1000/1000
Hot
Most Recent

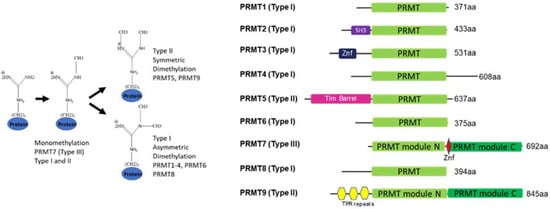
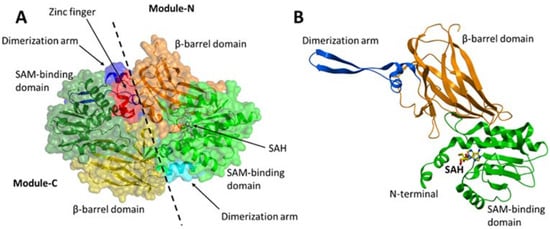
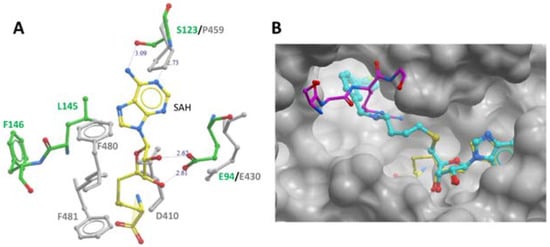
PRMT7 is a member of the protein arginine methyltransferase (PRMT) family, which methylates a diverse set of substrates. Arginine methylation as a posttranslational modification regulates protein–protein and protein–nucleic acid interactions, and as such, has been implicated in various biological functions. PRMT7 is a unique, evolutionarily conserved PRMT family member that catalyzes the mono-methylation of arginine. The structural features, functional aspects, and compounds that inhibit PRMT7 are discussed here. Several studies have identified physiological substrates of PRMT7 and investigated the substrate methylation outcomes which link PRMT7 activity to the stress response and RNA biology. PRMT7-driven substrate methylation further leads to the biological outcomes of gene expression regulation, cell stemness, stress response, and cancer-associated phenotypes such as cell migration. Furthermore, organismal level phenotypes of PRMT7 deficiency have uncovered roles in muscle cell physiology, B cell biology, immunity, and brain function.

| Substrate | R Methylation Sites | Function, Disease Relevance, Reference |
|---|---|---|
| DVL3 | 271, 342, 614 | DVL3 localization, wnt signaling, cancer [30] |
| EIF2S1 (EIF2 alpha) | 55 | Translation arrest, stress granule regulation, [31] |
| G3BP2 | 432, 438, 452, 468 | Wnt signaling, cancer, [32] |
| GLI2 | 225/227 | Cell senescence, [33] |
| Histone H4, H2A | H4R3, H2AR3 | Gene expression, [23][24][25][26][27] |
| HNRNPA1 | 194, 206, 218, 225 | Splicing, [28] |
| HSP70 | 469 | Stress response, [13] |
| NALCN | 1653 | Neuronal excitability, [34] |
| MAVS | 52 | Viral infection, [35] |
| MRPS23 | 21 | Oxidative phosphorylation, cell invasion, cancer [36] |
| P38MAPK | 70 | Myoblast differentiation, [37] |
| PRMT7 | 531 | Cell migration, cancer [16] |