1000/1000
Hot
Most Recent

Carbon capture use and storage (CCUS) is considered an effective mitigation strategy to reduce the most challenging emissions from heavy industries and gas processing. The safe transport of carbon dioxide via pipelines is an important aspect for developing large-scale Carbon Capture and Storage projects. Dispersion modeling for heavy gas such as carbon dioxide is considerably different from natural gas. The set up for modeling simulations is more challenging than conventional natural gas pipeline for several reasons, such as the differences in thermodynamics that must be considered. Moreover, when the carbon dioxide is transported in dense or liquid phase, the rapid phase changing, and possible consequent formation of solids should be considered. Finally, the equation of state required for accurate prediction of parameters is generally different than the ones applicable for natural gas.
The risk management strategy should be based on relevant industry good practice which focuses on inherent safety and the prevention of incidents with the potential to endanger people, the environment, or properties. Compared to natural gas, there are less companies with relevant experience in full-scale CCS projects development and few projects has been completed so far [1]. Thus, great care should be taken during hazard identification and management when dealing with CCS projects. According to DNV-GL [2], carbon dioxide pipelines should be designed with acceptable risk. The growing interests from energy companies in CCS will lead to the development of new generation CO2 pipeline systems that will require thorough risk assessments and to be design within acceptable risk levels as required by DNV-GL [2]. Since the fluid is usually transported in dense phase, there is the potential for large inventories of CO2 being released in the atmosphere which could reach populated areas with hazardous concentrations. According to ISO 13623 [3] and DNV-GL-RP-F104 [2], CO2 is categorized as a category C fluid. However, there is a guidance note stating that it is recommended that CO2 is categorized by category E unless long operational experience exists. This requires higher safety factors in the design.
To properly assess the risk of an installation, it is necessary to evaluate the potential consequence of an accidental release. The complex thermodynamic of the phenomena and the limited experience in handling CO2 are points of concern that need further investigation for a proper assessment.
Experimental release tests can be very expensive and usually are not affordable for small-medium laboratories, especially for large-scale scenarios. Several experimental works have been reported in the literature; these can be classified in large-scale and small scale experiments. Furthermore, the aim of the studies can be considered to be an additional parameter for the categorization. Most of the experimental work analyzed can be divided between far-field and near-field modeling. Studying the behavior and thermodynamics evolution in the near field is typically carried out in small scale set up or laboratory scale. During a release, the monitoring of far-field evolution has been developed in large-scale or full-scale outdoor experiments. Since the costs and infrastructure necessary for a large-scale set up are not easily sustainable by a single research center or university, several Joint Industry Projects (JIP) and Research Projects (RP) have been developed over the years. The most important JIPs and RPs are reported in Table 1 with the associated period, scale, and objectives.
Table 1. Relevant CO2 pipeline related JIPs and RPs programs during the years.
| JIP/RP Name | Years/Period | Scale | Objectives and Scope |
|---|---|---|---|
| CO2SAFEARREST | 2016–2019 | Full-scale | Burst tests research program. Two full-scale tests with buried pipeline (CO2-N2 mixture), 24 inches. |
| COSHER | 2011–2015 | Large-Scale | Obtain data to support the development of models to determine safety zones/consequence distances. |
| CO2PIPETRANS | 2009–2015 | Medium-Scale Large-Scale |
Fill the knowledge gap identified in the DNV-RP-J202. Results of the project were included in DNVGL-RP-F104 (2017). |
| COOLTRANS | 2011–2015 | Large-Scale | Identify and propose solutions to key issues relating to the safe routing, design, construction and operation of onshore CO2 pipelines in the UK. |
| CO2PIPEHAZ | 2009–2013 | Small Scale Large-Scale |
Improve the understanding of the hazards represented by CO2 releases. |
| CO2QUEST | 2013–2016 | Small Scale Medium-Scale |
Study the impact of the quality of CO2 on storage and transport. |
| CATO | 2004–2008 2010–2014 2015-ongoing |
N/A | A national program, which includes complete studies in all aspects of CCS. |
| CO2EUROPIPE | 2009–2011 | N/A | Outline guidance to elements of the European plan to develop large-scale EU CO2 infrastructure. |
| CO2RISKMAN | 2010–2013 | N/A | Development of industry guideline to assist the designer and projects on the emerging CCS industry. Potential hazards associated with handling CCS CO2 streams are discussed. |
Particular attention has been paid to large-scale tests for accuracy and readiness to develop validation and comparison studies by reducing the scale-up errors compared to operating pipelines. To highlight the main aspects related to a CO2 release, a schematic diagram has been reported in Figure 1. The release can be divided in near-field (phase change, expansion, air-mixing and solid formation) and far-field zone, where the atmospheric dispersion of the heavy CO2 cloud continues at large distances start.
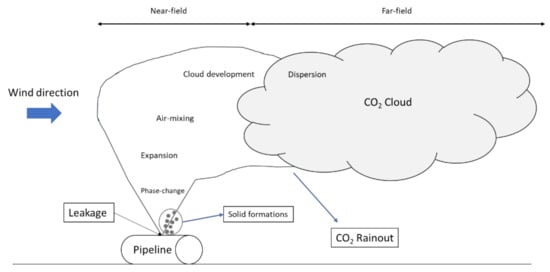
Figure 1. Schematization of a CO2 release from an onshore pipeline.
Since near-field modeling can strongly impact the far-field modeling and the definition of safety distances, particular attention should be reserved to these aspects. Pursell [4] presented some results from laboratory scale release tests performed in Health and Safety Laboratory (UK). The experiments were performed both for liquid and gas phase of CO2 from release orifices of 2 and 4 mm (diameter), the set up was connected to a pressurized vessel containing CO2 at pressure from 40 to 55 barg. The rapid expansion of the fluid downstream the orifice occurs approx. following an isenthalpic expansion to atmospheric pressure, then the CO2 jet continues to expand as it entrains and mixes with the surrounding air.
Useful data were obtained by analyzing the images, such as the length of the expansion zone and the effective diameter of the jet at the point where it reaches atmospheric pressure. Since it is not easy to define the location of the shock front in liquid releases, the effective diameter could be useful for dispersion calculations (assuming a gas phase equivalent diameter) since it is often used to define the size of the pseudo-source. Results showed that liquid releases led to an effective diameter up to 30% larger than the corresponding gas phase in the diameter of the analyzed orifice. Guo et al. [5] studied the near-field characteristics and dispersion behaviour of supercritical [6], gaseous, and dense-phase CO2 experimentally. A large-scale pipeline set up with an internal diameter of 233 mm and a total length of 258 m was constructed during the CO2QUEST project. Six tests have been performed, and several orifice diameters have been tested from 15 mm to full-bore rupture. The near-field behavior and the under-expanded jet flow structure have been analyzed; solid phase formation was observed. The sublimation of solid CO2 removes heat from the gas phase with a consequent temperature reduction. The formation of solid CO2 can impact the properties and the shape of the cloud. Based on the experimental data performed by Guo et al. [5], the development of the visible cloud can be divided into three stages: a “rapid expansion”, a “metastable stage”, and a “slow attenuation stage”. The distance of the 50,000 ppm concentration contour for three orifice diameters was determined in all the experiments. The maximum safety distance of 160 m has been measured for the full-bore rupture at the initial condition of 9.1 Mpa at 21.6 °C (dense phase). Safety distances measured for dense phase were consistently greater than gas phase tests.
An experimental study of supercritical CO2 leakage has been reported by Fan et al. [7]. The pressure and temperature conditions analyzed varied from 81 to 110 bar and 34.9 °C to 100.9 °C. The authors noted how the mass flow rate decreases with the increase of upstream temperature and length-diameter ratio and increases with the increase of upstream pressure. However, the effect of upstream temperature variation (at approx. 100 bar) on the jet structure was not so evident in the range between 37.6 °C and 40.1 °C.
An experiment with various measurement methods was developed by Teng et al. [8] to carry out controllable CO2 release from a high-pressure vessel. Pure liquid CO2 has been used for the experiments, orifice diameter of 1 and 2 mm. Initial pressure varies from 80 bar to 100 bar, while temperature from 301 K to 313 K. The lowest temperatures measured were −41.9 °C (1 mm diameter) and −45 °C (2 mm diameter). The maximum velocity along the jet centreline was 250 m/s. The results suggest that for a supercritical CO2 leakage, dry ice particles with size between 1 and 3 μm can be formed. The initial temperature shows limited impact on the size of dry ice particles, while a wider size distribution can be addressed to a higher initial pressure. Li et al. [9] developed a reduced scale facility with dry super-critical CO2 to analyse the jet plume’s early stage flow characteristics in the near-field. Initial pressure was set up to 8.02 Mpa; the velocity in the centreline of jet plume was measured from different leakage sizes (0.5, 1, 3, 5 mm), showing a correlation with the depressurization process during the leakage.
Ahmad [10] reported the results based on COSHER JIP; a large-scale rupture test was conducted on a loop test built in Spadeadam (UK). A 219 mm diameter pipeline buried underground filled with dense phase CO2 has been used. Temperature, pressure, concentration distribution and dispersion cloud have been considered with low wind conditions 1.9 m/s. Approximately 136 ton of CO2 were released in 204 s. The maximum height of the plume was registered at 60 m from the ground while the maximum horizontal extension at 400 m, the minimum temperature registered during the release was −78 °C. The test has been conducted at approx. 150 bar and the average temperature of the fluid in the reservoir was around 13 °C.
In 2017, other tests were carried out in Spadeadam (UK); two full-scale burst tests were performed during CO2SafeArrest JIP to evaluate the fracture propagation and arrest characteristics and CO2 dispersion in the atmosphere [11][12]. The outer diameter of the test section was a steel pipe 610 mm, 85 m long, connected to approx. 120 m long reservoirs at both ends. A mixture of 91% CO2 and N2 pressurized to about 15 MPa was adopted. The pipeline rupture was induced by initiating the crack with explosives; several sensors (temperature, pressure, oxygen cells) have been positioned over a pattern terrain in the vicinity of the crack. Two burst tests have been conducted; for the first test, all the pipe test section was buried under a 1 m deep soil cover, while in the second test, only half the length of the pipe test section was buried. The resulting crack propagated in both directions as the pipe wall was torn open sideways. The CO2 cloud reached an altitude peak of 250 m, as well as the debris thrown out of the crater formed. The crater extension measured approx. 45 m in the horizontal (pipe direction) while the perpendicular extension varied between 5.8 m and 9 m. The average width of the crater is about 7.4 m, which is 12 times of the outer pipe diameter.
In the work of Allason et al. [13], the COOLTRANS experimental campaign has been described and some considerations are reported. During the program, vertical pipe venting and puncture tests have been performed.
In the venting experiments no rain out of solid CO2 was observed and the plume dispersed upward and reached the ground some distance away downstream of the release but a low concentration. The pressure along the pipeline was observed to be relatively constant and there is an indication that some solid CO2 could form during vent operations. The experimental set up for the puncture experiments consisted of 914 mm outer diameter pipeline buried 1.2 m below ground and supplied with dense phase CO2. The puncture experiments have shown that the nature of the surrounding soil can impact the nature of the flow into the atmosphere. The flow from the punctures was observed to stall in the atmosphere above the source and, in low wind speed conditions, a ‘blanket’ was observed to form around and over the source. Moreover, the results from the puncture tests show that the random variations in wind direction and turbulent fluctuations within the plume, combined with the high toxicity index for CO2, lead to differences in the calculated integrated dose.
The experimental testing campaign review clearly highlights the differences between the management of a natural gas release and a dense phase CO2 release. The high CO2 density and the dry ice formation are two of the main points that should be analyzed carefully and can become challenging in the modeling phase. The rapid depressurization and the phase changing from liquid to gas is also a point of concern that will require a dedicated modeling approach that is substantially different from natural gas. As with natural gas the crater formation and the height reached from the CO2 cloud can impact the safety distance required, thus should be modeled accurately.
Modeling a release of a CO2 pipeline requires the assessment of some important aspects, such as transient conditions, multi-phase jet, as well as the dispersion behavior. A rapid pressure drop will follow the release of CO2 from a pipeline; the pressure and temperature reduction a phase transition from liquid-vapor is expected. Moreover, for lower temperature the formation of solids is also a possibility. The phase transition can impact the flow conditions within the pipeline and the properties of the fluid. The precise simulation of transient depressurization, with regards of flow rate and thermodynamic properties of CO2 during the release, will impact the accuracy of the cloud dispersion prediction. Specific focus must be reserved to phase transition and density prediction of the CO2 during transient operations in order to better predict solid formations. Release and dispersion studies are required for risk evaluations. Three main steps can be identified in dispersion modeling:
Some specific difficulties for modeling CO2 releases can be highlighted, which may constitute a limitation in developing accurate simulations, in particular: the selection of an Equation of State for an accurate description of the thermodynamic properties throughout the release process, the modeling of phase changing (from dense phase to gaseous), prediction of solid phase formation, the validity of homogeneous equilibrium (HEM) assumption. Another aspect to be considered is the very limited experience in CO2 pipeline modeling; for this reason, most codes and simplified models need to be assessed and validated with experimental tests data. Two main approaches are available to model an accidental release:
Simplified models
CFD models
The simplified models usually require very low CPU usage compared to CFD models; hence they are faster and optimized for risk analysis. However, a simplified model, such as an integral model, is based on several assumptions and simplifications to the physics of the phenomena; for these reasons they need to be extensively validated with real case data, experimental tests. CFD models can provide a very detailed description of the physics and the behavior of a CO2 release; this kind of approach is required when a complex topography, specific environment conditions or presence of buildings or other obstructions in the nearby area. These models require high experience and specific knowledge from the user to be set up and executed, compared to simplified integral models. Moreover, the uncertainties specific to CO2 related to limited experience and optimization often require custom-made inputs and user-defined functions to be implemented in commercial CFD software.
The most commonly available simplified models are the integral models implemented in existing commercially available software. Since most risks in the oil and gas industry come from flammable liquids or gases, none of them was originally designed to take into account inert gas such as CO2. The main models identified are:
FRED
PHAST
ALOHA
EFFECTS
Specific attention has been reserved to commercially available software and their capability to handle CO2 releases. The main CFD codes identified are:
ANSYS FLUENT
ANSYS-CFX
FLACS
OpenFOAM
FLUIDYN PANACHE
It is suggested to approach the near-field modeling and the far-field modeling in two separate ways. The near-field modeling of a CO2 release requires specific knowledge of the gas-dynamic phenomena in sonic and supersonic transitions, namely Mach number, wave motion, and sonic speed. The CFD code that performed better when compared to experimental tests was ANSYS-CFX. Even though simulating the entire phase transition is theoretically feasible, in practice, it is hard to simulate with a single CFD code, with issues mainly related to time steps and grid refinement. The unstructured hexahedrons mesh available by default in the ANSYS packages seems more suitable for specific cases when obstacles and hilly terrain need to be modeled. Finally, the terrain effects can be dominant for CO2 dispersion; for this reason, CFD modeling, especially in the vicinity of depression or large differences in terrain heights close to a CO2 pipeline route, can overcome the limitations of simple models such as integral models.