1000/1000
Hot
Most Recent

Rheumatoid arthritis (RA) is a common chronic inflammation-mediated disorder having systematic complications. RA triggers a self-directed inflammatory and immunological cascade that culminates in joint destruction. Though a range of treatment options are available, none of them are without adverse effects, leading researchers to search for alternative solutions. Nanomedicine has emerged as a powerful therapeutic alternative, and selenium (Se) is an essential micronutrient trace element that has a crucial role in human health and disease. The potential of SeNPs can be attributed to the effect of functional groups bound to them, concentration, and most importantly to their nano range size. The antirheumatic effect of SeNPs is considerable due to its potential in amelioration of oxidative stress-mediated inflammation via downregulation of radical and nonradical species, markers of inflammation, and upregulation of inherent antioxidant defenses.
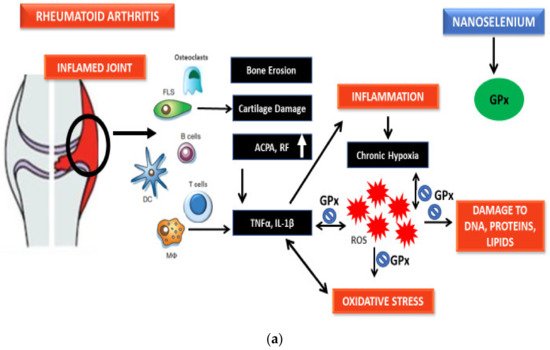
Rheumatoid arthritis (RA) is a common chronic inflammation-mediated disorder [1]. It is a long-lasting condition described as the inflammation of diarthrodial joints leading to symmetrical polyarthritis and synovial hyperplasia (swelling) that results in progressive destruction of cartilage and bones and loss of articular function that leads to the eventual deformation of joints [2]. Moreover, it is a systematic autoimmune disorder that can alter multiple organ systems [3].
The exact etiological milieu for RA is still not certain, but as an example of a chronic inflammation-mediated autoimmune disorder, it has been correlated to oxidative stress (OS), a state wherein a pool of reactive oxidative species (ROS) upregulates actively, either due to their enhanced generation, the decline in antioxidative defense mechanisms, or the combined effects of both, thus leading to altered redox signaling that is involved in the maintenance and progression of the disorder [4][5]. The therapeutic options for patients suffering from RA include nonsteroidal anti-inflammatory drugs (NSAIDs), glucocorticoids (GC), and disease-modifying antirheumatic drugs (DMARDs), but all of these available therapeutic remedies have associated adverse effects [1]. Thus, there is a prominent need to develop and test novel drugs that intend to ameliorate inflamed synovial joints and mitigate bone damage. Selenium (Se) is an essential micronutrient trace element having a crucial role in normal human functioning and has prominent relevance to several pathophysiological conditions [2].
In this regard, one of the most promising therapeutic solutions for RA is ‘nanomedicine’ [3] and has captured quite the amount of attention. Selenium nanoparticles (SeNPs) have become the centerpiece of attention due to their exclusive physical and chemical properties [6]. The SeNPs play an essential role in the antioxidant defense system that is crucial for reduction of oxidative stress [7].
In the past, lowered serum concentration of trace micronutrients has been demonstrated as a frequent event in autoimmune diseases [8]. Epidemiological reports proved that a low Se status can be a risk factor for RA, indicating the significance of antioxidants in controlling the maintenance and progression of the disease [9][10][11][12][13][14][15][16][17][18].
It has been reported that Se supplementation improves the condition of patients as well as reduces inflammation levels in experimental models, such as the granuloma pouch exudate, and in lupus mice or in adjuvant arthritis in rats [19]. Evidence has suggested that Se can decrease inflammation in autoimmune disorders [20].
Conventional treatment options for RA patients comprise NSAIDs, GC, conventional synthetic disease-modifying anti-rheumatic drugs (csDMARDs), and biologic DMARDs (bDMARDs), and all these available therapeutic remedies have associated side effects. Table 1 summarizes the diverse treatment options available for RA and their associated side effects.
| Drugs | Mode of Mechanism | Side Effects |
|---|---|---|
| NSAIDs | Inhibition of COXs | Cardiovascular risk, gastro-intestinal disorders, and renal malfunction |
| GCs | Inhibition of phospholipid release | Cardiovascular disorders, osteoporosis, insulin resistance, skin thinning, hypertension, and obesity |
| Conventional synthetic DMARDs | Disease altering activities | Interstitial pneumonitis, myelosuppression, hepatic cirrhosis, retinopathies, hypersensitivity, and allergic reactions |
| Biologic DMARDs | Inhibitors to immune mediators | Bacterial infections and high costs |
| Targeted synthetic DMARDs | Intracellular blockers of tyrosine kinase | Infections, headaches, hypertension, nausea, diarrhea, and high cholesterol levels |
RA continues to be a challenging disorder because all the above-mentioned recommended therapies do not often lead to a cure and are linked to frequent drug resistance and related side effects [21]. Hence, it is crucial to develop and test novel drugs that target inflamed joints and mitigate damage. In this regard, one of the most promising therapeutic solutions for RA is nanomedicine [3]. Hundreds of diverse nanomedicine formulations have been prepared and assessed over the years for various kinds of maladies. However, about 50 of such formulations are at present approved for clinical usage, and several nanomedicines are going through trials [22]. Nanoparticles (NPs) are defined as nano-range submicroscopic particles that have unique properties such as large surface area, nano size, surface charge, and chemistry, solubility, and multifunctionality [23]. NPs are deemed as being in a transitional stage between individual molecules and the analogous bulk materials, which allows them to possess peculiar properties that are unique from their molecular and bulk analogue counterparts [23][24]. Based on their unique properties, nanoscale materials and devices can interact with biomolecules from both the inside and on the cell surface that has the potential to detect disorders and deliver treatments. Hence, NPs have revolutionized healthcare as they facilitate research and development, help with early detection, enhance molecular imaging, and enable prevention, diagnosis, and control [25].
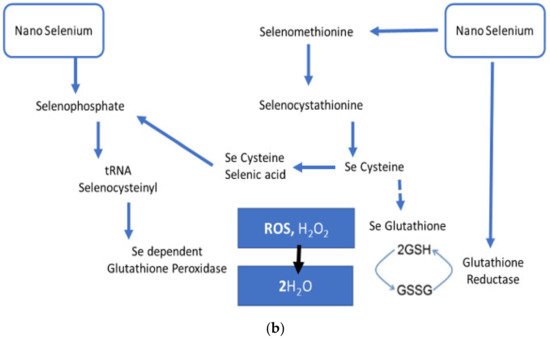
SeNPs have received attention due to their exclusive physical and chemical properties (i.e., mechanical, electrical, catalytic, and opt-magnetic properties) that are exhibited when this element is scaled down to the nano range as a result of high spatial confinement of nanomaterials, high surface-to-volume ratio, and large surface energy [26][27][28]. SeNPs, due to remarkable photoreactive, biocidal, anticancer, antidiabetic, antioxidant, antimicrobial, and anti-inflammatory properties in the healthcare arena, is being used in antimicrobial coatings, diagnostics, medical devices, nutritional supplements, and nanotherapeutics [29][30]. SeNPs have significantly emerged as a dual-targeting modality with both pro-oxidant and antioxidant potential dependent on subsequent duration, dose, and frequency as well as oxidation state. The pro-oxidant potential of SeNPs has been exploited in anticancer agents (chemotherapeutic drugs carriers). These NPs fundamentally localize in the malignant cells and lead to the production of reactive oxygen species, and hence cause cytotoxicity. The pro-oxidant mechanism of SeNPs follows the reduction of nano selenium via thioredoxin- and glutaredoxin-mediated redox signaling that leads to the generation of Se2− anion through the consumption of NADPH+H+ and stimulated production of ROS [31]. SeNPs play an important role in the antioxidant defense system, which is essential for reducing oxidative stress [7]. Se is an integral part of selenoproteins, such as glutathione peroxidases (GPxs) and TrxRs, which are needed for several biochemical reactions involved in normal antioxidant defenses [32]. SeNPs have been studied in different inflammation and redox imbalance-mediated disorders, such as cancer, diabetes, nephritis, and arthritis, and showed potential remedial uses [29].
Oral intake of NPs is regarded as the most suitable and cost-effective mode of supplementation. Nonetheless, the absorption of NPs is hindered by two gastrointestinal barriers: the intestinal mucosa and the mucus covering the intestinal mucosa [33]. In theoretical terms, NPs can pass through the intestinal epithelium via two transport methods: paracellular (between adjacent cells) or transcellular (through the cells) [34]. Intestinal epithelial cells can transfer NPs along with the mineral elements, though their ability is limited. Transcellular transport starts with endocytosis (pinocytosis or macropinocytosis) [35]. The absorption of the NPs depends on the size, surface hydrophobicity, and electric charge [36]. The epithelium of the digestive tract is comprised of lipids, resulting in a higher absorption rate of hydrophobic NPs than hydrophilic NPs. The absorption of 100 nm NPs in the digestive tract is about 15 to 250 times higher than that of larger NPs [37].
In a recent report, a comparison between SeNPs and selenomethionine (SeMet) in male C3H/HeJ mice to estimate the LD50 showed that SeNPs induce minor toxic effects compared to SeMet [38]. In short, SeNPs are less toxic, more bioavailable, and possess stronger biological properties than other organic and inorganic Se forms [39][40].
Qamar et al. reported the potential of SeNPs prepared from Trachyspermum ammi against RA in BALB/c mice models. SeNPs exhibited correction in a manner independent of dose in the redox state through the upregulation of antioxidant defenses and a reduction of paw edema as compared to the diseased group [41]. Hence, the antiarthritic potential of SeNPs is perhaps due to the reduction of ROS, inflammation-related markers, and increase in antioxidant protection as established from recent research.
Zingiber officinale (ginger)-derived SeNPs have been tested against aluminum chloride-induced hepatorenal toxicity in rats and provided significant antioxidant benefits through reduction in GSH, SOD, GPx, and malondialdehyde (MDA) levels [58]. Ginger-extract-made SeNPs have also been reported to have antioxidant-mediated anti-inflammatory properties and improved nicotine-induced renal inflammation-mediated impairment in rats [59]. Menon et al. also reported the antioxidant potential of Zingiber officinale-derived SeNPs through DPPH tests [60]. Given that long-term treatment using DMARDs and NSAIDs leads to renal and hepatic toxic damage in rheumatoid patients, [61][62] ginger-derived SeNPs can be a possible option to diminish the treatment-associated toxic effects in rheumatoid patients. Kameswari et al. reported on the potent free radical scavenging and anti-inflammatory potential of SeNPs derived from Acalypha indica extract [63]. Other SeNPs derived from plants have also showed significant antioxidant potential [64][65][66][67].