Porphyrias are a group of diseases that are clinically and genetically heterogeneous and originate mostly from inherited dysfunctions of specific enzymes involved in heme biosynthesis.
1. Introduction
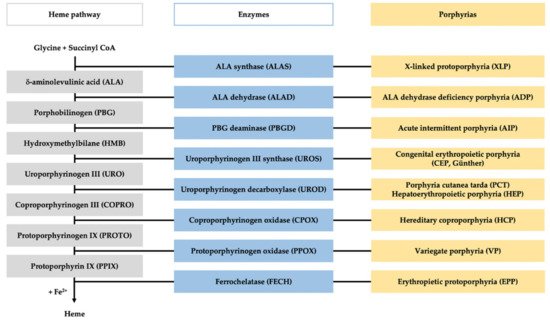
Porphyrias comprise a group of eight metabolic disorders originating from a genetically caused catalytic dysfunction of the enzymes involved in the heme biosynthesis pathway (
Figure 1)
[1]. Dominant or recessive inherited mutations in any of the genes encoding these enzymes lead to disturbance in heme synthesis along with the pathological accumulation and measurable excretion of the intermediates of the heme biosynthesis pathway
[2].
Figure 1. Heme pathway and porphyria. The figure represents the scheme of the heme biosynthesis (in gray), with the involved enzyme (in blue), and the specific porphyria related to each enzyme deficiency (in yellow).
An accumulation of the following two different kinds of metabolites may occur: one is the porphyrin precursors, such as 5-aminolevulinic acid (ALA) and porphobilinogen (PBG), which are linear non-fluorescent molecules, and the other kind is the porphyrins, such as uroporphyrins (URO), coproporphyrins (COPRO), and protoporphyrins (PROTO), which are circular molecules that emit fluorescence signals when excited. The accumulation of these metabolites occurs in different biological samples depending on their chemical properties. Since the hydrophobicity gradient increases as the heme synthesis progresses, the most hydrophilic metabolites (ALA, PBG, URO, COPRO) occur mainly in urine, while the relatively hydrophobic ones (COPRO, PROTO) occur in feces
[3].
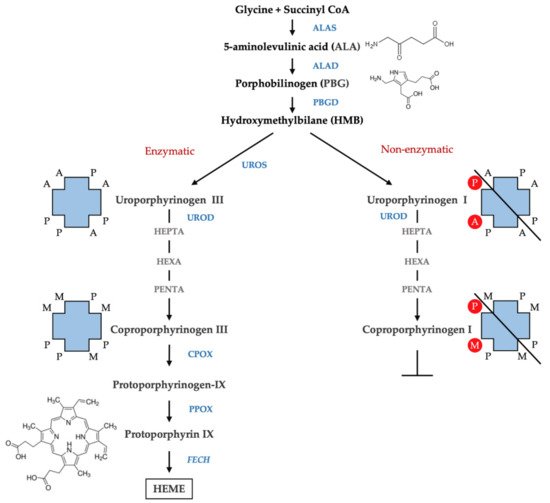
Porphyrins are the oxidized products of porphyrinogens, which are the actual substrates of the enzymes involved in heme biosynthesis. Porphyrins exist in different isomers depending on the arrangement of the substituents acetate (A), propionate (P), methyl (M), and vinyl (V) of the four pyrroles of the porphyrin ring. The most common isoforms are isoform III with asymmetrically arranged substituents and isoform I with symmetrically arranged substituents. The biosynthesis of heme involves the isoforms III since hydroxymethylbilane (HMB) is transformed, by the action of uroporphyrinogen III synthase (UROS), in uroporphyrinogen III. This undergoes subsequent decarboxylation by uroporphyrinogen decarboxylase (UROD) to form hepta-, hexa-, and penta-carboxyl porphyrinogen III, and finally, coproporphyrinogen III. However, in physiological conditions, a fraction of HMB escapes from the catalytic action of the UROS, and results in a non-enzymatic conversion of HMB to the uroporphyrinogen I isomer. Uroporphyrinogen I may subsequently undergo decarboxylation by the action of UROD to form hepta-, hexa-, and penta-carboxyl porphyrinogen I, and finally, coproporphyrinogen I; however, the reaction cannot proceed beyond this step to form heme as the next enzyme in the pathway—coproporphyrinogen oxidase (CPOX)—is stereospecific for the isomer III (
Figure 2). Therefore, the isomers I accumulate in the tissue and are excreted as porphyrins I. A large presence of porphyrins I as well as an abnormal isomer I/isomer III ratio in biological fluids are relevant in defining the presence of porphyria
[4].
Figure 2. Isomers of porphyrins. The figure represents the scheme of the heme biosynthesis with enzymatic (left) and non-enzymatic conversion of HMB (right). The arrangement of the substituents of the four pyrroles of the porphyrin ring is shown. A, acetate (-CH2COOH); P, propionate (-CH2CH2COOH); M, methyl (-CH3) and vinyl (CH=CH2).
Physiologically, heme synthesis is accomplished by the sequential action of eight enzymes and the whole process is finely regulated. The intermediate substrates other than the final product heme might exert a regulatory effect on the enzymes involved in the pathway
[5]. Moreover, the catalytic capacity of the enzymes in different segments of the pathway varies strongly, leading to variations in substrate pressure. Therefore, the dysfunction of a specific enzymatic activity may cause an accumulation of the substrate immediately before the blockage and of other previous metabolites of the same proceeding line
[6].
Specific combinations of metabolites (patterns) are associated with specific clinical features; sufficient evidence suggests that porphyrin precursors cause symptoms by injuring neurons, while porphyrins cause symptoms by injuring the skin and the liver
[7]. Usually, the levels of porphyrin precursors increase during the presentation of acute neurovisceral attacks induced by exogenous factors, such as medications, nutritional status, stress, exogenous hormones, infection, etc. Moreover, depending on the patient, these levels may return to the normal ranges or remain persistently elevated during the clinically asymptomatic phase
[8]. On the contrary, the accumulation of porphyrins is typically chronic, causing persistent cutaneous photosensitivity or skin fragility as a consequence of prolonged exposure to the sun
[9].
2. Qualitative Screening Tests
2.1. Plasma Scan
Plasma scan is a simple, rapid, and inexpensive screening test that allows for the detection of the presence of increased amounts of URO, COPRO, and PROTO in the plasma, based on the capacity of these photoactive molecules to fluoresce when irradiated with wavelengths close to 400 nm. Once the plasma samples are obtained, the assay may be performed and completed within a few minutes. Briefly, 100 µL of plasma, diluted five-fold in a 0.25 M potassium phosphate-buffered saline (PBS) at pH 6.7 is scanned inside a classical fluorescence spectrophotometer at the excitation wavelength of 405 nm
[10]. Otherwise, 1 mL of freshly drawn plasma may be centrifuged at 14000 rpm for 1 min and then 2 µL of the supernatant may be used directly for analysis in NanoDrop 3100, using a UV LED for excitation (maximum at 365 nm)
[11]. In both methods of conducting the test, the fluorescence emission spectrum is recorded in the wavelength range of 580 to 700 nm.
The fluorimetric emission scanning of plasma is a qualitative test, in which the presence or absence of porphyrins is detected as a positive or negative result, respectively. If positive, the analyst then identifies the wavelength at which the maximum emission peak is obtained, as this wavelength is typical of specific porphyria conditions. The emission maximum obtained within 626–628 nm is specific to variegate porphyria (VP)
[12][13], while a peak within 634–636 nm is characteristic of erythropoietic protoporphyria (EPP) and X-linked protoporphyria (XLP)
[10]. A fluorescence emission maximum within 618–622 nm corresponds to ALA dehydrase (ALAD) deficiency porphyria (ADP), acute intermittent porphyria (AIP), hereditary coproporphyria (HCP), congenital erythropoietic porphyria (CEP, Gunther disease), porphyria cutanea tarda (PCT), and hepatoerythropoietic porphyria (HEP) (
Table 1)
[14].
Table 1. Maximum fluorimetric emission peak in different porphyrias.
| Porphyrias |
ADP/AIP/HCP |
VP |
PCT/HEP/CEP |
EPP/XLP |
| Plasma peak (nm) |
618–622 |
626–628 |
618–620 |
632–636 |
The intensity of the fluorescence peak varies with the activity status of the disease and is always positive in all symptomatic patients, including those with a suspected attack of acute porphyria in progress (ADP, AIP, VP, HCP)
[15] and those with chronic high excretion of porphyrins (CEP, PCT, HEP, EPP). However, only 50% of asymptomatic VP carriers are detected using this method, which renders the plasma scan test inefficient for family studies involving the detection of gene carriers in both adults and children
[16][17]. Moreover, porphyrins in the plasma sample of patients with EPP and VP are particularly labile if exposed to light. Therefore, without adequate light protection, the levels of measurable porphyrin decline rapidly, resulting in false negatives
[18]. In addition, PCT patients treated with phlebotomy and patients with acute porphyria in the latent phase may not present any peak due to normal porphyrin concentration in their plasma samples. The absence of any peak in the emission spectrum, in a well-preserved sample, would imply that the diagnosis of active porphyria may be rejected.
On the contrary, patients undergoing maintenance hemodialysis therapy
[19][20] or those co-infected with immunodeficiency virus (HIV) and hepatitis C virus (HCV)
[21] may develop a secondary elevation of plasma porphyrin along with cutaneous lesions, similar to those observed in genetic PCT patients, and as a consequence, a characteristic fluorescence peak appears at 618–620 nm. Therefore, this test might be useful for monitoring the activity of all the conditions referred to as pseudoporphyria, which do not result from an enzymatic absence of genetic origin.
In conclusion, routine plasma fluorometric screening could be performed as the first diagnostic approach in cases with high levels of porphyrins in the plasma, such as in those with acute attack and the ones with chronic cutaneous porphyria. However, considering that several porphyria conditions have the same characteristic emission peak, the positive results should be confirmed with specific quantitative analysis of porphyrins to establish the correct diagnosis.
2.2. Fluorocytes
The excess of porphyrins in erythrocytes may be detected by the presence of fluorescent red cells (fluorocytes), which may be identified using three qualitative screening tests. The first one is to record, similar to the approach in the plasma scan test, the fluorescence emission spectrum of the erythrocytes diluted in buffered 0.9% saline at a ratio of 1:2000, in the wavelength range of 550–650 nm using 400–415 nm as the excitation wavelength range throughout, in a fluorescence spectrophotometer. The emission peaks for URO, zinc–protoporphyrin (ZnPP), and metal-free protoporphyrin (PPIX) are obtained at 620, 587, and 630 nm, respectively
[18][19][20][21][22].
Otherwise, a drop of freshly obtained blood could be smeared on a glass slide and viewed using a fluorescence microscope under ultraviolet light (380–450 nm)
[23]. Although an automated imaging system, which improved the traditional fresh blood film method, was described to facilitate population screening in EPP, this method requires special equipment and expertise and, therefore, is limited to specialized centers
[24]. Moreover, fluorescence microscopy presents problems associated with quantification, and the radiation focused on individual erythrocytes rapidly photodegrades protoporphyrin, thereby causing evanescence of the fluorescence
[25].
On the contrary, cytofluorimetric analysis is a rapid and sophisticated method for the measurement of the percentage of fluorocytes in the samples. In this method, a sample containing 10 µL of ethylenediaminetetraacetic acid (EDTA)-whole blood diluted in 10 µL of phosphate-buffered solution at pH 7.2 is processed directly in a flow cytometer without staining. Fluorescent erythrocytes emit red fluorescence at λ > 620 nm when excited at 488 nm using an argon laser. The results are expressed as the percentage of cells beyond the threshold of the autofluorescence of the control subjects
[26]. In contrast to fluorescence microscopy, this method does not present the problem of subjectiveness in the evaluation of the intensity of fluorescence in a possibly small fraction of rapidly fading fluorocytes.
Although high levels of fluorocytes may be detected in various forms of porphyria using this method, the test should be predominantly used in EPP
[27], in which the symptoms are not as severe and evident as in CEP and HEP. However, the test only quantifies the percentage of fluorescent erythrocytes and not the actual amount of total protoporphyrins. Even though the results of cytofluorometry are reported to correlate well with those of the quantitative HPLC test
[28], the absolute quantification of porphyrins is necessary to establish the diagnosis of EPP. Nonetheless, once the diagnosis is established, cytofluorometry might prove to be useful in the screening and follow-up of patients.
2.3. Hoesch Test
Rapid tests for detecting the presence of increased PBG in the urine are of great significance in the early diagnosis of an acute attack of porphyria
[29]. Initially, the test used in this setting was the one developed by Watson and Schwartz in 1941, which was subsequently modified in 1964
[30][31]. Although this test was termed as a screening test, it is actually a multistep procedure, and the number of reagents required, the time consumed for performing the test, and the expertise required for appropriately conducting and interpreting the test results have limited its use in clinical wards. Therefore, in current times, this test has been replaced by a simpler and rapid test, which is less prone to misinterpretation by inexperienced personnel, known as the Hoesch test
[32].
In the Hoesch test, two drops of fresh urine are added to 1 mL of Ehrlich’s reagent (solution of 0.2 g of p-dimethylaminobenzaldehyde (DMAB) in 10 mL of 6M HCl). In the case of increased PBG in the urine sample, as the urine contacts the reagent, a cherry-red color develops immediately at the surface of the solution and then throughout the tube under agitation
[33]. The sensitivity of the test is typically 10 mg/L, while the upper normal limit for urinary PBG is approximately 2.5 mg/L. Therefore, small increases in PBG might be missed when using this test, although these are not of practical importance when the test is being performed for symptomatic patients suspected of having an acute attack, as these patients present markedly increased levels of urinary PBG in the range of 10 to 20 times higher than the upper normal value
[34].
Moreover, the Hoesch test procedure appears to have higher specificity compared to the Watson–Schwartz test, as the former does not involve an interfering reaction in the presence of urobilinogen in patients with liver disease
[35]. The other possible sources of false-positive Ehrlich reactions have not been investigated, and the potential inhibitors of this reaction have also not been evaluated thus far
[36][37]. Using the Hoesch test, it is possible to detect the presence of high levels of PBG in the urine accurately and rapidly
[38]. Moreover, it is reported that Ehrlich’s reagent may be stored in a clear glass container for a minimum of nine months without a loss of activity
[35]. These data confirm the efficacy and feasibility of this first-line qualitative test in detecting an acute attack in progress. Nonetheless, further investigative procedures to confirm the diagnosis and identify the type of porphyria should be performed.
3. Quantitative Confirmatory Tests
3.1. ALA and PBG Determination
The quantification of ALA and PBG forms the first line of laboratory testing for acute porphyria in the event of potentially life-threatening acute neurovisceral attacks
[39]. For decades, the collection of urine samples over a period of 24 h was the rule leading to a harmful delay of the necessary therapeutic measures. Meanwhile, this procedure has been replaced by the spot urine sample procedure that is considered to be sufficient to estimate the activity of acute porphyrias and allow for a decision on therapeutic intervention. To date, in specialized porphyria diagnostic laboratories, ALA and PBG are commonly quantified after purification, from a spot urine sample, using two commercially available anion-exchange and cation-exchange columns (ClinEasy
® Complete Kit for ALA/PBG in Urine, Recipe GmbH, Munich, Germany; ALA/PBG by Column Test, Bio-Rad Laboratories, Hercules, CA, USA). This approach enables the selective purification of ALA and PBG, which removes the sample matrix, thereby preventing possible interferences from other compounds
[40]. In several certified European laboratories (EPNET), the preferred choice is the ClinEasy
® Complete Kit for ALA/PBG in Urine of Recipe for the quantification of both ALA and PBG in the urine. Briefly, urine samples are passed through two overlapping columns—the top column, containing an anion exchange resin, adsorbs PBG, and the ALA passes through this top column and is subsequently retained by the cation exchange column at the bottom. The adsorbed PBG is eluted from the top column using acetic acid and mixed with Ehrlich’s reagent, upon which the solution develops a purple color, whose intensity is proportional to the amount of the metabolite in the solution. The retained ALA is eluted using sodium acetate, and after derivatization with acetyl-acetone at 100 °C, it is converted into a monopyrrole, which is the measurable form capable of reacting with Ehrlich’s reagent. The absorbance of both PBG and ALA is measured against the blank reagent at 533 nm using a UV–VIS spectrophotometer. Metabolite concentrations are calculated through comparison to the appropriate calibrators (Urine Calibrator Lyophil RECIPE GmbH, Munich, Germany). The results are validated using normal and pathological controls (ClinChek Urine Control L1, L2, RECIPE GmbH, Munich, Germany) reconstituted in high purity water and stored in single-dose aliquots at −20 °C until used. The concentration values are expressed as µmol/mmol creatinine. As demonstrated by Stauch Th and colleagues in 2017, in the occasion of the international congress of porphyrins and porphyria in Bordeaux, ALA and PBG are considered over the normal limits if the concentration values are >5 and >1.5 µmol/mmol creatinine, respectively. A high level of ALA and a normal level of PBG indicate either the rare ALAD deficiency porphyria or the more common heavy-metal intoxication and hereditary tyrosinemia type I caused by the inhibition of ALAD by lead and succinyl-acetone, respectively
[41][42]. High levels, of up to 20–50 times the normal values, of both the metabolites establish the diagnosis of an acute attack, while moderate ALA and PBG levels could indicate VP and HCP
[6]. The majority of the patients affected by acute porphyria after the symptomatic period may revert completely, while a few might become clinically asymptomatic along with persistent moderate increments in the heme precursors.
Over the last few decades, the technique of liquid chromatography-tandem mass spectrometry (LC-MS/MS) has been employed in numerous clinical biochemistry applications. In comparison to traditional methods, LC-MS/MS has major advantages of higher analytical sensitivity, specificity, and diagnostic reliability. About porphyria, the LC-MS/MS technique has been applied successfully for the simultaneous quantification of ALA and PBG in urine and plasma samples
[43][44][45][46].
Notably, significantly lower levels of urinary ALA and PBG could be measured using MS-based methods, particularly for healthy individuals, confirming that these methods have a higher selectivity compared to the colorimetric ones
[40]. The difference is probably due to the presence of contaminant molecules in the urine, which react with Ehrlich’s reagent. Recently, LC-MS/MS measurements of a large number of healthy subjects established the upper limit of the normal values (ULN) of ALA and PBG as 1.47 and 0.137 µmol/mmol creatinine, respectively
[47], and it was reported that during an acute attack, these values could reach 40 and 55 µmol/mmol creatinine, respectively
[45]. Moreover, sample preparation using a solid-phase extraction (SPE) system allows the detection of concentrations as low as 0.05 µM
[46]. Therefore, measurements of porphyrin precursors in plasma and tissue samples have become achievable. Floderus et al. quantified the plasma levels of ALA and PBG in 10 asymptomatic AIP patients and reported the mean concentrations to be 1.7 and 3.1 µmol/L, respectively, which were significantly higher than those of the healthy subjects (0.38 and <0.12 µmol/L, respectively)
[43]. However, during an acute attack of porphyria, ALA and PBG concentrations may increase dramatically, up to 13 µmol/L
[44].
Since the variation over the time of the plasma concentrations of ALA and PBG is reported to be highly correlated with urinary concentrations
[43], these measurements may contribute to the monitoring of AIP patients during the course of an acute attack
[48], particularly if the patients are in an anuric state. Plasma ALA and PBG levels could also be useful in evaluating the safety and the pharmacokinetic effects of the existing or future therapies in AHP patients
[49]. However, not all hospitals are equipped with LC-MS/MS, restricting this technique to a few specialized centers.
3.2. Measurement of Urine Porphyrins
The differential diagnosis of porphyria relies on the measurements of porphyrins and relative isomers in urine, feces, and plasma. As plasma porphyrin separation finds a practical application only in patients with renal failure, this topic is not treated in this review.
Although various methods have been developed for the analysis of urine porphyrins, reverse-phase high-pressure liquid chromatography coupled with fluorescence detection (FLD-HPLC) has become the gold standard method for this purpose
[50][51][52]. The success of this technique is attributed to its capacity to separate the physiologically relevant porphyrins as free acids and resolve type I and type III porphyrin isomers simultaneously
[53]. Since the fluorescence detection enhances the specificity of the analytic method, matrix extraction procedures are not required, and acidic urine samples could be directly injected.
Typical chromatographic runs are performed on a C18-bonded silica stationary phase using a linear gradient elution system from 10% (v/v) acetonitrile in 1M ammonium acetate, pH 5.16 (phase A) and 10% acetonitrile in methanol (phase B). Porphyrins containing from two to eight carboxylic groups, including the resolution of type I and type III isomers could be achieved in less than 30 min. The excitation and emission wavelength ranges used are usually around 395–420 and 580–620 nm, respectively. Direct standardization is obtained by comparison to chromatographic runs of a suitable calibration standard that are now commercially available (RECIPE GmbH, Munich, Germany; Chromsystems GmbH, Gräfelfing, Germany). The total urine porphyrin measurement is calculated as the sum of the chromatographic fractions and the single fractions as relative percentages. As spot urine samples are commonly used, the results should be normalized on the creatinine concentration.
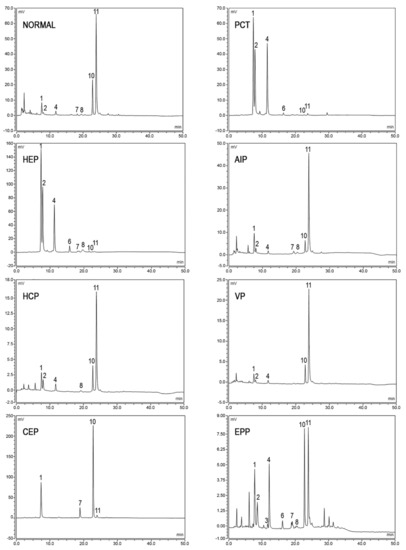
In normal subjects, the total porphyrins excretion is found below 35 nmol/mmol creatinine, COPRO predominate on URO, while hepta-, hexa-, and penta-carboxyl porphyrins are present only in small amounts. Moreover, relative concentrations of COPRO isomer III are higher than COPRO isomer I.
In porphyric patients, the excretion of urinary porphyrins varies in relation to the enzymatic defect underlying the particular type of porphyria and with respect to the disease stage
[54]. URO, both type I and type III isomers, and hepta- type III are evidently raised in PCT and HEP, since the type I isomers of URO and COPRO are detected in CEP almost exclusively. In acute hepatic porphyrias (AIP, VP, and HCP), porphyrins’ excretion is extremely variable from normal values in the asymptomatic phase to very high values observed during the acute exacerbation of the disease. In AIP, a pattern of the marked elevation of URO I and III isomers, along with less pronounced elevations of COPRO III and I, is detected, while HCP and VP demonstrate a marked elevation of COPRO III. In EPP, total urine porphyrins are normal; however, abnormal chromatographic profiles are frequently observed. In particular, higher relative amounts of COPRO I are commonly observed as a consequence of potential hepatic implications. In fact, increased levels of porphyrins excretion may also occur in several physio-pathological conditions including hereditary hyperbilirubinemias, toxic syndromes or liver diseases
[55]. Examples of porphyrins’ pattern of normal and pathological subjects are provided in
Figure 3.
Figure 3. Chromatographic profiles of urine porphyrins. The figure shows the patterns of excretion of healthy individuals (NORMAL) and patients (PCT, HEP, AIP, HCP, VP, CEP, EPP). Fluorescence peaks are identified as uroporphyrin I (1), uroporphyrin III (2), heptacarboxylic acid porphyrin I (3), heptacarboxylic acid porphyrin III (4), hexacarboxylic acid porphyrin III (6), pentacarboxylic acid porphyrin I (7), pentacarboxylic acid porphyrin III (8), coproporphyrin I (10), coproporphyrin III (11).
Recently, protocols using mass spectrometry (LC-MS/MS) to separate and detect porphyrins have also been reported to facilitate the clinical diagnosis of porphyria
[45]. However, the low prevalence of this equipment in hospitals restricts the application of such protocols to the identification and characterization of unknown porphyrins in research.
3.3. Analysis of Fecal Porphyrins
Two different systems may be employed for the analysis of fecal porphyrins. First, the total fecal porphyrins can be quantified using a spectrophotometric
[55][56] or fluorimetric
[57] method and then separated by HPLC to obtain porphyrin patterns
[58]. Second, total fecal porphyrins can be calculated as the sum of fractions following HPLC analysis
[59]. The former approach is considered more suitable for routine use in clinical analysis, while the latter is technically more correct and, therefore, allows for quantification with a higher reliability
[60].
In the widely employed method reported by Lockwood et al., porphyrins are extracted from a small sample of feces in the aqueous acid phase using the solvent partition
[56]. Briefly, 25–50 mg of feces is processed by the sequential addition of 1 mL of concentrated HCl to dissolve the organic matrix, 3 mL of diethyl ether to eliminate the contaminants—chlorophylls and carotenoid pigments, and 3 mL of water to avoid any alteration in protoporphyrin. The hydrochloric acid extract is then analyzed by performing a spectrophotometric scan between 370 and 430 nm, which includes the Soret region. After background signal subtraction, the absorbance measured at the maximum peak is used for calculating the total porphyrin content. It is also necessary to separately evaluate the water percentage in the feces sample to report the result as nmol/g dry weight (total fecal porphyrin normal value < 200 nmol/g dry weight)
[56]. Although no commercial reference material is available, an internal quality control (ICQ) could be prepared from the patient’s specimens to check the day-to-day reproducibility. The obtained hydrochloric acid extracts may be directly injected into HPLC systems for subsequent characterization using the same protocol as the one used for urine porphyrins chromatography. The calibration solution is prepared by adding appropriate concentrations of meso- and proto-porphyrin to the urine calibrators.
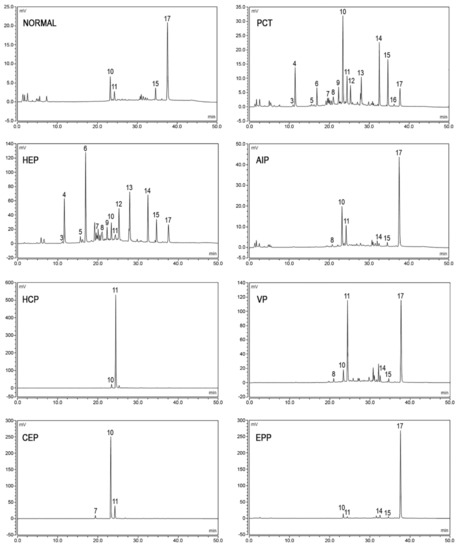
The fecal excretion of porphyrins increases in hepatic PCT, HCP, and VP, in erythropoietic CEP and HEP, and sometimes in EPP and XLP. The detection of the specific patterns of fecal porphyrins allows the differentiation of these enzymatic disorders that otherwise share similar clinical presentation and overlapping biochemical characteristics. Feces samples from healthy subjects, as well as from porphyria patients, contain varying amounts of dicarboxylic porphyrins, particularly deutero-, pempto-, and meso-porphyrins, in addition to protoporphyrin. These dicarboxylic porphyrins lack a diagnostic relevance, depending on the gut microflora
[61] and even on the diet
[62]. Moreover, gastrointestinal bleeding may interfere with feces analysis, causing an anomalous increase in the levels of protoporphyrin and the dicarboxylic porphyrins derived from it
[63]. Tricarboxylic porphyrins remain generally undetected, although the presence of harderoporphyrin in feces
[64] played a major role in the identification of a variant of homozygous HCP (harderoporphyria). Uroporphyrin is excreted prevalently in the urine and may be detected in feces only in trace amounts.
Fecal porphyrins profiles of PCT and HEP are the most complex, with a wide range of peaks. It is possible to recognize the COPRO I isomer, which always prevails over COPRO III, although increasing signals corresponding to epta-, esa-, and penta-porphyrins, with the prevalence of isomer III, are also observed. Since UROD is the enzyme that catalyzes the sequential decarboxylation of uroporphyrin to coproporphyrin, its activity deficit leads to the accumulation of all the porphyrin intermediates in PCT. In HEP, the activity of UROD is severely compromised and the porphyrins containing a higher number of carboxylic groups are represented more. PCT chromatographic profiles are also characterized by the presence of isocoproporphyrin and its derived metabolites hydroxy-, keto-, deethyl-, and dehydro-isocoproporphyrin. These molecules are commonly identified as a diagnostic sign of symptomatic PCT
[61]; nevertheless, diagnoses based on the presence of isocoproporphyrin in the feces are prone to be erroneous.
The chromatographic profiles of the fecal porphyrins of AIP and ADP patients are similar to those of healthy subjects. On the other hand, the fecal porphyrin chromatograms of HCP patients are easily recognizable by the presence of a prominent peak corresponding to COPRO III. The peaks for both PROTO and COPRO III are elevated in the chromatographic profiles of VP patients. In particular conditions, such as hepatitis or drug consumption, which inhibit the UROD activity, HCP and VP patients may exhibit fecal porphyrin patterns quite similar to those of PCT patients, including the isocoproporphyrin series. Since the relative abundance of COPRO III is always higher compared to isomer I in VP and HCP, while the opposite is true for PCT, the ratio between the coproporphyrin isomers serves as a suitable diagnostic parameter
[65][66]. The fecal porphyrins pattern in CEP contains an elevated peak corresponding to COPRO I. Finally, the chromatograms of EPP and XLP patients present an elevated PROTO peak. Examples of the porphyrins pattern of normal and pathological subjects are provided in
Figure 4.
Figure 4. Chromatographic profiles of fecal porphyrins. The figure shows the patterns of excretion of healthy individuals (NORMAL) and patients (PCT, HEP, AIP, HCP, VP, CEP, EPP). Fluorescence peaks are identified as heptacarboxylic acid porphyrin I (3), heptacarboxylic acid porphyrin III (4), hexacarboxylic acid porphyrin I (5), hexacarboxylic acid porphyrin III (6), pentacarboxylic acid porphyrin I (7), pentacarboxylic acid porphyrin III (8), hydroxyisocoproporphyrin (9), coproporphyrin I (10), coproporphyrin III (11), deethylisocoproporphyrin (12), isocoproporphyrin and dehydroisocoproporphyrin (13), deuteroporphyrin (14), pemptoporphyrin (15), mesoporphyrin (16), protoporphyrin (17).
3.4. Erythrocyte Porphyrins Measurement
Initially, to assay the total erythrocyte porphyrins, fluorometric methods involving the acid extraction procedure that converted ZnPP to its metal-free form PPIX were used
[67]. Later, when it became possible to detect ZnPP in the whole blood samples without prior acid extraction, hematofluorometry methods were developed for this assessment
[68][69]. Finally, the application of high-performance liquid chromatography (HPLC) coupled with fluorometric detection to separate and quantify unchelated PPIX and ZnPP simultaneously commenced
[70][71]. A method using derivative variable-angle synchronous fluorescence (DVASF) for determining PPIX and ZnPP simultaneously in a whole blood sample, avoiding the spectral compensation factor for PPIX and the chromatographic separation, has been also reported
[72]. A simple, rapid, and specific HPLC method is described here. Whole blood samples are collected in vacutainer tubes containing the anticoagulant K3EDTA and then stored at −20 °C in the dark. At the time of analysis, samples are thawed to room temperature and mixed well, followed by the dilution of 60-microliter aliquots in 200 µL of lysing solution (4% aqueous formic acid) and the extraction of porphyrins using 900 µL of acetone. An aliquot of the extracted porphyrins is injected directly into the HPLC system, using a Chromsystem C-18 column (Chromsystems GmbH, Gräfelfing, Germany) for chromatographic separations. ZnPP and PPIX are eluted in isocratic conditions (90% methanol in an aqueous 1% acetic acid solution) at 42 °C and subsequently detected using fluorescence excitation/emission wavelengths of 400/620 nm (1–7 min) and 387/633 nm (7–15 min), respectively. In order to quantify PPIX and ZnPP, homemade calibration curves are used, and the analytes are observed to be linear in the concentration ranges of 1.5–50 and 2–100 µg/dL, respectively. The results are reported as the sum of the PPIX and ZnPP peaks, and the concentrations in the blood are expressed as both µg/g Hb and relative percentage of each porphyrin. The normal level for the sum is <3 µg/g Hb and the normal percentage ranges are ZnPP 80–90% and PPIX 10–20%. Erythrocyte porphyrins are abnormally increased in EPP, XLP, CEP, and HEP, although the percentage of each porphyrin differs among these disorders, as described in
Table 2. It is noteworthy that elevated erythrocyte protopophyrins along with normal ZnPP support the diagnosis of the classical form of EPP, while an elevation in both components with balanced percentages occurs in the XLP. High levels of erythrocyte porphyrins also occur in the case of exposure to lead from both environmental and occupational conditions
[73], in iron deficiency
[74], as well as in sideroblastic anemia, all of which are conditions associated with elevated Zn PP. In CEP and HEP, the predominant porphyrins may be uroporphyrins or ZnPP, depending on the phenotype expression.