Nanoparticles are nanomaterials with three external nanoscale dimensions and an average size ranging from 1 to 1000 nm. Nanoparticles have gained notoriety in technological advances due to their tunable physical, chemical, and biological characteristics. However, the administration of functionalized nanoparticles to living beings is still challenging due to the rapid detection and blood and tissue clearance by the mononuclear phagocytic system. The major exponent of this system is the macrophage. Regardless the nanomaterial composition, macrophages can detect and incorporate foreign bodies by phagocytosis. Therefore, the simplest explanation is that any injected nanoparticle will be probably taken up by macrophages. This explains, in part, the natural accumulation of most nanoparticles in the spleen, lymph nodes, and liver (the main organs of the mononuclear phagocytic system). For this reason, recent investigations are devoted to design nanoparticles for specific macrophage targeting in diseased tissues. The aim of this review is to describe current strategies for the design of nanoparticles to target macrophages and to modulate their immunological function involved in different diseases with special emphasis on chronic inflammation, tissue regeneration, and cancer.
1. Introduction
Macrophages are plastic cells from the innate immune system that play different roles in the development, homeostasis, tissue repair, and immune response [1]. The local tissue microenvironment determines the macrophage polarization phenotype, M1-like or M2-like, in such a way that populations of both subsets can be found simultaneously coexisting in the same tissue. Macrophages are classically activated into the pro-inflammatory M1 phenotype in response to inflammatory stimuli such as lipopolysaccharides (LPS) or interferon-γ (IFN-γ). On the other hand, interleukin-4 (IL-4) and IL-13 alternatively activate the macrophage polarization to an anti-inflammatory M2 phenotype [2]. Polarized macrophages can be also reprogrammed by the combination of different agents promoting a phenotype reversion [3].
Damaged cells release specific molecules known as damage-associated molecular patterns (DAMPs) that activate the immune system in an analogous manner to pathogen-associated molecular patterns (PAMPs), small molecular motifs released from bacteria or viruses [4]. These endogenous molecules display physiological functions in cells, but are recognized as danger signals when released into the extracellular space leading to downstream inflammation. Therefore, tissue-specific macrophage subpopulations detect signals that are not found in healthy tissues following infection or injury and recruit monocytes that differentiate into macrophages [2]. DAMPs, PAMPS, or IFN-γ secreted by lymphocytes induce the pro-inflammatory M1 macrophage phenotype [3]. M1 macrophages secrete a variety of pro-inflammatory mediators such as IL-1 and tumour necrosis factor (TNFα) that stimulate inflammation, and IL-12, which activates T helpers 1 (TH1), initiating the adaptive immune response [2]. M1 macrophages also secrete reactive oxygen species (ROS) and nitrogen species that contribute to the elimination of invading organisms. During this process, they also trigger substantial collateral tissue damage to the host. To prevent further tissue damage due to the inflammatory macrophage response, macrophages undergo apoptosis or polarization to an anti-inflammatory and pro-regenerative phenotype that dampens the pro-inflammatory response and facilitates wound healing [1]. IL-4 and IL-13 alternatively activate the macrophage polarization to an anti-inflammatory M2 phenotype [2]. M2 macrophages secrete anti-inflammatory cytokines such as IL-4, IL-13, or IL-10 to dampen the proinflammatory response [2] and specific and numerous growth factors such as transforming growth factor (TGFβ1) and vascular endothelial growth factors (VEGFs) to promote cell proliferation and angiogenesis [5]. M2 macrophages can also regulate the proliferation and expansion of neighboring parenchymal and stromal cells and the activation of stem cells and local progenitor cell populations that participate in repair [5].
The inflammatory and anti-inflammatory responses orchestrated by macrophages need to be accurately regulated to prevent disease. Cytokines are the signals that mediate the coordination between immune cells to harmonize the balance between inflammation and tissue repair [6]. Imbalance of M1/M2 macrophage populations is associated with different diseases [3]. Uncontrolled inflammatory response driven by macrophages leads to chronic inflammation and autoimmune diseases [1]. Similarly, the dysregulation of anti-inflammatory response can contribute to tumour progression and metastasis [7]. In addition, prolonged inflammation and continuous activation of macrophages results in chronic diseases that may lead to the development of pathological fibrosis. In some diseases, extensive fibrosis can ultimately lead to organ failure and death [5].
Different types of biomaterials such as nanoparticles (NPs) and hydrogels are being extensively developed to target macrophages. Since macrophages are professional phagocytic cells, NPs can be exploited as vehicles that naturally target macrophages. These immune cells can easily incorporate NPs via phagocytosis, macropinocytosis, or receptor-mediated endocytosis [8]. Some types of NPs can interact with macrophages to directly modify their biological functions [6]. In addition, NPs can be used as drug delivery systems to treat macrophages involved in different diseases [9]. Several therapeutic options using functionalized NPs are being explored and developed to modulate macrophages.
This review summarizes the use of NPs to modulate macrophages involved in the initiation and progress of different diseases. We will particularly focus on NPs to target and treat macrophages involved in diseases characterized by chronic inflammation and in tumor-associated macrophages (TAMs).
2. Nanoparticles to Modulate Macrophages in Chronic Inflammation
The persistence of the harmful agent and the propagation of the inflammatory response leads to the imbalance between inflammatory and anti-inflammatory signals. In this scenario, macrophages are potential targets to treat inflammatory disorders such as rheumatoid arthritis, atherosclerosis, and inflammatory bowel disease. Drug-loaded NPs can be used to modulate the immunological activity of the pro-inflammatory M1 macrophages or to switch their phenotype from M1 to M2 [10][11]. These therapeutic strategies are designed to shift the dynamic balance from pro- to anti-inflammatory signals to mitigate the pathological process [3].
2.1. NPs Modulating Macrophages in Rheumatoid Arthritis
Rheumatoid Arthritis (RA) is an autoimmune disease characterized by stiffness, pain, and swelling of several joints
[12]. Macrophages play a major role in synovium inflammation. Jain et al. demonstrated that the injection of non-condensing alginate-based NPs encapsulating pDNA coding for IL-10 and modified with a tuftsin peptide (to target macrophages) was therapeutically effective treating RA in arthritic rats
[13]. M1 macrophages were successfully reprogrammed by these NPs to M2 macrophages preventing the progression of inflammation and joint damage.
Other strategies have been designed to target the synthesis of TNFα and IL-1 (both main pro-inflammatory cytokines involved in RA pathogenesis). NP-mediated TNFα knockdown in macrophages has been reported as an effective treatment for arthritis in murine models
[14]. Chitosan/small interference RNA (siRNA) NPs have been used to down-regulate TNFα expression to promote systemic anti-inflammatory effects
[15]. Down-regulation of TNFα and IL-1 has also been achieved by folate–Chitosan DNA NPs containing an Interleukin-1 Receptor Antagonist
[16]. These NPs demonstrated a precise activity blocking the effects of IL-1 by interfering with its binding to IL-1R, thus ameliorating joint damage in RA experimental models.
Folate receptors (FR) have also been targeted to treat RA
[17]. It has been reported that the expression of FRβ is abundant on activated macrophages and plays an important role on RA
[18]. The folate antagonist methotrexate (MTX) has been the primary drug used to treat RA for many years. Some groups have used NPs targeting FR to deliver MTX. Thomas and co-workers have designed folic acid (FA)-conjugated G5 dendrimers to target macrophages in vitro to deliver MTX in a collagen-induced arthritis (CIA) murine model
[19]. Zhao and colleagues have developed FRβ-targeting pH-responsive nanocarriers loaded with MTX in rat model of adjuvant-induced arthritis
[20]. These nanocarriers responded to an acidic pH delivering the drug efficiently and reducing the progression of RA.
2.2. NPs Modulating Macrophages in Inflammatory Bowel Disease (IBD)
Inflammatory bowel disease (IBD) is characterized by chronic inflammation of the gastrointestinal (GI) tract. There are two types of IBD: Crohn disease (CD) and ulcerative colitis (UC) [6]. Macrophages are an important source of proinflammatory cytokines (such as TNFα) that play an important role on the pathogenesis of IBD. TNFα has become an attractive target for IBD therapy using NPs. Most studies have investigated the use of different NPs containing TNFα siRNA as an interesting therapeutic strategy. Xiao and colleagues have used mannosylated NPs as delivery vehicles for TNFα siRNA [21]. TNFα siRNA demonstrated an effective activity to drastically reduce the TNFα expression and promoted anti-inflammatory effects in vitro and ex vivo leading to colitis attenuation in a dextran sodium sulfate (DSS)-induced colitis mouse model [21]. Similar results were obtained by H. Laroui and colleagues. They designed NPs with a Fab’ portion of the F4/80 antibody against murine macrophages, which also contained TNFα-siRNA showing high efficiency in the attenuation of colitis [22]. Wilson and colleagues have also encapsulated TNFα siRNA in NPs, but they have designed stimulus-responsive NPs. They have developed thioketal NPs (TKNs) that are reactive to high concentration of ROS [23]. Other strategies have combined the delivery of TNFα siRNA with siRNA against Cyclin D1 [24]. Kriegel and Amiji have used NPs in microsphere oral systems for dual siRNA (TNFα and Cyclin D1) delivery [24]. This dual treatment has shown to be more effective than each agent separately for treating IBD in a DSS-induced mouse model.
2.3. NPs Modulating Macrophages in Atherosclerosis
Numerous potential therapies based on NPs targeting macrophages have been developed to treat atherosclerosis. Atherosclerosis is a chronic inflammatory disease characterized by the narrowing and thickening of the arteries caused by the build-up of plaques containing low-density lipoproteins (LDLs) and immune cells in the artery wall
[25]. Some strategies for the treatment of atherosclerosis have focused on reducing LDL accumulation in macrophages. Macrophages uptake oxidized LDLs through scavenger receptors (SRs) and release LDLs through the lipid transporters ABCA1 and ABCG1
[26]. Some authors have designed NPs to reduce the macrophage expression of SRs such as SR-A or oxidized LDLs receptor 1 (LOX-1) using siRNA in murine models
[27][28]. Other authors have used mannose functionalized dendrimer NPs (mDNPs) to deliver the liver-x-receptor (LXR) ligand, which regulates the expression of cholesterol transporters
[29]. Other authors have used NPs to deliver siRNA to block the expression of chemokine receptors associated with monocyte recruitment as a potential treatment for atherosclerosis
[26].
A different strategy for the treatment of atherosclerosis is the modulation of macrophages to enhance efferocytosis. Tao et al. have tested the efficacy of S2P-conjugated 1,2-Distearoyl-sn-glycero-3-phosphorylethanolamine (DSPE) polyethylene glycol (PEG) NPs delivering siRNA to macrophages to block the expression of CaMKIIγ and they have demonstrated that this therapy promotes plaque stabilization in mice
[30].
3. Nanoparticles to Stimulate Tissue Repair and Regeneration
M2 macrophages are actively involved in tissue repair and wound healing. For this reason, many investigations nowadays are dedicated to design NPs to stimulate the M2 macrophage phenotype for tissue or organ regeneration [31].
3.1. NPs Modulating Macrophages in Myocardial Infarct Repair
Myocardial infarction (MI) is the result of partial or complete coronary artery occlusion that leads to blood flow reduction [6][32]. The appropriate myocardial healing is guided by macrophages [26]. For this reason, numerous NPs have been developed to target macrophages and achieve myocardial infarct repair.
Harel-Adar et al. have investigated intravenous administration of phosphatidylserine (PS)-presenting liposomes as a new strategy to achieve myocardial infarct repair in a rat model of acute MI
[33]. These authors designed liposomes with PS to mimic apoptotic cells and to induce macrophage transition and myocardial infarct repair. Cardiac healing has also been investigated by Bejerano and colleagues. They delivered miRNA-21 encapsulated into hyaluronan-sulfate (HAS)-Ca
2+ NPs to cardiac M1 macrophages at the infarct zone inducing their repolarization to an anti-inflammatory and reparative M2 phenotype
[32]. miRNA-21 upregulation increased angiogenesis, reduced the number of apoptotic cells, and improved cardiac healing in mice by promoting inflammation resolution.
Courties et al. incorporated siRNA targeting interferon regulatory factor 5 (transcription factor that up-regulates genes associated with M1 macrophages) into lipidoid NPs to achieve attenuation of M1 macrophage polarization, inflammation resolution, and tissue regeneration in a mouse model of MI
[34]. M. Tokutome et al. achieved M2 polarization and subsequent cardiac healing in animal models encapsulating pioglitazone in PLGA NPs, activating the macrophage peroxisome proliferator-activated receptor-gamma (PPARγ)
[35]. PPARγ is a master regulator of inflammatory responses.
3.2. NPs Modulating Macrophages in Chronic Liver Injury
During the past decades, it has been demonstrated that hepatic macrophages hold central functions in the initiation, propagation, and perpetuation of inflammation in liver injury
[36]. He and co-workers have suggested the use of mannose-modified trimethyl chitosan-cysteine (MTC) conjugate NPs containing TNFα siRNA
[37]. These NPs attained a significant reduction in TNFα expression, blocked liver injury progression and reduced mortality in mice with acute hepatic injury.
Other authors have described PLGA NPs with a Spleen Tyrosine kinase (SYK) pathway inhibitor to target and treat macrophages in chronic liver injury
[38]. SYK inhibition using these NPs resulted in a significant reduction in inflammation, hepatic injury, and fibrosis in mice. Similar results have been obtained in experimental models of acute and chronic liver injury in mice using liposomes loaded with dexamethasone
[39]. Dexamethasone induced anti-inflammatory M2 polarization of hepatic macrophages and reduction of liver injury and liver fibrosis.
Other authors have focused on stimulating the selective production of collagenases to reduce liver fibrosis
[40]. Dendrimer-graphene nanostars have been designed to deliver a plasmid expressing the collagenase metalloproteinase 9 into inflammatory macrophages in cirrhotic livers. These NPs efficiently degraded collagen in vitro and promoted a phenotypical transformation from inflammatory to pro-regenerative and anti-inflammatory macrophages in a mouse model of liver fibrosis caused by chronic intraperitoneal administration of CCl
4. This targeted gene therapy reduced selectively and locally the presence of collagen fibers, decreased hepatic injury, and allowed hepatic regeneration
[40].
4. Nanoparticles to Target and Treat Tumour-Associated Macrophages
4.1. Specific Differential Phenotype of Tumour-Associated Macrophages
Tumor-associated macrophages (TAMs) are the most abundant immunosuppressive cells in the tumor microenvironment. They play a fundamental role in the promotion of tumor initiation, growth, and progression
[41]. In the initial phase of cancer, they activate antitumor immunity. When the tumor is firmly established, they promote angiogenesis, immunosuppression, and metastasis
[42][43]. Numerous tumor-derived chemoattractants are crucial for recruiting monocytes into the tumor milieu and to promote their transition to TAM. These include chemokines such as CXCL12 (stromal cell-derived factor 1α), CCL3 (macrophage inflammatory protein (MIP) 1α), CCL2 (MCP-1) and CCL4 (MIP1β), interleukins (IL-6 and IL-1β), and cytokines (colony stimulating factor 1 (CSF-1) and VEGF A)
[44][45][46][47][48].
TAMs have been recognized as potential therapeutic targets for cancer immunotherapy. Macrophage-related therapeutic approaches are already in clinical trials, but they still need to be supplemented with conventional cancer treatments such as standard cytoreductive therapies, angiogenesis inhibitors, and check-point inhibitors immunotherapy
[41].
NPs have been widely designed as drug delivery systems to treat cancer. They provide the opportunity to deliver drugs directly to TAMs. Engineered NPs can target TAM components and transform immunosuppressive TAMs into immunocompetent macrophages to improve the efficacy of cancer immunotherapy
[49].
4.2. NPs to Target TAM for Cancer Diagnostics and Prognosis
The fact that TAM display a very particular phenotype can be used to design selective diagnostic strategies for some types of solid tumors and even to define their prognosis because TAM invasion into tumor stroma has been associated with a worse prognosis in several types of cancers
[44]. NPs designed to target tumors are preferentially phagocytosed by macrophages as any other NPs. In this context, Leimgruber and colleagues have demonstrated that AMTA680 magnetofluorescent injectable NPs label endogenous TAM allowing the tracking of these immune cells within the microenvironment of soft tissue sarcoma, lung carcinoma, and colon adenocarcinoma
[50]. AMTA680 NPs consisted of a fluorescent dye for optical imaging (VT680) that emitted in far-red wavelength and a super-paramagnetic core for MRI. AMTA680 preferentially targeted “M2-like” TAMs expressed high levels of F4/80 and VEGF
[50]. Iron oxide NPs have also been employed for MRI diagnosis in tumors and to quantitatively monitor the TAM presence in breast cancer
[51]. Ferumoxytol NPs (clinically approved iron NPs that are naturally engulfed by TAM) have been used for the MRI diagnosis of anaplastic thyroid cancer
[52] and melanoma
[53]. Perfluorocarbon nanoemulsions have also been employed for murine breast cancer MRI diagnosis
[54]. Pérez-Medina et al. have developed an
89Zr-modified reconstituted HDL (rHDL) nanotracers for TAM PET imaging in a murine model of breast cancer
[55]. These nanotracers accumulated in TAM-rich tumor areas. The rHDL were prepared by mixing dimyristoylphosphatidylcholine (DMPC) vesicles with apoA-I resulting in discoidal NPs
[55].
64Cu loading in NPs has been investigated for PET imaging
[56]. Kim et al. have demonstrated that
64Cu-labeled polyglucose NPs were effective for PET breast cancer diagnosis. In another study, Locke et al. used mannosylated liposomes loaded with
64Cu for PET pulmonary adenocarcinoma diagnosis
[57]. Near-infrared fluorescent silica coated iron oxide NPs have also been used for the accurate delineation of glioblastoma multiforme
[58]. This type of glioblastoma has abundant infiltration of macrophages at the tumor margins. Fluorescent labeling of TAM served for its complete surgical resection. Indeed, these authors demonstrated that these NPs cross the blood–brain barrier
[58].
4.3. NPs to Inhibit Macrophage Recruitment and to Deplete TAM in Tumors
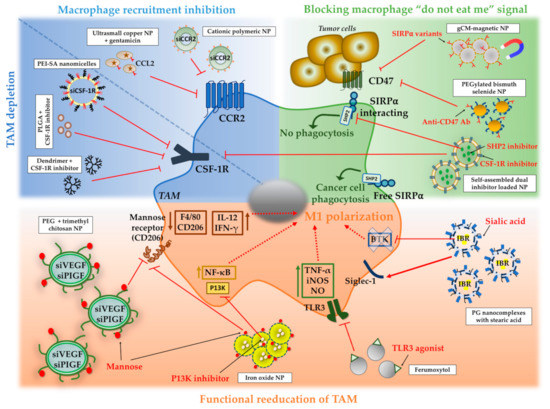
In recent years, different strategies have been suggested for TAM-targeted therapies. In the first stages of cancer, many efforts have been focused on designing NPs to prevent macrophage recruitment to the tumor area (
Figure 1). The inhibition of pivotal signaling pathways in monocyte recruitment, such as CCL2/CCR2, has been investigated to design NPs that block monocyte accumulation in tumors and transformation to TAM
[49][59][60]. Shen et al. designed cationic polymeric NPS shaped with copolymer poly(ethylene glycol)-block-polylactide (PEG5K-b-PLA11K) and a cationic lipid (
N,
N-bis(2-hydroxyethyl)-
N-methyl-
N-(2-cholesteryloxycarbonyl aminoethyl) ammonium bromide) that were engulfed by circulating Ly6C
hi monocytes
[59]. These NPs encapsulated a siRNA that blocked CCR2 receptor expression preventing TAM formation in an orthotopic murine model of breast cancer
[59]. In a different study, ultrasmall copper NPs were designed to target CCR2 with an antibody
[60]. These NPs encapsulated a chemotherapeutic agent resulting in a targeted therapy for pancreatic ductal adenocarcinoma with reduced toxicity.
Figure 1. Strategies for the targeting of TAM using NPs. Four main strategies have been described for TAM-targeted therapies: (1) inhibition of macrophage recruitment to the tumor (blue); (2) TAM depletion in the tumor microenvironment (blue); (3) blockade of macrophage immune checkpoint (“do not eat me” signal) (green); and (4) functional re-education of TAM to a M1 phenotype (orange). The image illustrates different molecular targets and different types of NPs that have been described to treat TAM. NP: nanoparticles; PEI-SA: polyethylenimine and stearic acid; PLGA: poly (lactic-co-glycolic acid); CSF-1R: Colony stimulating factor 1 receptor; TAM: tumor-associated macrophages; CCR2: C-C Motif Chemokine Receptor 2; SHP2: Src homology region 2 phosphatases; SIPRα: macrophage signal-regulatory protein alpha; Ab: antibody; gCM: genetically engineered cell membrane; PEG: polyethylene glycol; VEGF: vascular endothelial growth factor; PIGF: placental growth factor; IFNγ: interferon γ; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; P13K: Phosphoinositide 3-kinases; TNF-α: tumor necrosis factor; iNOS: nitric oxide synthases; NO: nitric oxide; TLR: toll-like receptor; IBR: Ibrutinib; BTK: Bruton’s tyrosine kinase.
Depletion of TAM has also been reported as a potential strategy to treat the tumor microenvironment and to prevent tumor progression and dissemination (
Figure 1). Cancer cells secrete CSF-1 to activate CSF-1/CSF-1R pathway and TAM formation. NPs targeting CSF-1R have been suggested as a possible therapy for TAM depletion. For example, a composition of polyethylenimine and stearic acid (PEI-SA) nanomicelles containing a siRNA against CSF-1R has been used to block CSF-1 receptor
[61]. Qian et al. have also used NPs containing a siRNA against CSF-1R
[62]. These NPs consisted of 1,2-dimyristoyl-sn-glycero-3-phosphocholine (DMPC) and cholesterol oleate. TAM targeting was carried out with a peptide with high specificity for M2 macrophages (M2pep)
[62]. The use of NPs loaded with CSF-1R inhibitors, such as BLZP945 and PLX3397, has also been suggested by several authors
[63][64][65][66]. PLGA NPs loaded with PLX3397 have been designed to deplete TAM in B16F10 tumors, a melanoma experimental model
[63]. Cuccarese and colleagues have designed fluorescent injectable NPs loaded with the same PLX3397 CSF-1R inhibitor to treat murine pulmonary carcinoma
[66]. Both types of NPs reduced “M2-like” TAM number. NPs targeting CSF-1 in TAM have also demonstrated improved response to therapy in glioblastoma
[64]. This study was carried out using hydroxyl dendrimer NPs with BLZ945 as CSF-1R inhibitor
[64]. This therapeutic approach resulted in “M2-like” TAM depletion (a decrease in arginase levels) and intratumoral accumulation of cytotoxic T cells indicating an increase in antitumor immunological activity
[64]. Dextran-grafted poly(histidine) copolymer NPs have also been used to target TAM and release BLZ945 CSF-1R inhibitor in a breast cancer experimental model
[65]. Therefore, the use of NPs targeting TAM in aggressive tumors (such as glioblastoma or melanoma) may be a promising approach to improve immunological therapies and to reduce the side effects caused by conventional cytotoxic therapies.
4.4. NPs to Block the Macrophage “Do Not Eat Me” Signal
Cancer cells develop diverse mechanisms to escape macrophage phagocytosis, thus promoting an immunosuppressive tumor microenvironment. The overexpression of membrane CD47 (also known as integrin associated protein) is a potent “do not eat me” signal for macrophages and allows cancer cells to evade macrophage phagocytic activity
[67][68]. The crosstalk between the cancer cell CD47 and the macrophage signal-regulatory protein alpha (SIRPα) has been designated as the innate immune checkpoint
[67]. CD47/SIRPα crosstalk has been conceptually chosen to develop NPs that could efficiently block cancer resistance to macrophage-mediated phagocytosis (
Figure 1).
Rao and colleges have constructed genetically engineered cell-membrane (gCM)-coated magnetic NPs overexpressing SIRPα variants with high efficiency for CD47
[69]. This allowed the blockade of CD47/SIRPα crosstalk under magnetic activation in murine models of melanoma and triple negative breast cancer. This therapy prolonged mouse survival, controlled tumor growth, promoted cancer cell cytophagy, boosted T-cell mediated devastation of cancer cells, and improved antigen presentation by macrophages and dendritic cells
[69]. CD47-targeted bismuth selenide (Bi2Se3) NPs have also been employed to deplete TAMs in tumors and to promote macrophage-mediated phagocytosis of cancer cells
[70]. These NPs presented an excellent photothermal performance and efficiently increased the temperature of the tumor. This photothermal therapy reduced tumor size in 4T1 tumor-bearing mice, a breast cancer model. A further modification of Bi2Se3 NPs consisted of polyethylene glycol (PEG) functionalization and an anti-CD47 antibody (Ab) coating to target cancer cells
[70]. Ab-PEG-Bi2Se3 also were useful for IR thermal imaging, which helped to track the location of the agent in cancer cells
[70].
Src homology region 2 phosphatase (SHP2) is a downstream effector responsible for SIRPα function in neurons, dendritic cells, and macrophages
[68]. Some authors have designed self-assembled dual inhibitor-loaded NPs to target “M2-like” TAM
[71]. These NPs consisted of different self-assembled colipids: 1,2-Distearoyl-sn-glycero-3-phosphorylethanolamine (DSPE), PEG, and Phosphatidylcholine (PC). These constructs have been used for the delivery of two key TAM-related pathway inhibitors, SHP2 and CSF-1R inhibitors. NPs with both inhibitors increased macrophage phagocyte ability as compared to free drugs
[71].
4.5. NPs to Switch TAM to an Antitumor “M1-Like” Phenotype
Macrophage activity is molded by cancer cells to help tumor growth. However, macrophages are very plastic cells that may be re-educated by functionalized NPs to switch from “M2-like” TAMs to an M1 pro-inflammatory phenotype to detect and destroy cancer cells (
Figure 1)
[43][72][73].
M1 polarized macrophages help to combat tumor growth via activation of phagocytosis and T-cell mediated immune response. The differential phenotype of TAMs is still being investigated to find therapeutic targets to re-polarize these macrophages to an “M1-like” phenotype
[41][42][43]. For example, VEGF and Placental growth factor (PIGF) are overexpressed in “M2-like” TAM. Song et al. designed NPs with PEG and trimethyl chitosan modified with mannose
[74]. They included two siRNAs to block the expression of VEGF and PIGF in TAMs. Mannose modification allowed NPs to efficiently target M2 macrophages, as its receptor is highly expressed in TAMs. They found lower colocalization of F4/80 and CD206 (mannose receptor) due to the treatment with these NPs, indicating a decrease in the M2 phenotype. In contrast, IL-12 and IFN-γ cytokine levels increased, resulting in a polarization towards the M1 phenotype
[74]. Gold NPs decorated with thiolated-PEG-COOH polymer, M2pep to target TAM, and thiolated anti-VEGF siRNA have also been developed for the induction of long lasting gene therapy and TAM M1 polarization via VEGF silencing
[75].
It has been described that some microRNAs, such as miR-125B, are involved in the repolarization of macrophages from a pro-tumoral M2 phenotype to an antitumor M1 phenotype
[76]. In this context, Parayath and colleagues developed CD44-targeted hyaluronic acid-poly (ethylenimine) (HA-PEI)-based NPs loaded with miR-125b to treat non-small cell lung cancer in mice
[77]. These NPs promoted an increase in the synthesis of iNOS and a decrease in arginase-1 promoting M1 macrophage polarization
[77]. Other authors have used anionic magnetic NPs to transfect the same miR-125b
[78].
Iron oxide NPs have also been employed for TAM re-education as they display intrinsic ability to promote M1 polarization in TAM
[79][80][81][82]. In this context, ferumoxytol NPs, clinically approved iron oxide NPs, have been used to design a targeted therapy to TAM
[79]. A toll-like receptor 3 (TLR3) agonist was attached to the NP surface to target TAM. Ferumoxytol displayed anti-tumor effects inducing a pro-inflammatory response in macrophages. Its combination with TLR3 agonist increased macrophage polarization to a M1 phenotype in a melanoma cancer model
[79]. These NPs efficiently increased TNF-α, iNOS levels, and NO secretion, inducing a pro-inflammatory response in macrophages and the activation of T-cell immunity. The therapy with these NPs induced regression in primary melanoma and in lung metastases
[79].
NF-κB is a family of highly conserved transcription factors and a master regulator of inflammatory responses in macrophages
[83]. In recent years, numerous investigations have arisen using modified NPs to transform TAM into M1 macrophages via induction of NF-κB-related inflammatory pathways. For example, iron oxide NPs loading a P13K inhibitor, 3-methyladenine (3-MA), modified with mannose have been used for functional TAM reeducation
[84]. The transcription factor NF-κB successfully activated TAM to induce M1 polarization
[84]. Iron oxide NPs coated with biomimetic membranes from LPS-treated macrophages have also been designed and tested for TAM re-education in a murine model of breast cancer
[85]. These NPs were decorated with a TLR7 agonist to boost the activation of NF-κB
[85]. Other authors have also suggested the use of TLR7 and TLR8 agonists to induce the activation of NF-κB to enhance immunotherapy efficacy and to increase TNF-α and IL-6 secretion
[86]. For example, Shan et al. used human ferritin heavy chain nanocages modified with M2pep to deliver a TLR agonist in a murine breast cancer model
[87]. TLR agonist triggered TLR signaling and polarized TAM into M1 inflammatory macrophages
[87].
Bruton’s tyrosine kinase (BTK) is a non-receptor kinase belonging to the Tec family of kinases and has been found overexpressed in TAM
[88]. Qiu and colleagues developed NPs with phosphatidylglycerol nanocomplexes conjugated and decorated with stearic acid
[89]. They have encapsulated a small molecule BTK inhibitor, Ibrutinib (IBR), for targeted immunotherapy. They also included sialic acid linked to Sigle-1 cell adhesion molecule (overexpressed in TAM) on the NPs’ surface to target TAM. The inhibition of BTK in TAMs resulted in a reduced release of TH
2 tumorigenic cytokines and impaired angiogenesis. The nanoconstructs efficiently suppressed tumor growth and polarized TAM to a M1 phenotype
[89].
5. Nanocomposite Hydrogels to Modulate Macrophages
Hydrogels are three-dimensional cross-linked polymer networks that contain high amounts of water
[90]. Hydrogels are drug delivery vehicles that can be used as site-specific drug-controlled release systems. The main characteristics of hydrogels are protection, targeting, and local and controlled drug delivery by swelling and shrinkage
[91]. Enzymatic, hydrolytic, or environmental stimuli trigger hydrogel drug release at the implantation site
[91].
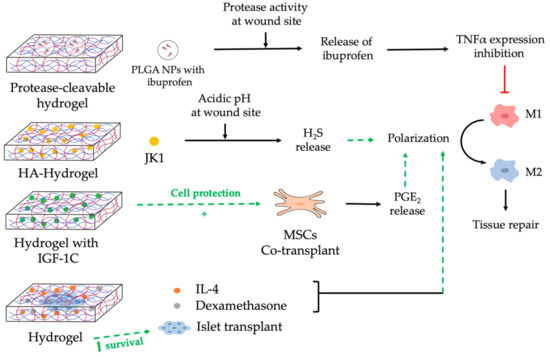
J. Chen and colleagues synthesized a system of double hydrogel layers on titania nanotubes
[92]. They introduced IFN-
γ between the double hydrogel layers and IL-4 was encapsulated into the titania nanotubes. This dual system allowed the initial release of IFN-
γ, which stimulated the switch of macrophages to M1 in three days and the subsequent release of IL-4 at day 4, which displayed anti-inflammatory effects mediated by the macrophage switch to M2 macrophage phenotype
[92]. Hydrogels loading anti-inflammatory drugs have also been designed to modulate macrophages. For example, protease-cleavable hydrogel has been designed to locally deliver PLGA NPs loaded with ibuprofen to inflamed tissues where elevated protease activity is found
[93]. Delivered ibuprofen inhibited TNFα expression in RAW 264.7 murine macrophages (
Figure 2).
Figure 2. Nanocomposite hydrogels to modulate macrophages. This figure illustrates four examples of hydrogels designed to induce macrophages polarization for tissue repair. The first hydrogel encapsulates NPs with anti-inflammatory drugs that are released upon exposure to protease activity. The second hydrogel contains a donor (JK1) that can be triggered in a pH-dependent manner to release pro-regenerative molecules (H2S). The third hydrogel is loaded with molecules (IGF-1C) that promote co-transplanted MSCs survival, which modulates macrophage function. The fourth hydrogel encapsulates transplanted cells (islet transplant) and pro-regenerative molecules (IL-4, Dexamethasone). PLGA: poly (lactic-co-glycolic acid); NPs: nanoparticles; TNFα: tumor necrotic factor alpha; HA: hyaluronan; H2S: hydrogen sulphide; Insulin-like growth factor 1 (IGF-1C); MSC: mesenchymal stem cells; PGE2: prostaglandin E2; IL: Interleukin.
Nanocomposite hydrogels have also been used to modulate macrophages to boost wound repair. M. Xian and collaborators have designed hydrogen sulphide (H
2S) donors called JK1 that release H
2S in a pH-dependent manner
[94]. H
2S is known to have an important role in wound repair due to its anti-inflammatory activity through the conversion of macrophages to a M2 phenotype
[95]. It has been demonstrated that the acidic pH in wounds during the inflammatory stage triggers the release of H
2S from JK-donors
[94]. J. Wu and coworkers designed HA-hydrogels as delivery systems for JK1 and they achieved a pH-dependent prolonged H
2S releasing profile
[96]. HA-JK1 hydrogel promoted the conversion to M2 macrophages that contributed to wound healing in mice (
Figure 2).
Hydrogels have also been used for tissue and organ regeneration. MSCs transplant has been considered a good treatment for several immune disorders because they are known to secrete prostaglandin E
2 (PGE
2), which promotes M2 macrophage polarization and tissue repair
[97]. MSCs have been used in chitosan-based injectable hydrogels combined with entrapped C domain peptide of insulin-like growth factor 1 (IGF-1C) for colitis in mice
[98]. Hydrogels containing growth factors mimic native stem cell microenvironments
[99]. Therefore, these authors provided a supportive niche for transplanted stem cells that facilitated their survival and enhanced their therapeutic effects. Overall, IGF-1C hydrogels protected MSCs, which released high levels of PGE
2 in an experimental mouse model of colitis
[98]. PGE
2 released from MSCs in these hydrogels induced the M2 macrophage phenotype reducing inflammation and promoting colon regeneration (
Figure 2).
NPs in silk hydrogels to modulate macrophages are also a promising treatment for type 1 diabetes. Kumar and colleagues designed hydrogels containing IL-4 and Dexamethasone to encapsulate pancreatic islets
[100]. The structure of these hydrogels allowed the preservation of islet function and viability. The release of IL-4 and Dexamethasone from these hydrogels modulated macrophage phenotype to stimulate tissue regeneration in mice (
Figure 2).