1000/1000
Hot
Most Recent

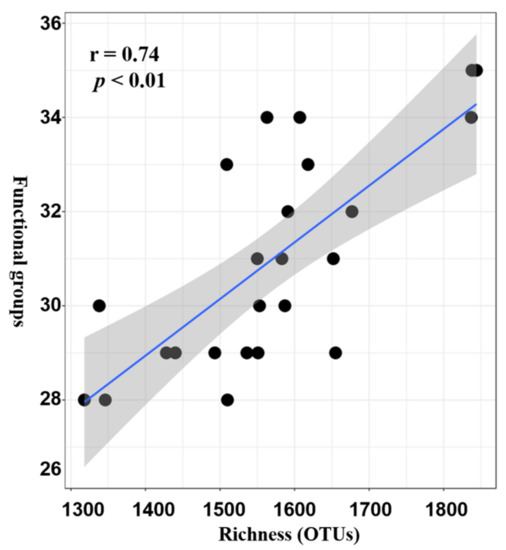
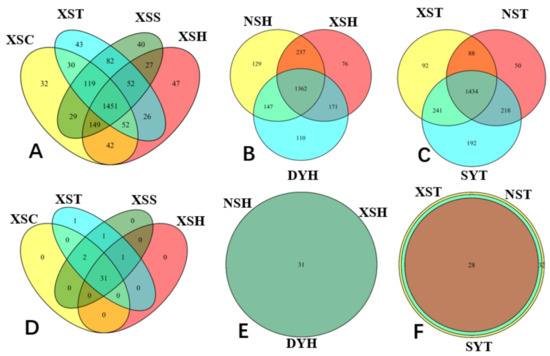
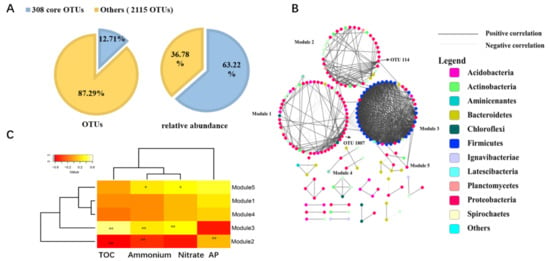
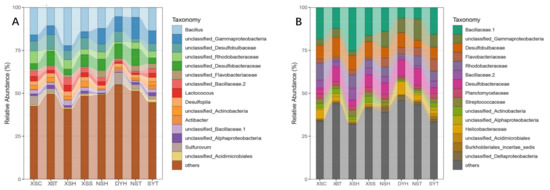
Seagrasses are an important part of the coral reef ecosystem, and their rhizosphere microbes are of great ecological importance. However, variations in diversity, composition, and potential functions of bacterial communities in the seagrass rhizosphere of coral reef ecosystems remain unclear. This study employed the high-throughput sequencing based on 16S rDNA gene sequences and functional annotation of prokaryotic taxa (FAPROTAX) analysis to investigate these variations based on seagrass species and sampling locations, respectively. Results demonstrated that the seagrass rhizosphere microbial community was mainly dominated by phylum Proteobacteria (33.47%), Bacteroidetes (23.33%), and Planctomycetes (12.47%), while functional groups were mainly composed of sulfate respiration (14.09%), respiration of sulfur compounds (14.24%), aerobic chemoheterotrophy (20.87%), and chemoheterotrophy (26.85%). Significant differences were evident in alpha diversity, taxonomical composition and putative functional groups based on seagrass species and sampling locations. Moreover, the core microbial community of all investigated samples was identified, accounting for 63.22% of all obtained sequences. Network analysis indicated that most microbes had a positive correlation (82.41%), and two module hubs (phylum Proteobacteria) were investigated. Furthermore, a significant positive correlation was found between the OTUs numbers obtained and the functional groups assigned for seagrass rhizosphere microbial communities (p < 0.01). Our result would facilitate future investigation of the function of seagrass rhizosphere microbes.
| Sample | DYH | SYT | XST | XSH | XSC | XSS | NXH | NXT | |
|---|---|---|---|---|---|---|---|---|---|
| Latitude (° E) | 22.54 | 18.2 | 16.84 | 16.84 | 16.84 | 16.84 | 9.71 | 10.22 | |
| Longtitude (° N) | 114.45 | 119.47 | 112.34 | 112.34 | 112.34 | 112.34 | 114.29 | 114.2 | |
| Water | pH | 8.14 (0) |
8.15 (0.01) |
8.24 (0.01) |
8.24 (0.03) |
8.24 (0.03) |
8.24 (0.01) |
8.15 (0.01) |
8.21 (0.01) |
| Salinity | 28.37 (0.07) |
24.7 (0.3) |
34.52 (0.05) |
34.52 (0.15) |
34.57 (0.08) |
34.52 (0.05) |
34.31 (0) |
34.54 (0.09) | |
| DO (mg/L) | 5.82 (0.05) |
6.7 (0.2) |
9.47 (0.17) |
9.47 (0.51) |
9.64 (0.59) |
9.47 (0.17) |
7.64 (0.03) |
8.36 (0.13) | |
| Nitrate (mg/L) | 0.153 (0.004) | 0.044 (0.002) | 0.049 (0.002) | 0.049 (0.005) | 0.046 (0.001) | 0.049 (0.002) | 0.06 (0.001) |
0.035 (0) |
|
| Nitrite (mg/L) | 0.001 (0) |
0.025 (0.001) | 0.016 (0) |
0.016 (0.001) | 0.016 (0.001) | 0.016 (0) |
0.001 (0) |
0.001 (0) |
|
| Ammonium (mg/L) | 0.069 (0) |
0.121 (0.002) | 0.094 (0.002) | 0.094 (0.005) | 0.091 (0.006) | 0.094 (0.002) | 0.048 (0.001) | 0.032 (0.001) | |
| Phosphate (mg/L) | 0.007 (0.001) | 0.015 (0) |
0.01 (0.001) |
0.01 (0.002) |
0.01 (0.001) |
0.01 (0.001) |
0.006 (0) |
0.006 (0) |
|
| Sediment | Ammonium (mg/kg) | 3.02 (0.01) |
5.97 (0.02) |
4.37 (0.01) |
2.91 (0.03) |
1.45 (0.01) |
4.78 (0.02) |
6.39 (0.03) |
5.48 (0.12) |
| Nitrate (mg/kg) | 22.3 (2.63) |
24.4 (3.68) |
44.0 (2.10) |
62.4 (3.26) |
69.4 (4.06) |
44.0 (2.24) |
32.60 (1.12) |
45.60 (1.56) | |
| AP (mg/kg) | 18.0 (2.01) |
15.0 (1.28) |
14.0 (3.06) |
11.0 (1.85) |
15.0 (2.30) |
15.0 (1.60) |
18 (2.45) |
17 (3.01) |
|
| TOC (%) | 18.4% (1.89) | 32.5% (2.36) |
43.4% (1.03) |
33.2% (2.64) |
40.3% (3.12) | 21.8% (3.56) |
36.80 (2.63) |
42.06 (3.42) |
| Microbial Communities |
Microbial Communities |
Phylogenetic Composition (PD) |
Taxonomic Alpha Diversity |
|||
|---|---|---|---|---|---|---|
| Richness | Shannon | Simpson | ||||
| Species | p | p | p | p | ||
| XSC | XST | 0.9420 | 0.6700 | 0.5250 | 0.3240 | |
| XSC | XSH | 0.4550 | 0.3870 | 0.7380 | 0.7380 | |
| XSC | XSS | 0.5250 | 0.6700 | 0.3240 | 0.5250 | |
| XST | XSH | 0.0802 | 0.9690 | 0.0810 | 0.0330 | |
| XST | XSS | 0.2180 | 0.1060 | 0.9870 | 0.9870 | |
| XSS | XSH | 0.0240 | 0.0330 | 0.0330 | 0.0810 | |
| Location | ||||||
| H. ovalis | XSH | DYH | 0.9500 | 0.3700 | 0.0200 | 0.0200 |
| XSH | CGX | 0.2300 | 0.3700 | 0.3700 | 0.3700 | |
| DYH | CGX | 0.1300 | 0.0200 | 0.3700 | 0.3700 | |
| T. hemprichii | ||||||
| XST | SYT | 0.5490 | 0.5500 | 0.3700 | 0.8960 | |
| XST | NXT | 0.5490 | 0.3000 | 0.0370 | 0.0650 | |
| SYT | NXT | 0.0930 | 0.0300 | 0.0200 | 0.1730 | |
| Microbial Community |
Microbial Community |
Delta Unifrac |
P Unifrac |
Delta Bray |
P Bray |
Delta Euclidean |
P Euclidean |
Delta Sorensen |
P Sorensen |
|
|---|---|---|---|---|---|---|---|---|---|---|
| Species | ||||||||||
| XSC | XST | 0.184 | 0.087 | 0.294 | 0.016 | 1.260 | 0.156 | 0.099 | 0.293 | |
| XSC | XSH | 0.177 | 0.084 | 0.308 | 0.109 | 1.590 | 0.294 | 0.152 | 0.282 | |
| XSC | XSS | 0.049 | 0.100 | 0.088 | 0.100 | 1.040 | 0.500 | 0.061 | 0.300 | |
| XST | XSH | 0.219 | 0.025 * | 0.366 | 0.015 * | 1.429 | 0.017 * | 0.136 | 0.124 | |
| XST | XSS | 0.176 | 0.035 * | 0.284 | 0.017 * | 1.073 | 0.009 ** | 0.083 | 0.016 * | |
| XSH | XSS | 0.169 | 0.011 * | 0.299 | 0.012 * | 1.403 | 0.037 * | 0.135 | 0.109 | |
| Location | ||||||||||
| H. ovalis | XSH | NSH | 0.039 | 0.100 | 0.088 | 0.100 | 1.024 | 0.100 | 0.111 | 0.100 |
| XSH | DYH | 0.035 | 0.100 | 0.125 | 0.100 | 1.024 | 0.100 | 0.114 | 0.100 | |
| NSH | DYH | 0.033 | 0.100 | 0.122 | 0.100 | 0.667 | 0.100 | 0.042 | 0.100 | |
| T. hemprichii | XST | NST | 0.070 | 0.100 | 0.110 | 0.100 | 20.104 | 0.100 | 0.130 | 0.100 |
| XST | SYT | 0.064 | 0.100 | 0.112 | 0.100 | 19.395 | 0.100 | 0.132 | 0.100 | |
| NST | SYT | 0.026 | 0.100 | 0.105 | 0.100 | 18.762 | 0.100 | 0.121 | 0.100 |