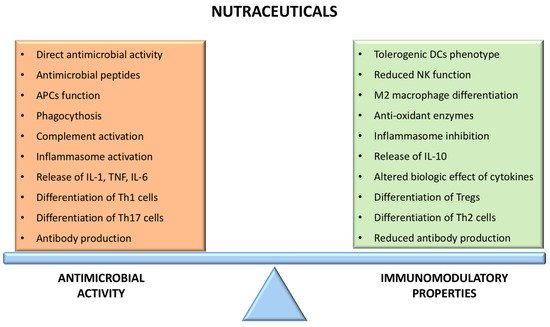
The antiviral effects of lactoferrin depend on the inhibition of different phases of the infectious cycle. Indeed, it can interfere with the binding of viral pathogens with heparin sulfate glycosaminoglycan molecules on the cellular surface, thus blocking the interaction between the virus and its receptor and its entry into the host cell
[57][60]. In vitro studies performed on different pathogens (influenza A virus, parainfluenza virus, RSV, and others) evidenced that lactoferrin inhibits viral replication, with an effect that is more pronounced when adequate cellular zinc levels are present
[9][57]. Additionally, lactoferrin has complex immunostimulating properties that promote viral clearance, enhancing both innate and adaptive responses
[65]. The effect of lactoferrin on the different cellular targets is mediated by its interaction with cellular receptors (surface peptidoglycans and glycosaminoglycans, TLRs, and others), which promote its internalization. Following this, lactoferrin can act through the modulation of signaling pathways (interacting with kinases and phosphatases) and transcription factors
[62]. Concerning the innate response, lactoferrin acts as a chemotactic factor for leukocytes, enhancing cellular activity, cytokine release (particularly IFN), and function of NK cells (thus promoting phagocytosis), neutrophils, and macrophages, and participates in reinforcing and maintaining the integrity of the gut mucosal barrier
[61][66]. Different studies have shown that lactoferrin accelerates the maturation of enterocytes (mainly through the activation of MAP kinase (MAPK)-dependent molecular pathways), thus reducing the permeability of the mucosal barrier and the systemic spreading of infectious agents, and promotes the development of the gut microbiome
[65][67]. This latter effect is of particular relevance in newborns, since the microbiome prevents gut colonization by pathogens
[66] and has a direct local and systemic immunomodulatory role through the stimulation of the release of IL-10
[68] and the downregulation of Th1- and Th17-dependent cytokines
[69]. The action on innate immunity is also partly responsible for the effects of lactoferrin on the adaptive immune response. Indeed, lactoferrin stimulates the maturation and activation of DCs and macrophages and enhances their antigen-presenting function and secretion of cytokines (such as TNF-α, IL-8, and IL-12), finally promoting a Th1-mediated immune response
[70]. Additionally, lactoferrin directly influences lymphocyte maturation and proliferation, as different experimental studies showed that lactoferrin enhances the maturation of T helper lymphocytes and B lymphocytes and their antigen-presentation function as well as the production of immunoglobulin, both at the mucosal level and systemically
[65][71][72]. Activation of MAPK pathways contributes to the promoting effect of lactoferrin on T cell maturation, while the enhanced release of IL-12 is important in driving Th1 differentiation
[65][73].