1000/1000
Hot
Most Recent

Thyroid cancer is the most common endocrine malignancy, representing 2.9% of all new cancers in the United States (US). It has an excellent prognosis, with a five-year relative survival rate of 98.3%, and has a higher frequency in females than males, with ratio of 3:1. Differentiated thyroid cancer (DTC) represents 90% of all thyroid malignancies and includes three main types: papillary thyroid cancer (PTC), the most common type, comprising 85% of all DTC; follicular thyroid cancer (FTC); and the rare subtype, Hürthle (oncocytic) cell thyroid cancer (2–5%).
Thyroid cancer is the most common endocrine malignancy, representing 2.9% of all new cancers in the United States (US). It has an excellent prognosis, with a five-year relative survival rate of 98.3%, and has a higher frequency in females than males, with ratio of 3:1 [1]. Differentiated thyroid cancer (DTC) represents 90% of all thyroid malignancies and includes three main types: papillary thyroid cancer (PTC), the most common type, comprising 85% of all DTC; follicular thyroid cancer (FTC); and the rare subtype, Hürthle (oncocytic) cell thyroid cancer (2–5%) [2].
In the last ten years, the incidence of DTC has dramatically increased. This tendency is mainly as a result of the diffusion of imaging systems, the use of ultrasound-guided Fine Needle Aspirations Cytology (FNAC), and improvements in histological evaluations [3][4].
Actually, papillary microcarcinomas (PMCs) represents the most diagnosed thyroid tumors, with a 35% incidence of occult papillary thyroid microcarcinomas in autopsy studies [5]. The increased diagnosis of these malignancies, associated with a low risk of recurrence and death, has led to the need for redefining of the multimodal therapeutic approach to avoid potential overtreatments. With regard to treatment, historically, the only option was surgery. In the last few years, active surveillance (AS) has been established as an alternative approach; it is aimed at identifying patients who would likely benefit from rescue surgery [6].
Considering the data in the literature and the available evidence, we reviewed the current management of low-risk DTC, and PMCs in particular, to better clarify the indications for active surveillance for low-risk thyroid cancers.
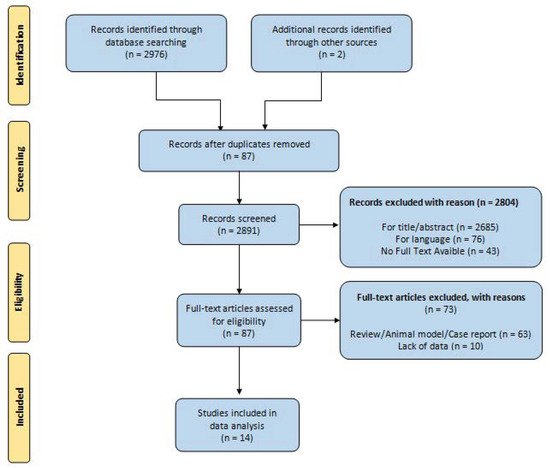
According to the PRISMA flowchart (Preferred Reporting Items for Systematic reviews and Meta-Analyses), a systematic search was performed of electronic databases (PubMed, Web of Science, Scopus, and EMBASE). We used medical subject headings (MeSH) and free-text words, using the following search terms in all possible combinations: “differentiated thyroid cancer”, “micro papillary cancer”, “management”, and “active surveillance”. The last search was performed in May 2021. Attention was focused on the following primary outcomes: growth of the primary tumor, metastatic disease (lymph node or extra nodal), tumor recurrence after delayed thyroid surgery (DTS), and thyroid-cancer-related mortality. The secondary outcomes selected were decreased volume of primary tumor (>3 mm), overall mortality, and incidence of/indication for thyroidectomy.
The retrospective application of the surveillance criteria to patients surgically treated for thyroid nodules was not one of inclusion criteria for the studies reporting on AS of low-risk PTC; AS was limited to employment of surveillance strategies. Low-risk PTC was defined as T1a or T1b, N0, M0 disease. The search strategy was limited to articles written in the English language; moreover, papers on animal studies, review articles, editorials, and case series were excluded.
R.P. and G.C., two independent authors, analyzed all the papers, selected the suitable manuscripts, and performed the data extraction independently. All duplicate studies were removed. Two other authors (N.V. and S.M.) then checked the eligibility of the studies selected. Discrepancies were resolved by consensus.
We identified a total of 2976 articles, of which 87 articles were selected for full text review. After full text review, 14 studies were included for the final analysis. The results are summarized in the PRISMA flowchart ( Figure 1 ) [7][8][9][10][11][12][13][14][15][16][17][18][19][20].
A total number of 4830 patients under AS were included in this review.
All studies assessed tumor growth during AS; a total of 451/4830 (9.4%) patients experienced an increase in diameter of up to 3 mm. Conversely, in five studies, a decrease in tumor size > 3 mm was assessed in 172/1324 (12.9%) patients during AS.
DTS after AS was performed in 609/4830 (12.6%) patients, as reported by all authors. Ten authors reported involvement of cervical lymph nodes during AS in 88/4213 (2.1%) patients, while in six studies, 4/3589 (0.1%) patients were reported to have extra-nodal metastatic disease. No study reported mortality due to thyroid cancer during AS.
Active surveillance should be proposed for low-risk PMC only after clinical trials proving the validity of this approach. For high-risk PMC, (lymph node or distant metastasis, extra thyroid extension, closeness to recurrent laryngeal nerve or trachea, high-grade cytology, or growth during observation), a surgical approach (lobectomy with or without paratracheal dissection) is necessary. Better knowledge of papillary cancer natural history and biological behavior might be useful in the design of multimodal management.