Laser-Induced Breakdown Spectroscopy (LIBS) has been firstly introduced and proposed for analytical applications almost immediately after the invention of the laser in 1960. Since then, it has been proposed and today is widely used as an alternative analytical method for numerous applications. The operating principle of LIBS is quite simple and is based on the interaction of a powerful enough laser beam, focused usually on or in a sample, inducing a dielectric breakdown of the material, thus resulting in plasma formation consisting of excited and non-excited atoms and molecules, fragments of molecular species, electrons and ions, and emitting characteristic radiations, whose spectroscopic analysis can in principle provide the elemental composition fingerprint of the material. The required instrumentation consisting basically of a laser source, and a spectrometer/monochromator equipped with the appropriate light detector (nowadays being almost exclusively some CCD or ICCD type detector) is relatively simple and economically affordable, while significant progresses have been achieved to small size and/or portable equipment, facilitating largely the in situ operation.
1. Overview
Laser-Induced Breakdown Spectroscopy (LIBS), having reached a level of maturity during the last few years, is generally considered as a very powerful and efficient analytical tool, and it has been proposed for a broad range of applications, extending from space exploration down to terrestrial applications, from cultural heritage to food science and security. Over the last decade, there has been a rapidly growing sub-field concerning the application of LIBS for food analysis, safety, and security, which along with the implementation of machine learning and chemometric algorithms opens new perspectives and possibilities.
2. Laser-Induced Breakdown Spectroscopy (LIBS)
Laser-Induced Breakdown Spectroscopy (LIBS) has been firstly introduced and proposed for analytical applications almost immediately after the invention of the laser in 1960
[1]. Since then, it has been proposed and today is widely used as an alternative analytical method for numerous applications
[2]. The operating principle of LIBS is quite simple and is based on the interaction of a powerful enough laser beam, focused usually on or in a sample, inducing a dielectric breakdown of the material, thus resulting in plasma formation consisting of excited and non-excited atoms and molecules, fragments of molecular species, electrons and ions, and emitting characteristic radiations, whose spectroscopic analysis can in principle provide the elemental composition fingerprint of the material. The required instrumentation consisting basically of a laser source, and a spectrometer/monochromator equipped with the appropriate light detector (nowadays being almost exclusively some CCD or ICCD type detector) is relatively simple and economically affordable, while significant progresses have been achieved to small size and/or portable equipment, facilitating largely the in situ operation
[3][4].
The main attributes of LIBS are its capability to provide the simultaneous multi-elemental composition of a sample, of any state of matter (i.e., solid, liquid, or gas, dielectric or conductive), with either little sample preparation or none at all, and due to developments in photonic technology, its instrumentation is of relatively low cost and allows for in situ and on-line applications, as well as applications where the measurement is performed remotely
[3]. LIBS-based instruments, either portable or even handheld are now commercialized and are readily available; they are used mainly for the detection of metallic elements in alloys, paints, rocks, soil, etc.
[5].
For many years, LIBS was considered mainly for applications related with industrial diagnostic purposes
[6]; however, later, it has been applied to environmental, cultural heritage, and space applications
[7][8]. Despite the vast field of applications for which LIBS has been proposed, two important issues arise that are considered as drawbacks: the reproducibility and the relatively large detection limits, at least compared with other competing technologies (as e.g., ICP, atomic emission/absorption, etc.). Both these issues are attributed to the relatively lower reproducibility of the laser-induced plasma that take place during the plasma formation
[9] and very often to matrix effects
[10].
During the past decade, the scientific interest about LIBS-related applications has been importantly revived, mostly due to the implementation of chemometric and machine learning tools for the analysis of the LIBS spectroscopic data
[11][12]. In comparison with other spectroscopic techniques, LIBS is superior in terms of collected data, as it can provide enormous datasets with thousands of variables in very short acquisition times. In that sense, the first LIBS benchmark dataset has been published by Képeš et al.
[13] which contains LIBS spectra from 138 soil samples belonging to 12 classes. Moreover, based on this benchmark dataset, a comparative classification contest has been performed during the EMSLIBS 2019 conference by Vrábel et al.
[14].
Recently, a challenging application of LIBS that received considerable attention is the implementation of LIBS for foodstuff analysis. Specifically, LIBS was employed for the detection of heavy metals in various foodstuffs, for the detection of adulteration and for determining the designation of origin for various types of foodstuffs
[15][16]. In particular, during the last decade, more than 100 scientific articles have been published that cover various aspects of LIBS applications in food related analysis. Despite this renewed interest, the related research efforts performed so far concerning the most commonly adulterated foods (i.e., olive oil, honey, and dairy products) are rather limited, to the best of our knowledge.
3. LIBS principle of operation
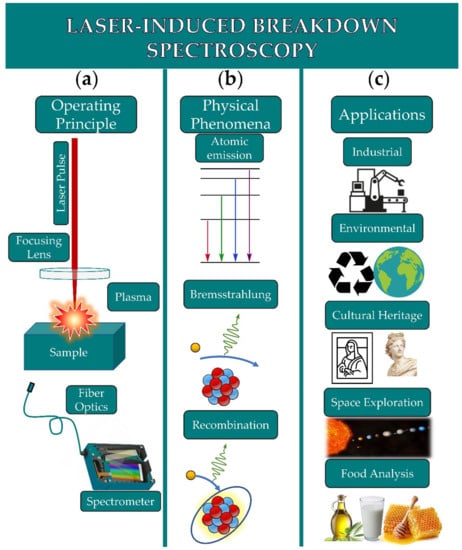
LIBS is based on the spectroscopic analysis of the plasma-emitted radiation resulting from the interaction of a strong enough and focused laser beam with a sample. A typical LIBS setup requires a laser, a lens for the focusing of the laser radiation, a lens system or a fiber optic for the collection of radiation emitted from the plasma, and a spectrometer to analyze the collected plasma emission. A schematic of the setup is shown in Figure 1a.
Figure 1. (a) Schematic of a LIBS setup. (b) Radiative processes occurring in the plasma (atomic emissions, Bremsstrahlung, and recombination). (c) Typical applications of LIBS.
When a strong enough laser beam is focused on the sample’s surface, the material is heated, vaporized, and finally removed; i.e., laser ablation is occurring. The amount of material that is removed depends on the intensity and the wavelength of the laser beam, the laser pulse duration, as well as the material itself (i.e., color, reflectance, surface morphology, etc.). Then, the vaporized ablated material is rapidly expanding above the sample’s surface, forming the so-called high-temperature plasma plume
[17].
In general, the laser-induced plasmas contain various species, namely atoms and molecules (excited or not), ions, electrons, and often small diatomic molecules. The latter are either formed following the fragmentation of the sample’s constituents or can be formed through chemical reactions occurring within the plasma volume
[18]. The radiation emitted from the plasma usually exhibits several spectral features, arising from radiative transitions of the plasma species, and radiative recombinations and bremsstrahlung, presenting the form of discrete or continuous spectra. Discrete spectra of radiation are due to transitions occurring between bound states of atoms, ions, and molecules. Atomic and ionic transitions exhibit spectral lines, while molecular transitions exhibit band-shaped spectral features. These types of transitions are well studied, and their characteristics (e.g., wavelength, energy levels, etc.) can be found in various databases such as the NIST atomic and molecular spectra database
[19][20]. On the other hand, continuous spectra of plasma radiation are due to processes such as Bremsstrahlung and/or to a recombination of electrons with atoms
[3][17][18]. A schematic of the atomic and molecular emissions, Bremsstrahlung, and recombination processes are shown in
Figure 1b.
The laser-produced plasmas are in general short-lived, typically lasting up to a few microseconds. Their temporal evolution and analytical description are governed by rather complex dynamics and are out of the scope of this paper. However, for the sake of completeness, a simplistic description of the temporal evolution of the plasma-emitted radiation will be given. So, within few tens of nanoseconds, after the plasma creation, the emitted radiation is basically due to Bremsstrahlung and recombination of electrons with ions, corresponding to a continuous spectrum. A few hundred nanoseconds after the plasma creation, ionic and atomic spectral lines start to emerge and increase in intensity while the continuum decreases rapidly, thus becoming clearly observable, together with the molecular bands.
The analysis and treatment of LIBS data can be demanding, making the extraction of qualitative and/or quantitative information not a trivial task. For instance, LIBS spectra may be complex, containing a large number of features not allowing for the extraction of valuable information about the sample characteristics, whereas spectra from different samples may exhibit the same spectral features, thus impeding the sample discrimination. To overcome such difficulties and by exploiting the ability of LIBS to create large datasets, machine learning and chemometric techniques can be employed. Machine learning algorithms and chemometrics can perform tasks suitable for categorizing/classifying data into desired and distinct classes, calibration measurements, pattern recognition, and outlier detection. These algorithms have been widely applied on LIBS data, for industrial, environmental, cultural heritage, space exploration, and food analysis applications (see, e.g., Figure 1c).
4. Conclusions
LIBS, being a simple, versatile, and powerful technique, has gained significant popularity during the last decades, and it has been proposed for a plethora of applications in different scientific fields. It is only during the last few years, that LIBS applications for food analysis-related applications have emerged. However, this advancement would have remained limited unless modern machine learning and chemometrics methods are implemented, as the complexity of organic matter and the similarities of the respective LIBS spectra do not allow for safe conclusions. So, LIBS assisted by modern data analysis tools, such as machine learning and chemometrics started to become a valuable method for food control and quality assurance.