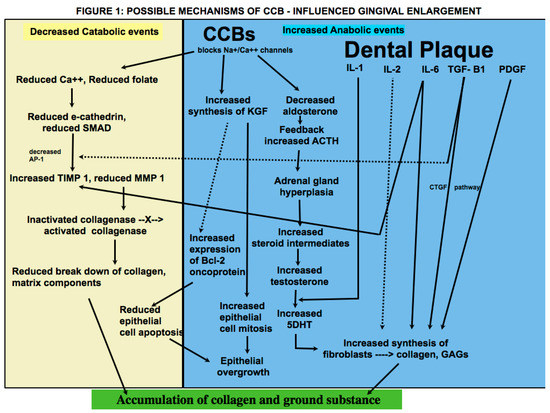
At the cellular level, drugs across genres associated with GE appear to have a similar mechanism of action
[1][9]. Brown et al. presented a hypothesis that consolidates previous theories on the pathogenesis of DIGE
[9]. These drugs have an inhibitory effect on sodium/calcium cation channels (Na/Ca++), which results in blockage of influx of calcium ions into fibroblasts
[9]. Due to regulatory effect of calcium channels on folate (FA) uptake, this results in reduced uptake of FA into fibroblasts
[9]. This in turn causes a decline in e-cathedrin, a calcium-dependent gene molecule crucial for epithelial cell to cell adhesion, and SMAD (Small Mothers Against Decapentaplegic) gene, a transducer-signaling protein for transforming growth factor-beta 1 (TGF beta1)
[9]. The TGF-beta family of growth factors regulates cell development and proliferation via an incredibly complex system of cross-talking signaling pathways of which the SMAD pathway appears to play a key role in DIGO through its action on tissue inhibitor of metalloproteinase-1 (TIMP-1) and matrix metalloproteinases (MMPs)
[9]. Interleukin-6 (IL6), a pleotropic cytokine, also acts on TIMP-1
[8]. There is a resultant increase in TIMP-1 and a decrease in MMP-1
[8][9], which is required to activate collagenase. The reduced collagenase activity results in decreased collagen breakdown, and accumulation of collagen and other extra cellular matrix (ECM) content occurs as a consequence. In addition, nifedipine has been demonstrated to cause an increased synthesis of keratinocyte growth factor (KGF) by fibroblasts in vitro
[9]. KGF is known to regulate epithelial growth by stimulating cell mitosis and reducing apoptosis of epithelial cells
[9][13]. Increased synthesis of B-cell lymphoma 2 (BCl2) oncoprotein, which inhibits apoptosis of keratinocytes, has also been found in amlodipine-associated GO
[9]. It is also important to remember that gingiva is a target tissue for androgen metabolism and contains receptors for 5-alpha-dihidrotestosterone (DHT). Nyska et al. proposed a mechanism by which CCB-influenced GO developed in a canine model
[19]. CCBs block aldosterone synthesis due to their calcium flux inhibiting properties, which creates a feedback increase in pituitary ACTH secretion
[19]. This causes adrenal gland hyperplasia and increased synthesis of steroids, such as pregnenolone
[19]. There is a resultant increase in production of testosterone, an anabolic steroid that causes proliferation of fibroblasts and collagen via DHT receptors
[19]. Sooryamurthy et al. similarly reported exaggerated DHT production from testosterone by gingival fibroblasts in patients with GE who were on nifedipine and/or cyclosporine
[20].
Figure 1 shows the possible mechanisms of CCB influenced gingival overgrowth.
A plethora of studies have shown that presence of dental plaque is critical in the development and manifestation of DIGE
[1][7][9][13][21]. Bacterial plaque in gingival crevicular fluid (GCF) causes an increase in synthesis of both pro inflammatory cytokines, such as interleukins IL-1
[1], IL-2
[21], 1L-6
[8], and anti-inflammatory cytokines and mitogens, such as TGF-beta 1
[22], platelet-derived growth factor (PDGF), and insulin-like growth factor (IGF)
[23]. It has been proposed that stimulation of gingival fibroblasts occurs when they are exposed to CCBs and pro-inflammatory cytokines, such as IL-1
[1][8]. Kasasa et al. reported that IL-1 caused an increase in production of 5DHT from labelled testosterone in human gingiva and periodontal ligament (PDL)
[24]. In addition, increased expression of IL6 has been found in cyclosporin-, phenytoin-, and nifedipine-influenced GE
[8]. IL-6, which has pro- as well as anti-inflammatory tendencies, causes an increase in fibroblast proliferation and glycosaminoglycans synthesis by targeting fibroblasts and accumulation of connective tissue (CT) components by increasing TIMP-1 synthesis
[8]. Growth factors, such as TGF-beta, connective tissue growth factor (CTGF), and PDGF, regulate fibroblast proliferation and collagen synthesis and reduce cell apoptosis
[25]. Taken together, DIGE appears to be a result of an imbalance between anabolic and catabolic events, and the cumulative effect is accumulation of gingival collagen and ground substance. There is conflicting evidence on whether CCBs have a direct or indirect effect on gingival fibroblasts. Nishikawa et al. demonstrated in an in vitro study that in lower doses, nifedipine did not have any effect on fibroblast growth and proliferation
[21]. They concluded that the effect of nifedipine is indirect, as the presence of local inflammatory factors is necessary for nifedipine to stimulate cytokine and/or DHT production by gingival fibroblasts, resulting in increased collagen synthesis.
[21]. On the other hand, it has been theorized that inflammation may develop as a result of direct effects of the drug in gingival crevicular fluid (GCF)
[9].
Effect of Dose on Pathogenesis
The dose of the drug is considered to be an important factor in the development and severity of DIGE by some authors who noted that the severity of the condition was directly related to the dose
[7][13], while others have disproved the theory due to presence of the condition even in some patients who were taking relatively lower doses
[26][27].
It has been shown that GE occurs when nifedipine is taken at a dose of 5 mg/day, 10 mg/day, or 20–40 mg/day
[7][21][22][28]. The condition is either noticed or treated about 2–18 months after medication is started
[21]. Amlodipine has been associated with GE even in doses as low as 2.5 mg/day taken over a longer duration of time
[13]. Khzam et al. reported a case of DIGE that occurred within a few days after use of felodipine 400 mg for hypertension
[18]. Ikawa reported a case where gingival enlargement was seen in a patient taking manidipine at a dose of 20 mg/day for several months before surgical intervention
[29]. The range of dose and time of occurrence may be attributed to factors such as the drug used, oral hygiene habits, concurrent use of medication, such as statins, and other existing diseases, such as diabetes mellitus. The appearance of DIGE even in low doses should be factored in while determining the possibility of drug modulation as the initial course of treatment.
4. Conclusions
When enlargement of an organ occurs due to an increase in the number of cells, the condition is referred to as hyperplasia, whereas when enlargement occurs due to increase in the size of the cells, it is referred to as hypertrophy. What was termed as drug-induced gingival hyperplasia is now known as drug-influenced gingival enlargement, categorized under “dental biofilm induced gingivitis” in the most recent classification scheme for periodontal diseases
[30]. This change in terminology reflects an evolution in understanding of the pathogenesis and development of DIGE. Whereas earlier studies focused on the theory that DIGE is caused by an increase in connective tissue elements, more recent studies suggest that it is a result of an imbalance between catabolic and anabolic mechanisms that are necessary for regulating the periodontium
[9]. Historically, the pathogenesis of DIGE has been described as being caused by inflammatory and noninflammatory mechanisms; however, they are not distinct or separate; rather, the pathways appear to be confluent in various steps of the disease. In addition, there is insurmountable evidence that dental plaque is necessary for the development and progress of the condition and that the offending drug mainly alters the response of the periodontium to plaque bacteria. It is also important to remember that plaque-induced gingival inflammation does not only contribute to the occurrence of DIGE but is also a consequence of it due to limited access for oral hygiene. The literature involving pathogenesis of CCB-influenced GE, treatment and clinical outcome of various treatment modalities is conflicting. Nevertheless, the subject is of great relevance due to the widespread use of the drugs that cause it, the pervasiveness of the systemic diseases that require their use, and the functional and cosmetic implications of DIGE. Within the limitation of this article, it may be inferred that genetic makeup, plaque induced gingival inflammation, type of drug used, duration of drug use, lifestyle and oral hygiene habits, treatment modalities utilized, and patient compliance are important prognostic factors that affect the prevalence and severity of the condition, long-term clinical outcome, and risk of recurrence following periodontal treatment. More studies that focus on predisposing factors should be undertaken that will aid in identifying individuals who are at greater risk for developing DIGE. Patients taking these drugs must be forewarned about this side effect so that they take adequate oral hygiene measures that will help prevent or at least minimize the condition. Longitudinal studies on causes of recurrence will help shed light on why periodontal therapy is successful in some patients, whereas in others, there is recrudescence of gingival enlargement.