The ear and brain work together in the hearing process. Sound energy in the audible frequency range (20 Hz–20 kHz) propagates through air medium as an acoustical mechanical wave which enters from the outer ear towards the middle ear to vibrate the eardrum.
1. Introduction
The ear and brain work together in the hearing process. Sound energy in the audible frequency range (20 Hz–20 kHz) propagates through air medium as an acoustical mechanical wave which enters from the outer ear towards the middle ear to vibrate the eardrum. In a healthy cochlea, the eardrum vibrates the small bones called the ossicles (malleus, incus and stapes) and the sound vibrations travel to the cochlea which resides in the inner ear. The basilar membrane (BM) in the cochlea mechanically filters sound while the hair cells turn BM vibrations into electrical nerve impulses. In this mechanoelectrical transduction, BM vibrations displace hair cells (stereocilia) resulting in the gating of ion channels and generating bioelectrical potentials. The auditory nerves are stimulated and the brain would interpret the information received from the generated nerve impulses.
There are three main types of hearing loss that can either affect one or both ears. Conductive hearing loss occurs when the outer ear and middle ear could not become the transmission mediums to conduct sound properly. People with severe outer or middle ear malformation and chronic ear infection may experience conductive hearing loss. The sensorineural hearing loss happens due to missing or damaged hair cells in the inner ear that lead to frequency selectivity and sensitivity disorders. Mixed hearing loss is the combination of both conductive and sensorineural hearing losses, a result associated with issues related to both outer/middle ear and inner ear. Different types of hearing devices like cochlear implant (CI) and middle ear implant (MEI) have been developed to cater to different types of hearing problems. Hearing device usage significantly improves the social, psychological, emotional and physical aspects of life for hearing impaired persons.
CI is the most successful neural prosthesis to date with a mature technology in biomedical device implantation. It mainly treats patients with severe to profound sensorineural hearing loss.
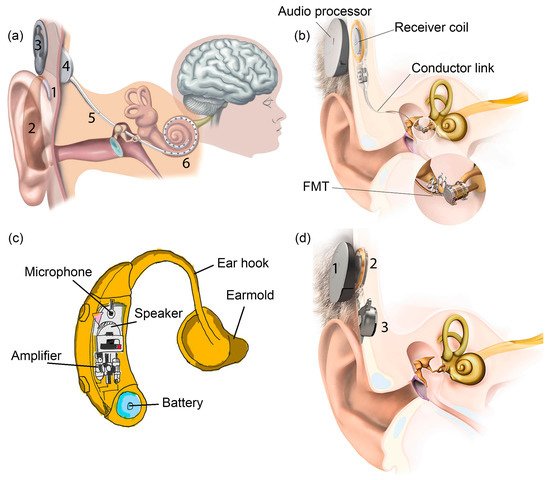
Figure 1a displays the externally worn audio processor (2) with a battery pack behind the ear and a microphone (1) hooked onto the ear
[1]. The microphone transduces acoustic waves from the environment into electrical signals before feeding them into the processor. Bank of filters in the processor decompose the electrical sound signals digitally into different frequency bands/components and implement certain specific processing strategies to extract the right features from the detected sound. The signals are encoded into radio frequency (RF) electrical signals. RF transmitter coil (3) is positioned in place and aligned to the RF receiver coil (4) by a magnet. The coded signals are transmitted to the receiver coil implanted under the skin from the transmitter coil via transcutaneous magnetic induction mechanism. The hermetically sealed implanted stimulator circuit (4) derives power from the RF signal and translates the coded signals into electrical pulses. A conductor link (5) threaded into the cochlea and the multichannel stimulation electrodes (6) at the end of the conductor link connect to the hearing nerves. The electrical pulses are transmitted from the stimulator to the stimulation electrode array inserted in the cochlea via the conductor link and directly stimulate the auditory nerves. All of these processes are supported by a battery, supplying each component of the CI with power
[1][2].
Figure 1. The schematic of (
a) cochlear implant (CI)
[1], (
b) middle ear implant (MEI), photo credit: MED-EL
[3], (
c) hearing aid and (
d) bone-anchored hearing aid (BAHA), photo credit: MED-EL
[4].
MEI has a similar arrangement like CI with the extracorporeal audio processor and surgically implanted receiver coil under the skin (
Figure 1b)
[3]. Magnetic attraction holds the audio processor directly over the implant. A microphone detects sound and converts it into electrical signals. The processor processes the electrical signals and transmits them to the implant before being relayed to the floating mass transducer (FMT) via a conductor link
[5]. FMT converts the electrical signals into mechanical vibrations that set the middle ear structures into motion and subsequently transfer the vibrations to the cochlea. At this stage, the cochlea may proceed with its natural functionalities by which the BM mechanically filters the sound vibration and the hair cells transform the vibration into bioelectrical potentials. There is a variety of types of MEI based on how FMT is attached in the middle ear. FMT can be clamped to the incus of the ossicular chain or inserted into the round window of the cochlea
[6]. The working operation of MEI is independent of skull growth and thus suitable to be implanted in children
[7]. MEI can aid people with a conductive, mixed or mild to severe sensorineural hearing loss.
Figure 1c shows a hearing aid which is basically an acoustic amplifier
[8]. The detected sound by microphone is digitally amplified by the audio processor. The increased sound volume is then transmitted to the ear canal via a customised earmold worn in the ear. The loudspeaker converts back the electrical signals into sound signals and delivers them to the ear drum in the middle ear. The ossicles and cochlea will naturally process the sound signals. Deaf patients with less than 70–80 dB of sensorineural hearing loss can use a hearing aid
[9].
Bone-anchored hearing aid (BAHA) is a hearing system based on bone conduction method, specifically designed for people with damaged outer or middle ear. It utilises the skull’s bones to conduct sound into the inner ear, facilitating hearing for people with a conductive or mixed hearing loss condition. In
Figure 1d, the externally worn microphone and audio processor (1) pick up sound and convert it into electrical signals
[4]. These signals are transmitted to the RF receiver coil (2) which has been surgically positioned transcutaneously under the skin around the temporal bone. An actuator/vibrator (3) transduces the electrical signals into mechanical vibrations and the signals are transmitted to the cochlea through the skull. The vibrations make the cochlea fluid move and generate nerve impulses
[10][11]. Adhesive bone conduction system is a newly developed non-surgical device where the users can simply stick an adhesive adapter onto the skin behind the ear
[12][13]. The audio processor clicks onto the adapter where it picks up sound and vibrates the adapter. The adapter then gently vibrates the bone behind the ear. The bone conducts the vibrations towards the cochlea. The system comfortably avoids pressure onto the skin and can be worn unnoticeably behind the ear.
A summary detail on the commercially available hearing systems is tabulated in
Table 1. HiRes Ultra 3D by Advanced Bionics is a CI system with 16 stimulation electrodes that can deliver up to 120 spectral bands of sound
[14]. Greater spectral resolution improves speech perception in noise and music appreciation. Nucleus
®Systems by Cochlear Americas possesses 22 stimulation electrodes that provide access to the full spectrum of sound and optimal hearing coverage. The recipients’ number of Nucleus
®Systems has grown to more than 400,000 worldwide
[15]. It is the first CI with removable magnet, allowing users to undergo magnetic resonance imaging (MRI) brain scans. Vibrant Soundbridge by MED-EL is a semi-implantable MEI that implemented mechanical stimulation using electromagnetically driven transducers
[16]. Fully implantable piezoelectric-based MEI, Envoy Esteem by Envoy Medical is widely used in the United States and Europe
[17]. Evoke by Widex is the first brand that incorporates machine learning into the hearing aid
[18]. The intelligent hearing aid is capable of learning from different situations and user’s inputs in order to evolve its own functions, so that high quality sound can be heard. More by Oticon is a recently developed hearing aid with internet-enabled and Bluetooth-enabled functions
[19]. Baha
®Attract is a BAHA system manufactured by Cochlear corporation
[11]. The vibrator is magnetically retained externally with the audio processor to the bone conductor. Soft pad is used to distribute pressure over the area of skin contact for magnetic attachment
[13]. On the other hand, BONEBRIDGE by MED-EL is a different BAHA system that has an implanted vibrator in the mastoid area to directly connect to the skull
[13][20]. The exterior processor is quite light due to the exclusion of the vibrator’s weight.
Table 1. Descriptions of the commercially available hearing systems.
| Hearing System |
Hearing Loss Condition |
System Operation |
Manufacturer |
| Cochlear implant (CI) |
Severe to profound sensorineural |
Bypass outer ear, middle ear and inner ear’s BM and hair cells. Direct stimulation of auditory nerves. |
-
HiRes Ultra 3D by Advanced Bionics [14]
-
Nucleus ®Systems by Cochlear Americas [15]
|
| Middle ear implant (MEI) |
Mild to severe sensorineural, conductive or mixed |
Bypass the outer ear. Transduce mechanical vibrations in the middle ear and transmit them to stimulate the cochlea. |
-
Vibrant Soundbridge by MED-EL [16]
-
Envoy Esteem by Envoy Medical [17]
|
| Hearing aid |
Mild to moderate sensorineural |
Amplify and transmit sound to the cochlea via ear canal and middle ear structures. |
|
Bone
-anchored hearing aid (BAHA) |
Mild sensorineural, conductive or mixed |
Bypass the outer ear and middle ear. Transmit sound to the cochlea via skull’s bones. |
-
Baha ®Attract by Cochlear Corp. [11]
-
BONEBRIDGE by MED-EL [20]
|
Even though there are huge technological improvements in these hearing devices with tremendous technical and clinical success, the number of hearing device adoption rate has not increased
[21]. More than 75% of hearing impaired population did not benefit from the advanced hearing devices technology, making hearing loss as the largest chronic sensory condition that remains untreated
[16]. The main reason for rejection of becoming a hearing device user is the stigma of wearing the device itself. Patients are concerned with society’s perspectives on the devices’ cosmetic appearance and wish for their deafness to be hidden. In addition, there are risks for the device to be damaged as the exterior parts can be subjected to trauma, humidity or dirty conditions. The magnetic fixation of the RF transceiver coils can cause problems in the skin tissue due to continuous pressure exercised by the antennas. Other limitations include the restriction from enjoying physical activities like swimming due to water exposure or extreme sports due to perspiration. Simple daily routines like showering and sleeping require the users to temporarily remove the external part
[7][16][21][22]. Many users desire the hearing devices to work without the exterior components on or off the ear with 24/7 operation. It has become a quest for researchers to invent a totally implantable hearing system that would make the device invisible.
Only a few fully implanted MEI systems have been reported and the semi-implanted MEIs have been found to have less performance problems compared to the fully implantable ones
[23]. Hearing aid is not an implant but an externally or internally worn device. Behind the ear (BTE) and receiver in the canal (RIC) are body-worn hearing aids where the loudspeaker is located behind the ear and in the earmold, respectively. In the ear (ITE) fits in the outer ear bowl while a completely in the canal (CIC) hearing aid is placed deep in the ear canal, barely visible. An invisible in canal (IIC) hearing aid is positioned completely inside the canal leaving no visible trace. It is not possible to have a fully implanted system for BAHA while for CI, many are still in the research phase and not yet commercialised. Recently, MED-EL announced the very first surgery of a fully implanted CI in Europe and will receive its market approval in the next several years
[24]. In this paper, we will look closely at the efforts done towards achieving the fully implanted systems and discuss the key challenges and limitations that impede the process of attaining one. The battery, microphone and processor from the externally worn compartment of the hearing systems are thought to be the three main issues that need to be addressed and discussed thoroughly. Therefore, this review article is structured into two sections: energy harvester and implantable microphone. A review of the low-powered speech processing strategies and the programmable processor circuit implementation for the hearing system’s processor unit will not be presented in this paper. For each section of the microphone sensor and energy harvester, comprehensive background information on the electromechanical energy sensing/harvesting and the available transduction mechanisms are discussed in depth. None of the recent reviews compile both sensor and energy harvester in one literature which focuses specifically on hearing system application.
Microfabrication technology offers device size reduction and compatibility with the silicon processes, allowing the microphone and energy harvester to easily integrate with the digital signal processing circuitries in one small chip that can fit into the ear. In the end, the choice of materials to be implemented is of utmost important. The microphone and energy harvester for fully implanted hearing systems are desired to operate efficiently, pose low health risks and be compatible with complementary metal-oxide-semiconductor (CMOS) processes. In vivo energy harvesters especially need to be flexible and stretchable. The combination of piezoelectric materials and polymers into a nanocomposite or a multi-layered thin film appeals as a promising structure for both energy harvesters and microphones. Thus, in this paper, we will mainly focus on the sensing and energy harvesting of polymer-based piezoelectric transducers for totally implanted hearing systems. The design principles of the piezoelectric/polymeric devices are carefully examined, which include the materials development, configuration structure and fabrication, operation modes, packaging strategies and considerations of biocompatibility.
2. Energy Harvester for Totally Implanted Hearing Device System
Generally, an implanted medical device is used for sensing or stimulating by which it serves as a diagnostic tool or is used for treatment practices. A patient who is suffering from neural system disorder uses implantable stimulators
[25], Parkinson’s symptoms can be eased via deep brain stimulators
[26] and a hearing impaired person uses a cochlear implant for hearing restoration
[27]. These medical implants, including cardiac pacemakers, retinal implants and infusion pumps, are considered small-sized and low-powered electronic devices. The electrochemical battery to power up these devices needs periodic recharging or has a limited lifetime that requires replacement. The implanted lithium battery for a pacemaker, neurostimulator and insulin infusion pump needs to be replaced every ~6.5 years, ~4 years and ~2 years on average, respectively
[28][29]. The implanted battery for a MEI can stay for ~6 years while an external worn battery has to be charged every week
[5][30]. Higher energy usage will reduce the lifespan of an implant’s battery and its small energy storage requires frequent charging. The removal and replacement of an implanted battery requires surgery that could pose risks to the well-being and health of the patients. The risks of infection, bleeding, inflammation, long healing process and additional cost have called for the development of self-powered medical implants to reduce these physical, psychological and financial burdens on users. Self-sustainable energy generation may perhaps ensure the longevity of the implanted medical devices.
2.1. Energy Harvesting Technologies for Implanted Medical Devices
Energy harvesting can extend the working life of electronics. Battery contributes an unwanted weight and volume in a biomedical system. A battery that can last longer presages an increase of size and weight of the implanted devices. Therefore, an energy harvester is essential in the manufacturing of medical implants. There are various accessible energy sources like solar, heat, electromagnetic radiation, ultrasonic waves, fluid flow and vibration which could be used to generate electrical signals. From these sources, there are different types of transduction mechanisms that can be implemented in implanted biomedical devices like piezoelectricity (mechanical strain), electromagnetism (magnetic induction), electrostaticity (capacitance), thermoelectricity (temperature gradient), triboelectricity (frictional contact and electrostaticity) and pyroelectricity (thermal fluctuation). Two energy harvesting concepts are coined, either from the environmental source or human body.
Energy sources from the outside of a human body can be utilised by the implanted energy harvesters to generate electrical energy. Wireless energy transfer is a reliable and convenient through-skin energy transmission method to charge up the implanted biomedical device or energy storage device without additional surgery
[25]. Reported here are few examples of implantable wireless energy harvesters that a hearing device could adopt for its own fully implanted system. In
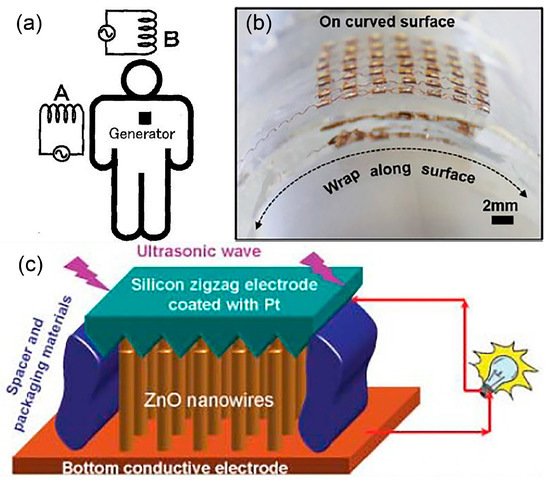
[31], electromagnetic induction has been used to power up a pacemaker. A human body is proposed to be placed in a low frequency rotating magnetic field that triggers the implanted microgenerator (
Figure 2a). Two phase excitation coils are set up outside of a human body while the microgenerator, metal magnet and high-ratio gear are implanted inside. A rotating magnetic field is applied onto the implanted magnet causing it to rotate. High-ratio gear increases magnet rotation and drives the microgenerator to generate high voltage. The system produces small eddy current due to low frequency excitation and thus it is safe to be implemented in a human. They estimate that 4 W of power is needed to be generated from the system to fully charge a pacemaker for 3 h.
Figure 2. (
a) Electric power generating system for implanted medical devices is proposed by positioning a human body in a rotating magnetic field. © 1999 IEEE. Reprinted, with permission, from
[31]. (
b) Optical image of the flexible piezoelectric transducers array for ultrasonic energy harvesting. Reprinted from
[32], Copyright (2019), with permission from Elsevier. (
c) A schematic diagram of a nanowire piezoelectric nanogenerator driven by an ultrasonic wave
[33].
In
[32], the wireless energy transfer employs an acoustic energy to supply power for the implanted medical devices. A flexible piezoelectric transducers array is developed (
Figure 2b) for ultrasonic energy harvesting. It generates continuous ~2 peak-to-peak voltage (Vpp) output and ~4 µA current under ultrasonic excitation, when implanted on both planar and curved surfaces. The developed flexible device demonstrated auspicious performance with weak attenuation on curved surfaces. The proposed design solved the bulky and rigid issues of other ultrasonic energy harvesters that restrict them from attaching to soft and curvy surfaces. Wang et al. have also driven an implanted piezoelectric-based nanogenerator device using ultrasound that can produce continuous direct current (DC) output for an implanted device
[33]. In
Figure 2c, the nanogenerator is made of vertically aligned zinc oxide (ZnO) nanowires grown on a polymer substrate and covered by a zigzag electrode.
Semi-implanted hearing systems, however, have already adopted the wireless energy transfer method via inductive coupling based on the electromagnetic field between the RF transmitter and receiver coils. The coils have a dual function of delivering power and auditory information, simultaneously. The external battery with rechargeable characteristics continuously supplies power to the internal implant coil wirelessly. For a cochlear implant, a small implant coil is adequate with a power requirement of ~20 mW
[25]. For a totally implanted hearing system, it is impractical to develop an implanted wireless energy harvester that harvests energy from the external sources. It is a hassle for the user to stay inside the magnetic field or ultrasound wave set up for the hearing device to be continuously powered and operated, unless there is also an implanted energy storage device which will then require periodic recharging
[34]. In addition, there might be restrictions or health and safety concerns on the users with implanted electromagnetic-based wireless energy harvester to undergo head MRI. Also, long term magnetic radiation exposure might cause decrease in fertility, muscle stiffness with loss of protein and neuroendocrine system degradation due to change in DNA, while electric fields may cause unnecessary stimulation of muscles, nerves and sensory organs
[25].
The implanted medical device or energy storage device can be charged internally, directly, continuously and unconsciously by the energy harvested from human body. In vivo implantable energy harvesters are aimed at powering electronic devices embedded inside the human through the body’s thermal gradient or internal biomechanical motion like lung inflation, stomach deformation, cardiac motion, muscle contraction/relaxation or blood flow. A human body at rest consumes ~100 W to maintain internal organs, tissues and cells functioning while it produces ~81 W when asleep and 1630 W during sprint walk
[28].
Heartbeat vibrations can generate and supply power to pacemakers or sensors that stimulate heart muscles, regulate its contraction and monitor vital signs like pulse rate or blood pressure. In
[35][36], fan-folded piezoelectric beam stacked structure uses heartbeat to generate more than 10 µW to power up a lead-free pacemaker. Fan-folded design is chosen in order to utilise three-dimensional space to the energy harvester and the added tip mass and link mass help to reduce the high natural frequency of the energy harvester. Small natural frequency of the device (16.18 Hz) generates more power. Less than 1 µW is needed for a pacemaker and the proposed energy harvester of size 2 cm × 0.5 cm × 1 cm generates sufficient energy for the medical implant. No magnet is incorporated into the proposed device and thus it is MRI compatible. Dong et al. developed a cardiac energy harvester using a porous piezoelectric helical thin film structure that transforms the mechanical energy from a pacemaker’s lead into electrical signals
[37]. This strategy allows the battery of a cardiac pacemaker to be charged without the energy harvester coming into contact with the heart and thus interferes with its function. The bioinspired self-wrapping helical configuration of the energy harvester allows flexible integration with the lead of the pacemaker and the bending motion of the energy harvester generates ~0.6 Vpp output. A 10 × 10 array of helical devices wrapping all through the lead is estimated to extend the pacemaker’s battery lifetime by 1.5 years. Kim et al. developed a self-powered cardiac sensor from a flexible piezoelectric transducer that can generate 17.8 V and 1.74 μA from the contraction and relaxation of a porcine heart
[38]. In
[39], an implantable blood pressure sensor in aorta could monitor high blood pressure using the aorta’s pulsating energy in piezoelectric generator film, producing a maximum output voltage, current and power of 10.3 V, 400 nA and 631 nW, respectively.
Other than heartbeat, the temperature difference between the skin and body core can be used to generate electricity and charge the battery of a pacemaker. Thermoelectric energy harvester converts temperature gradients into electrical energy and such a small temperature difference could provide high power output of more than 100 µW
[28]. Cardiac pacemaker does not require constant battery loading and consumes low energy where 10 µW can sufficiently power up the device
[28][35][40]. The biggest consideration of implementing a thermoelectric energy harvester in an implanted medical device is the biocompatibility of the materials used. Bismuth telluride is a common thermoelectric material that is moderately toxic but can be fatal in large quantity
[28].
A brain pacemaker is a neurosurgical treatment which stimulates certain area in the brain via implanted electrodes that generate continuous electrical impulses. Deep brain stimulation helps to relieve neurologic and psychiatric disorders like epilepsy and Parkinson’s disease. This neurostimulator consumes more energy compared to a cardiac pacemaker. A flexible piezoelectric thin film energy harvester on plastic substrate by Hwang et al. can be utilised as a self-powered deep brain stimulator using slight movement of human muscles or organs
[41]. The fabricated energy harvester yielded a maximum output current and voltage of 0.57 mA and 11 V, respectively. The energy harvester manages to stimulate a specific target area of a mouse brain that instantaneously induces its forearm movement. In
[42], Fan et al. have proposed an energy harvesting system utilising the deformation of a piezoelectric material attached onto the human mandible to power up the implanted deep brain stimulator. The piezoelectric energy harvester was mounted onto a synthetic mandible and a novel mandibular loading set up was developed to copy the human mastication forces; 1 Vpp output was measured and the improved device performance could power up a commercial deep brain simulator in the future.
Depending on the stimulation strategies, the average total power consumption for a CI can be around 10 mW. Žák et al. proposed a multidisciplinary ambient energy harvesting system from the combination of thermal gradient, mechanical movement (shocks) and bending movement of neck muscles or arteries in the head area for autonomous powering in a totally implanted CI
[43]. For energy harvesting from the mechanical movement, they have suggested the electromagnetic-based electromechanical conversion principle, utilising resonance at the excited mechanical movement/vibration frequency. Although human muscles and skeleton cause a damping effect, it can be neglected for small weight of proof mass. The electromagnetic energy harvester consists of the flexible cantilever structure with seismic mass (permanent magnet) which moves against a fixed printed coil that induces voltage. At 18 Hz, a maximum output voltage and power of ~1.4 Vpp and ~1.8 mW have been simulated, respectively, during walking in the time range of 10 s. As for the thermoelectric energy conversion, the waste heat produced in the region of human head (~15 W) is converted into useful electrical energy via the Seebeck effect. Thermal gradient around 5 °C is desired with the thermal resistance of human tissue between body core and skin surface variying with respect to physical activities, age and health conditions. Thermoelectric module with 100 mm
2 area is able to provide more than 300 µW of power. Additionally, the electrochemical gradient in the inner ear’s fluid can be used as an energy harvesting source. The difference in ionic concentration between the endolymph and perilymph fluid induces endocochlear potential. In
[44], an endoelectronic chip is positioned in the middle ear between two electrodes that connect to the endolymph and perilymph. The energy harvester extracts a minimum of 1.12 nW from the endocochlear potential in guinea pigs continuously for 5 h.
Table 2 summarises the various in vivo implantable energy harvesters for powering the implanted medical devices.
Table 2. Summary of in vivo energy harvesters for implanted medical devices.
| Energy Source |
Transduction Mechanism |
Device
Application |
Input Excitation |
Output
Performance |
| Mechanical |
Piezoelectricity |
Lead-free cardiac pacemaker |
Heartbeat vibrations |
10 µW [35][36] |
| Mechanical |
Piezoelectricity |
Cardiac pacemaker |
Pacemaker’s lead/heart motion |
0.6 Vpp [37] |
| Mechanical |
Piezoelectricity |
Cardiac sensor |
porcine heart motion |
17.8 V and 1.74 μA [38] |
| Mechanical |
Piezoelectricity |
Blood pressure sensor |
aorta’s pulsating motion |
10.3 V, 400 nA and 631 nW [39] |
| Heat |
Thermoelectricity |
Cardiac pacemaker |
Temperature gradient between skin and body core |
100 µW [28] |
| Mechanical |
Piezoelectricity |
Brain pacemaker |
Muscles/organ motion |
0.57 mA and 11 V [41] |
| Mechanical |
Piezoelectricity |
Brain pacemaker |
Human mandible motion |
1 Vpp [42] |
| Mechanical and heat |
Electromagnetism and thermoelectricity |
Cochlear implant |
neck muscles/arteries motion (electromagnetism) and temperature gradient between skin and body core (thermoelectricity) |
~1.4 Vpp and ~1.8 mW (electromagnetism) and 300 µW (thermoelectricity) [43] |
| Electrochemical gradient |
Electrochemistry |
Cochlear implant |
Ionic concentration gradient in inner ear’s fluid |
1.12 nW [44] |
In the search of materials and methods for creating an energy harvester that supports the development of a fully implanted hearing system, the mechanical energy appeals as the most omnipresent energy source available from the ambient environment surrounding the hearing device. The mechanical energy is relevant because it is closely related to the hearing process and can be easily captured before being converted into useful electrical power via mechanoelectrical transduction. The in vivo energy harvesting through human organ motion can supply clean power for the implanted medical device without affecting the organ or its functions. As the electronic device shrinks, so does the energy consumption. Piezoelectric transduction mechanism transduces mechanical vibrations into microwatts and milliwatts of power level and the approach is suited for in vivo energy harvesting in cochlear devices. Therefore, piezoelectric energy harvesting (PEH) can generate sufficient electrical output to meet the requirement of a totally implanted device.