Most of the published studies on COVID-19 patients with complications of secondary infections were retrospective and reported single-centered data on a small number of patients, but the incidence and the time of development of the secondary infections were almost homogeneous. Karulli A. et al. reported 16 out of 32 patients developing an MDR infection with the prevalent isolates of CPE, and the highest mortality rate of 72%. Although the sample size was small, some considerations might be made about the high number of patients on empirical antibiotic therapy at admission (78%) and the high percentage of patients that were on steroids (53%)
[11]. Similarly, Patel A. et al. reported a spread of MDR secondary infections caused by
Gram negative bacteria; the whole sample of patients described had an MDR infection and the majority of infections reported were caused by CPE (46%). Moreover, nearly the whole studied population was on an empiric antibiotic therapy upon admission (97%) compared to the study by Karulli, and 35% of them received steroids
[21]. Baiou A. et al. reported 33% of MDR secondary infections with around 10% of the isolated microorganisms as carbapenem-resistant
Klebsiella pneumoniae and
Pseudomonas aeruginosa. In the latter, differently from the two previous studies, patients were younger and few of them received steroids (4.7%). The only risk factor independently associated with MDR secondary infections was the invasive mechanical ventilation (OR 1.062; 95% CI 1.012–1.114)
[22]. A reduction of antibiotic prescriptions has been described between January 2020 and April 2020 (OR 0.28, 95% CI 0.08–0.98) and a lower prescription was also observed in studies evaluating children (38.5%, 95% CI 26.3%–52.3%) when compared to studies including only adult population (83.4%, 95% C.I. 76.6–88.5%), while a higher antibiotic administration was described with the increasing of median or mean patient age, per 10 years increase (OR 1.45, 95 CI 1.18–1.77) and in patients requiring invasive mechanical ventilation (OR 1.33, 95% CI 1.15–1.54), with a 10% increase
[23]. Cultrera R. et al. reported the microbiological characteristics of a high number of isolates in secondary infections on a small sample of patients: the incidence of MDR-acquired infections was similar to other retrospective studies. They found a higher frequency of methicillin-resistant
Staphylococcus Aureus (MRSA), vancomycin-resistant
Enterococcus faecium and carbapenem resistant
Acinetobacter baumannii [12]. Bogossian E.G. et al. compared the incidence of multi-drug resistant organisms (MDRO) between patients admitted with COVID-19 and a control group of patients with subarachnoid hemorrhage (SAH), complicated by sepsis, admitted in a period before the outbreak. The incidence and characteristics of MDR secondary infections reported were similar to most of the data coming from the published articles on COVID-19 and did not show any significant difference in the multivariable competing risk analysis (sHR 1.71 (CI95% 0.93–3.21)) between the two groups; therefore, they concluded that COVID-19 was not an independent risk factor associated with MDR infections acquisition
[13]. Nasir N. et al. reported that patients who were severe to critically ill at the time of admission with COVID-19 had an over fourfold probability to develop secondary infections (OR 4.42; 95% CI 1.63–11.9). In fact, the majority of those patients needed more invasive devices such as an endotracheal tube and central venous catheter; undergoing more frequent to invasive mechanical ventilation and, as a consequence, they were more frequently admitted to the ICU compared to the controls. Moreover, the authors showed that all patients developing secondary infections received the empiric antibiotic therapy at admission and an elevated percentage (92%) were on steroids treatment, which was an independent risk factor strongly associated with secondary infections (OR 4.6; 95% CI 1.24–17.05)
[24]. Giacobbe D.R. et al. and Bonazzetti C. et al. both reported data on the incidence of blood stream infections (BSI) in critically ill COVID-19 patients
[14][25]. The patients described in both papers were similar in the number of cases and age, but Bonazzetti C. et al. reported a higher number of MDR infections, caused especially by
Enterococcus spp., with a higher rate of mortality (49% vs. 26%). Moreover, they observed that patients who developed BSI during their ICU stay had a higher sequential organ failure assessment (SOFA) score (9.5, IQR 8–12 vs. 8, IQR 5–10), but they did not report whether their patients were on an empirical antimicrobial therapy at ICU admission and if they received steroids
[25]. Instead, Giacobbe D.R. et al. reported that 96% of their patient sample were on an empiric antimicrobial treatment at ICU admission and 31% received steroids: they found that the anti-inflammatory treatment was an independent risk factor for BSI development (caused-specific hazard ratio [csHR] 3.95; 95% CI 1.20–13.03 for methylprednisolone and csHR 10.69; 95% CI 2.71–42.17, for methylprednisolone plus tocilizumab)
[14]. Interestingly, Luxemburger H. et al. reported that the use of proton pump inhibitors (PPI) (OR 2.37; 95% CI 0.08–5.22) and gastroesophageal reflux disease (OR 6.40; 95% CI 1.50–35.51) were independent predictive factors of developing secondary infections in patients affected by COVID-19. These results underlined the role of microaspiration in the pathogenesis of secondary bacterial infections of the lower respiratory tract
[26]. Furthermore, a short term and current use of PPI during admission for COVID-19 increased the worst-outcome risk (OR 1.90; 95% CI 1.46–2.77 and OR 1.79; 95% CI 1.30–3.10) due to an increased probability of ICU admission, invasive mechanical ventilation, and death
[27]. A recent meta-analysis confirmed such findings on a bigger population coming from seven papers (OR 1.46, 95% CI 1.34–1.60)
[28]. Ripa et al. reported data on both BSI and low respiratory tract infections (pLRTI) among 86/731 (11.8%) admitted to the ICU
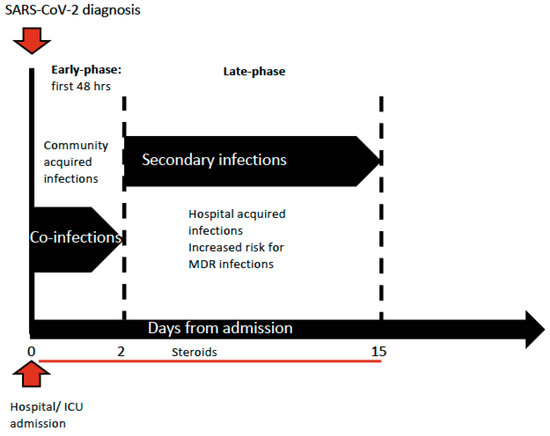
[29]. Critically ill patients more frequently developed a secondary infection. During the follow-up of 1,318 patient-days (PDFUs), 40/731 patients (5.5%) had 51 secondary infections during ICU stay with an incidence rate of 38.7 (28.8–50.9) per 1000 PDFUs, which was considerably higher when compared to patients outside the ICU, with an incidence rate of 4.0 (2.9–5.5) per 1000 PDFUs. Among factors associated with an increased risk for secondary infections, there was ICU admission within the first 48 h from hospital admission (HR 2.51, 95% CI 1.04–6.05), together with decreasing PaO
2/FiO
2 ratio < 100 (HR 3.67; 95% CI 1.24–10.90). The results confirmed the median time for the first secondary BSI at 13 days from hospital admission with a prevalence of Gram-positive among the isolates (
Staphylococcus epidermidis and
Enterococcus faecium), while at 16 days for the first pLRTI, mainly caused by Gram-negative MDRO (carbapenem-resistant
Acinetobacter baumannii, CP-Kp, CPE and carbapenem-resistant
Pseudomonas aeruginosa). The authors observed an increase of antimicrobial resistance compared with an historical cohort of patients before the SARS-CoV-2 outbreak, with a higher incidence rate of Gram negative MDRO among BSIs, mainly due to
Acinetobacter baumannii, while among Gram-positive isolates there was an increase of vancomycin-resistant
Enterococcus faecium.
Only two multicenter retrospective studies reported the incidence and risk factors for secondary infections in critically ill patients
[8][30]. The former, published by Grasselli G. et al. evaluated patients with severe COVID-19 admitted to eight Italian hub hospitals
[8]. They reported data on a sample of 774 patients, in which 35% developed MDR infections, among 359 patients (46%) that developed hospital acquired infections (HAI). The most frequent secondary infections were ventilator associated pneumonia (VAP, N = 389 [50%]), with 26 (95% CI 23.6–28.8) per 1000 patient-days of invasive mechanical ventilation, and BSI (N = 183 [34%]), with 11.7 (95% CI 10.1–13.5) per 1000 ICU patient-days. They observed an incidence of MDR infections like most of the single center, and in the majority of the cases the isolated microorganisms were
Enterobacterales with resistance to carbapenems and
MRSA, especially in VAP. The number of patients that were on empirical antibiotic therapy was similar to the other single-center studies (69%), but nearly half of these patients received broad spectrum drugs. Notably, at the multivariable analysis they showed that treatment with broad spectrum antibiotics was independently associated with HAI (HR 0.61; 95% CI, 0.44–0.84), which paradoxically described a protective role of antimicrobial treatment against the development of secondary infections; however, they reported that the policy of all participating ICUs was to interrupt empiric antimicrobial therapy early if culture results were negative for bacterial infections at admission. In addition, the authors observed that patients complicated with septic shock resulted in a doubled mortality rate (52%). The other multicenter study, published by Giacobbe D.R. et al. involved nine Italian hub hospitals that admitted 586 patients with severe COVID-19 during the first wave of the outbreak between February and May 2020. Among them, 171 (29%) patients developed an infection with diagnosis of VAP. The incidence rate of VAP was slightly lower when compared to the study by Grasselli et al. (18 vs. 26 events per 1000 ventilator days). However, the incidence of MDR infections was similar and very close to all the other studies that reported this data. The most frequently isolated germs were
Pseudomonas aeruginosa (35%),
Staphylococcus aureus (23%) and
Klebsiella pneumoniae (19%). Among the MDR microorganisms, the most frequently isolated species were methicillin-resistant
S. aureus (10%) and carbapenem-resistant Gram-negative bacteria (32%). They expressed the outcome of their population at 30 days as a 30-day case-fatality and it was quite high (78/171, 46%), but it was evaluated only on patients who developed a secondary infection. Other studies reported the mortality rate over the whole COVID-19 population; therefore, no conclusion could be drawn comparing the outcome of the results among the selected studies. Interestingly, the authors found that septic shock and ARDS at VAP onset were strongly associated with an increase of 30-day case-fatality (OR 3.30; 95% CI 1.43–7.61 and OR 13.21; 95% CI 3.05–57.26 respectively). This association was confirmed when they analyzed the subgroup of patients who developed VAP with the BALF culture positive. The incidence rate of VAP reported in patients for both studies was surprisingly high, especially when compared to that related to non-COVID-19 critically ill patients (one out of 19 episodes per 1000 ventilator days)
[31][32]. The increase of VAP incidence rate in COVID-19 critically ill patients might be explained by multiple triggering mechanisms: (i) one of the main hypothesis might be that the viral infection causes a direct damage of the lower airway epithelial cells associated with impaired mucociliary clearance, which allows bacterial binding to cell surfaces
[33]; (ii) a second hypothesis might be an up-regulation of the airway cell receptors
[33]; (iii) a third reason might be a decreased immune response or the abnormal release of inflammatory mediators during the early stages
[34][35]. Furthermore, some authors speculated on the hypothesis that there might have been an overestimation during the COVID-19 period because in the majority of cases the diagnosis was clinical and performing a BALF was rare, especially at the beginning of the outbreak. This was due to the high risk of viral transmission from the patient to the operator despite the presence of PPE, as in several hospitals in Northern Italy where a shortage was reported during the first wave.