1. Introduction
The entire sweat gland system represents 1.6–5 million apocrine and eccrine glands distributed throughout the body surface
[1] as an integral part of one of the largest organs—the skin
[2]. The eccrine sweat glands are the most numerous, spread over almost the entire surface of the body. They are responsible for the largest volume of the sweat secretion, which fulfills the thermoregulatory function of the body
[3]. The eccrine glands develop until the age of 2–3 years—when they become active, and their quantity stays constant throughout life. The development of apocrine sweat glands begins at birth, but they become active later during the stimulation by androgens or estrogens—associated with puberty. After a period of sexual maturity, their activity gradually decreases with age. The main concentration of apocrine glands is on the hairy parts of the body (in the axillae, mammary areolae, and periumbilical and genital areas). The armpit contains the most significant number of apocrine glands on the entire surface of human skin
[2]. Although a mixed-type apoeccrine gland has been described
[4], other authors have not confirmed its presence
[5]. Therefore, only two types of sweat glands are usually mentioned in the literature.
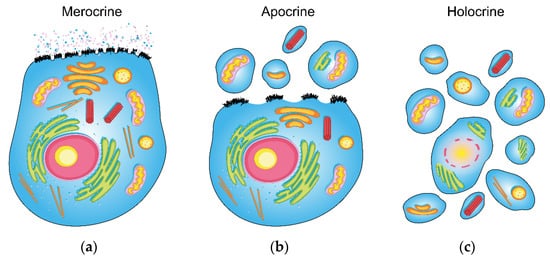
To characterize sweat glands by their secretory mechanism, they can be divided into three main groups: holocrine, merocrine (also called eccrine) and apocrine (
Figure 1). (1) The holocrine secretion is defined as a complete disintegration of a cell into a secretion (
Figure 1c), which is typical for sebaceous glands. They are also commonly found near the hair follicles and other sweat glands. However, they cannot be considered sweat glands, as they form the lipid-rich secretion by breaking its whole cell down. On the other hand, the eccrine and apocrine glands secrete in the true sense of the word, and do not undergo complete disintegration. (2) Merocrine secretion is based on the formation of vacuoles from Golgi’s apparatus and the cell membrane. Formed vacuoles and secretory granules, containing mainly water-soluble metabolites, ions and proteins, leave the cell via the process of exocytosis without disrupting it (
Figure 1b). (3) Apocrine secretion is characterized by separating a part of the cell containing cytoplasm and cell membrane (
Figure 1a). Unlike holocrine secretion, which occurs after death and the breakdown of the entire cell, apocrine secretion is formed from a living cell. It delivers part of the cell’s cytoplasm and cell membrane, containing intracellular fragments, to the outlet of the apocrine gland
[6]. The released part of the cell grows back, and the cell continues to function. Therefore, its utility lies in the advantage of apocrine secretion that reflects the intracellular space’s metabolic ratios. On the other hand, routine clinical materials such as urine or blood plasma (or serum) mainly reflect the metabolic situation in the extracellular space, and hence only partially and indirectly the intracellular and membrane metabolic processes
[6]. Although the mechanism of apocrine sweat formation and function has been studied for a long time, it has not yet been completely elucidated
[7].
Figure 1. Schematic visualization of the three main types of sweat secretion (the design was adopted from Graphics RF,
www.vecteezy.com, accessed on 1 July 2021). The general epithelial cell (with apical membrane) was chosen for the demonstration of: (
a) merocrine, (
b) apocrine and (
c) holocrine secretion (a more detailed description of the mechanisms is provided in the introduction).
The easier acquisition of eccrine sweat, which can be supported thermally or by physical activity, led to its better chemical characterization than apocrine sweat. Still, due to compositional similarities, both will be discussed together. The sweat consists of three main parts: (1) water which makes up the majority of sweat (almost 99%); (2) ions—Na
+, Cl
−, Ca
2+, K
+, Mg
2+, PO
43−, NH
4+ and others, including ionic species, and (3) organic molecules—amino acids, hydroxy/keto acids, free fatty acids, saccharides, urea, creatinine, uric acid, vitamins, volatile compounds and intact/metabolized exogenous chemicals and pharmaceuticals
[8]. Sweat is also often attributed to the function of removing metabolic wastes through perspiration, but so far, this function seems to be relatively negligible and mostly reserved for the kidneys
[9]. However, it appears that various exogenous substances and toxins such as pharmaceuticals
[10], drugs
[11], toxic metals
[12] or other organic substances
[13] are transferred into a sweat (and the concentrations in sweat are often higher than that of blood and/or urine), which could be used for monitoring or screening purposes.
Additionally, apocrine sweat represents the chemical nature of body odor, caused by the microbial (mainly the
Corynebacterium genus) transformation of steroid molecules, volatile and other organic fatty acids and lipids secreted on the skin surface by sweat glands
[14]. This makes apocrine sweat a sensitive indicator for specific odor modulating substances, e.g., in some spices, and it is also possible to observe these compounds metabolically transformed
[15]. To clarify the composition and origin of the apocrine secretion (not only sweat but also, for example, saliva, tears, or milk), various proteomic analyses were performed, describing cytoskeletal, membranous, microsomal, ribosomal, mitochondrial, and even, nuclear and nucleolar proteins in these biofluids
[6][16]. In terms of similarity of the proteomic composition of biofluids produced by apocrine glands (milk, sweat, tears, cerumen, saliva and cerebrospinal fluid), apocrine fluids contain between 30 to 65% identical proteins regardless of their anatomical origin
[6]. Furthermore, the proteomes of all the human apocrine fluids compared to plasma or serum revealed that 38 (cerebrospinal fluid) to 91% (milk) of the entries were identical to plasma proteins
[17]. This fact points to a link between the apocrine secretion (on the surface of the body) and the plasma, representing the situation in the blood circulation.
The sampling of apocrine sweat possesses a challenge as the secretion volume is very small, equivalent to a few microliters in one sample taken
[18]. Nevertheless, under certain circumstances (heat, exercise, influence of pain, topical application), apocrine sweat is diluted by eccrine sweat. Among other properties, apocrine sweat differs from eccrine in its pH. Apocrine sweat (pH 5.0–6.5) has an average pH approximately 0.5 units higher than eccrine sweat (pH 4.0–6.0). Using the pH value, the ratio between apocrine and eccrine sweat in the sample can be approximately estimated
[18]. Another technique to distinguish between the two types of sweat is fluorescence under ultraviolet light, as only apocrine sweat shows fluorescence
[18]. A reliable way to collect apocrine sweat, which is not diluted by eccrine sweat, is to expel the contents of the apocrine glands using external compression, vacuum or capillary forces
[4][18]. These techniques produce extra apocrine sweat without significant contamination of the eccrine component (if the subject’s physiology is not further influenced by heat and/or exercise). A major issue in the collection of apocrine sweat from armpit apocrine glands is the frequent use of antiperspirants (which leads to obstruction of the apocrine gland outlets, as well as damage to the apocrine gland itself) and difficult reproducibility of sampling (which is caused by an individual amount of secretion and a non-specific volume of the sample taken)
[19].
Future trends in clinical diagnostics are moving toward small sample volumes, high sensitivity of analytical methods, non-invasive collection techniques and a shift toward alternative biofluids. Although apocrine sweat is not yet used as a routine clinical material, it offers potential applications in screening (drugs, pharmaceuticals), monitoring (diseases, therapeutic drugs) or diagnostics
[20][21]. For clinical applications, apocrine sweat has the following advantages and significance:
-
It is a secretion that reflects the contents of the cellular cytoplasm (including cellular fragments).
-
It reflects the components contained in the cell membrane of apocrine gland secretory cells.
-
The components correspond to the cytoplasm composition and the cell membrane of a living cell (as opposed to a holocrine secretion, which is composed of the remnants of dead cells).
-
The lipid nature of apocrine secretion may be used in the future alongside diagnostic targets to identify/quantify lipid xenobiotics, lipophilic pharmaceuticals, and lipophilic narcotic drugs.
Apocrine sweat is still an incompletely studied biological fluid in metabolic and lipid composition, as comprehensive studies are lacking. The future use of apocrine sweat in clinical practice will become apparent when more studies comprehensively characterize this material. However, it requires the availability of highly sensitive microscale analytical techniques as only a few microliters of a sample can be acquired. The current study aimed to develop a robust and straightforward apocrine sweat sampling technique and verify the reproducibility of such a procedure. The sampling technique was evaluated by determining the lipidomic profile in healthy subjects following a standard sampling protocol. Subsequently, the data obtained were statistically evaluated further to understand the variability and behavior of apocrine sweat.
2. Novel Approach for Sweat Sampling
In the study presented here, we focused on characterizing apocrine sweat as a potentially clinically relevant biofluid, from multiple points of view. First, we have developed and described a simple, easy-to-use sampling technique, using a custom 3D-printed attachment and a commercially available porous glass filter disk. This concept not only offers an opportunity for other researchers and clinicians to adopt our technique for further studies, but it also brings a unifying aspect to apocrine sweat sampling, as the 3D blueprint can be easily shared and modified for further development. We have also described a standardized protocol with recommendations for consistent sampling. The usage of antiperspirants and other cosmetics with underarm application needs to be avoided before the sampling of apocrine sweat as it can lead to obstruction of apocrine sweat glands
[19]. Due to the physiology of the human secretory glands, it is not possible to obtain pure apocrine sweat because in the axillary region, in addition to the high density of apocrine glands, eccrine glands and sebaceous glands (in the hair follicles) are also present
[1]. It should also be noted that participants’ physiology was not altered by heat or physical activity (stimulating sweat production in other glands), and therefore an assumption was taken that the majority of sampled material is of apocrine gland origin and minor parts are of eccrine, sebaceous, or even microbial sources. To avoid this contamination as much as possible, the armpit was carefully cleaned with distilled water prior to the sampling. A perfectly adequate name for the material studied in this work is therefore armpit sweat (the main component of which is apocrine sweat). With defined and optimized the sampling technique, we proceeded to a detailed description of the material itself.
2. Lipidomic Methodology
Lipidomic analysis is increasingly showing its potential in the field of clinical diagnostics as well as offering unique insights into the biochemistry and biophysics of biological fluids and tissues
[22]. Apocrine sweat is an ideal material for lipidomic analysis as it contains not only intracellular metabolites but also lipid bilayers, which are released by apocrine glands
[6]. The absolute quantitative LC-MS analysis of lipids can be achieved only using HILIC separation, where all lipids are separated based on their polar head groups and not the side chains. Such mechanism is providing identical ionization conditions and matrix effects for each lipid class
[23]. On the other hand, the complementary reversed-phase mode (RP) provides separation based on acyl-chain length and number of double bonds, which offers a resolution of isomeric lipids. Moreover, using lipid pattern plots allows easy deciphering of misidentifications. Recently, it has been shown that RP and HILIC-based LC-MS analyses of lipids yield equal quantitative results for many lipid classes
[24]. As our goal was to comprehensively describe the lipid composition of the sampled sweat, as well as to quantify these lipids, the RPLC system was used. However, it needs to be addressed that for absolute quantitation multiple lipid standards within each lipid class would have to be used.
3. Data Processing Solution to the Problem of the Variability
Descriptive statistical evaluation of the studied material is necessary for its use in clinical applications. As sweat had already been shown to be a highly variable material as regards water content and concentration of ions
[1][20][25] and considering the fact that our approach did not account for differences in the volume of the sample, a robust normalization strategy had to be chosen. For the analysis of urine, the creatinine concentration is commonly used as a normalization parameter of the dilution, but it still does not solve all problems when performing metabolomic urinary analysis
[26]. The Probabilistic Quotient normalization (PQN) offers a solution to the dilution-induced sample inconsistencies as it transforms the metabolomics data according to an overall estimation of the most probable dilution
[27]. Although this normalization was first applied to NMR data, it is now becoming increasingly popular in metabolomics and lipidomics as it outperforms other normalization strategies
[28]. Our goal was to obtain data of as low non-biologically induced variability as possible while at the same time preserving information about individual biological variability. Finally, the application of lnPQN transformation was able to reduce CVi and CVg to approximately half that using natural logarithm alone, while the variability of QC samples remained unchanged, indicating the selectivity of the lnPQN approach in avoiding an overestimated normalization of the data.
The general principle and usefulness of the lnPQN were additionally proven by multivariate statistical analysis, particularly in the hierarchical cluster analysis, where 9 out of 10 individual participants were clustered, while an effect of the different day or side of sampling was not seen. Further insight into biological variability was offered by the results from PCA, in which all individual samples clustered close together, but 3 participants (C, I, F) lay further away from all other samples. This outcome highlights one imperfection of our study, namely the uneven representation of ages in our small cohort. These results are also in compliance with larger cohort studies investigating correlations between plasma lipids, age, and sex of participants, where intra-/interclass trends in lipids showed a good correlation with age
[29].
4. Description of the Sweat Lipidome
In terms of the lipid concentration in apocrine sweat, FA dominated, followed by glycerolipids (TG and DG) and then CE and phospholipids (SP and GP). Takemura et al.
[30] discovered that the technique of sweat collection significantly affects its composition, and that the FA content of physically scraped sweat can be up to 100-fold higher than that of clean sweat (collected passively without scraping). As far as we know this is the first work comprehensively describing the apocrine sweat lipidome including estimation of lipid concentrations. We compared our results with those from other studies of sweat from various origins. Peter et al.
[31] subjected mechanically collected eccrine sweat to quantitative FA analysis and found that FA concentration oscillated between 0.1–5.47 μg/mL, which is approximately one order of magnitude below that in our measurements (in which FA ranged from 0.03–44.4 μg/mL). The difference can be explained by the different biological origin (eccrine gland) of the sweat in the aforementioned study and the higher extraction efficiency of our sampling procedure (due to pre-soaking the disk in 80% IPA). The high concentration of lipids (specifically FA) is consistent with the assumption that apocrine sweat is a lipid-rich material and, unlike eccrine sweat, cannot be completely evaporated
[18]. As the release of apocrine sweat is promoted by physical stimulation, we chose the scraping approach for its collection, which can lead to an increased content of FA in the final sample, similarly to the presence of sterols and ceramides of sebaceous and epidermal origin. The heterogeneous lipid composition of the sample was also reflected in the number of individual lipids (in parentheses), where Cer (
n = 61) were the most represented, followed by TG (
n = 37), SM (
n = 23), PC (
n = 20), DG (
n = 17) and FA (
n = 17). At first glance the large number of ceramides of structurally heterogeneous character (various long-chain bases: 16:0, 16:1, 18:0, 18:1, 20:1) may be striking; however, they are found abundantly as constituents of the cutaneous lipidome, where their function (water permeability barrier of the skin) and their role in pathobiochemical processes (e.g., atopic dermatitis or psoriasis caused by disruption of the permeability function or inflammatory disbalance of lipids in the epidermis) have been well documented
[32][33][34].
To understand the biochemistry and biological behavior of a particular material, it is helpful to focus on the trends within and between groups of studied biomolecules in a particular biofluid or tissue. By constructing a correlation map, we could easily trace the relationship trends between lipid classes. We could also observe that one lipid class generally behaves in a consistent manner compared to another class, which is commonly found in studies of plasma lipidome
[35]. Glycerolipids (TG and DG) in apocrine sweat did not show a similar correlation pattern and were frequently showing opposite trends compared to each other. In contrast, in plasma these classes usually correlate in a similar pattern together
[36]. Interestingly, all glycerophospholipids showed similar trends in correlations between and within lipid classes (except for the correlation of PS and PI to PEO). On the other hand, in common with other researchers, we have also found correlation trends in the classes themselves and in some cases even individual lipids with opposite behavior to the rest of the class (indicating different origin, function, or regulation)
[36].
To fully describe the studied material, differences between the sides of sampling were also evaluated. Although systematic trends in lipid class levels were observed between the left and right sides of sampling, these changes were considered nonsignificant after the application of
p-value correction (Benjamini-Hochberg). Furthermore, since these differences for the majority of lipids were only up to +/−15%, this contribution was minor compared to the interindividual variability of individual lipid classes (which are mostly greater than 50%). On the other hand, these differences could be of significant importance for diseases affecting only one of an organ pair (e.g., breast cancer), or they could also reflect the physiological dominance of the right or left hand and/or lymph nodes
[37]. However, further studies will be needed to clarify these hypotheses.
5. Future Development
The results presented above show the potential of apocrine sweat sampling and its analysis by advanced mass spectrometry coupled with liquid chromatography, as a promising noninvasive alternative to established methods in routine clinical analysis. Based on the results of our research and also from other studies, it is likely that new unsuspected applications for various microsampling techniques will be emerging for both diagnostic and therapeutic purposes
[38]. Given the large number of different techniques already described for the collection of apocrine sweat (which usually involve stimulation by heat, physical activity, pain, or the intradermal injection of adrenaline), it would be useful to compare them with each other and with the SLIDE technique in the following studies. Large-scale studies will be necessary to define the influence of age, gender, BMI, and other factors on the lipidome of apocrine sweat. Due to the low cost of the sampling method itself, the potential future application can be also seen in screening programs. This study was conducted to improve characterization of the apocrine sweat lipidome, as other pilot experiments have already shown the usefulness of sweat analysis for (1) drug screening
[11], (2) detection of early epidermal immunomodulation disruption in atopic dermatitis
[33], (3) glucose monitoring in diabetic patients
[39] and (4) detection and measurement of pharmaceuticals
[10]. This work was carried out because a reliable sampling technique and analysis of the composition of apocrine sweat in healthy individuals is a necessary basis for the subsequent use of this biofluid in modern clinical diagnostics.