The APO-1 monoclonal antibody is an antibody against the glycoprotein FAS on the cell membrane surface, which can bind to APO-1 on the breast cancer cell membrane surface to induce cell apoptosis [37]. For some breast cancer cells with low FAS expression, the FAS antigen expression vector can also be transfected into cells to promote high expression before using APO-1 monoclonal antibodies.
The cytokines of the body’s immune system, such as TNF-α, INF-α and IL-2, can reduce the expression levels of MDR1 gene mRNA and P-gp, increase the sensitivity of breast cancer cells to neoadjuvant chemotherapy drugs, and reverse resistance to neoadjuvant chemotherapy drugs [38].
TME and tumor cells interact with each other. Tumor cells can create a suitable living environment for themselves by changing TME, and TME in turn regulates tumor cells, including promoting drug resistance. At present, TME is a hot spot in tumor research. It can provide new ideas for tumor treatment and reverse tumor drug resistance to a certain extent [39].
In addition to the above-mentioned HIF inhibitors and targeted inhibitors developed for the TME hypoxic environment and acidic environment, current research focus is the use of nanomaterials when analyzing TME models, and the focuses on the mechanism of nanomedicine targeting CAFs [40].
Moreover, the combination of anti-angiogenic therapy and immunotherapy can lead to a reversal of the immunospressive TME [41]. In breast cancer, anti-angiogenic therapy can induce the normalization of tumor blood vessels, and promote the recruitment of immune cells and the maturation of dendritic cells (DC) [42]. The use of immune checkpoint inhibitors can further alleviate the immunosuppressive state and promote the normalization of TME, thereby reversing resistance of neoadjuvant chemotherapy drugs induced by TME.
More importantly, the heterogeneity of TME is related to breast cancer subtypes, and the treatment plan for TME will help realize the potential of “precision medicine.”
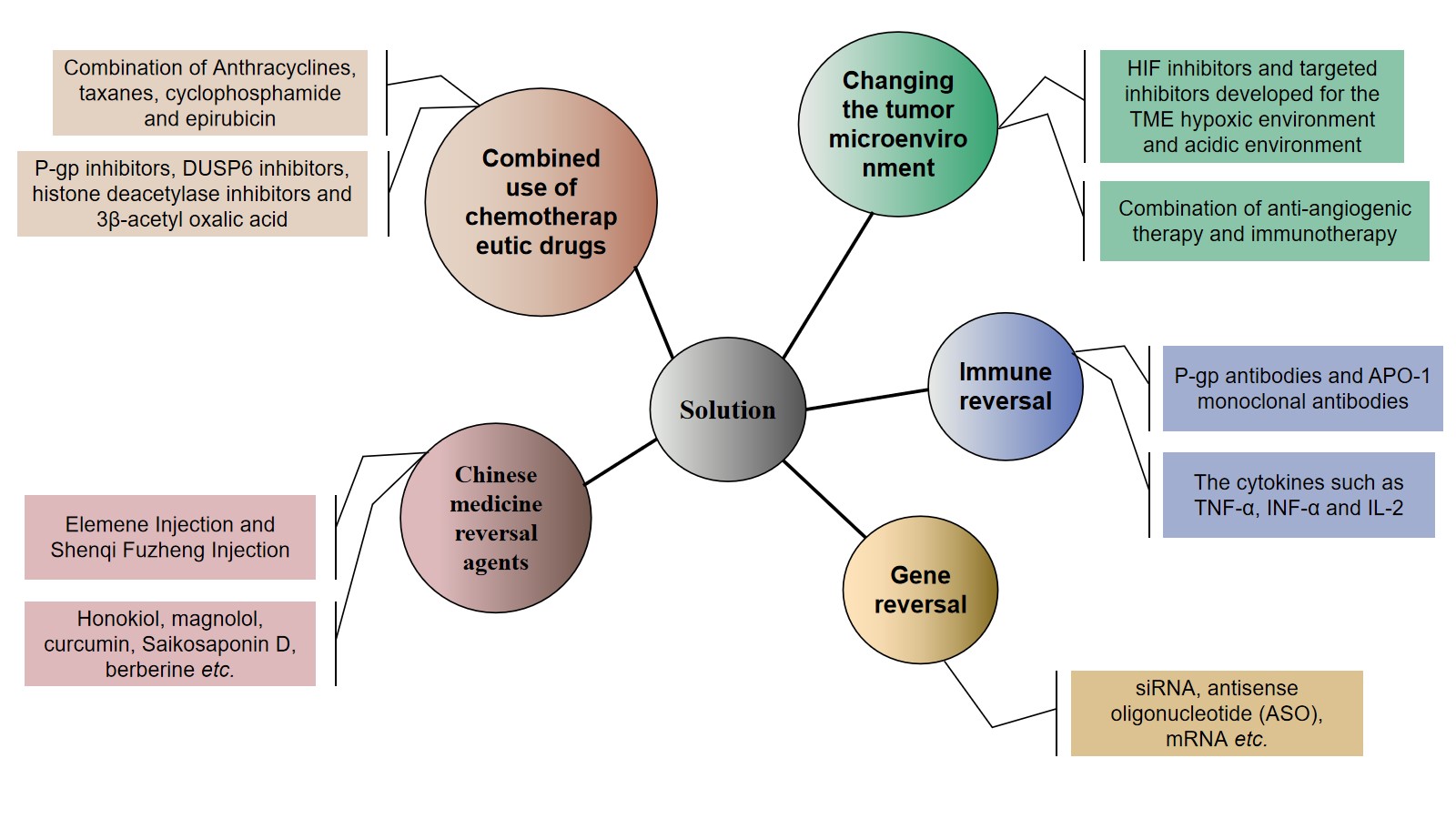
Figure 2. Strategies to combat resistance to neoadjuvant chemotherapy drugs in breast cancer.