2. Translating Personalized Medicine into Clinical Practice: The Andalusian Experience
Andalusia, located in the south of Spain and with 8.5 million inhabitants, is the third most populated region in Europe, and it is larger than half of the countries of the European Union. Remarkably, Andalusia has the whole population under a unique universal electronic health record, thus forming the largest resource of this kind in the European Union. Under this scenario, all the decisions and strategies taken around this huge clinical database acquire enormous relevance. Since 2001, the data recorded by the Andalusian Public Health System (SSPA) are systematically uploaded to the Population Health Base (BPS), making it one of the largest repositories of highly detailed clinical data in the world (with over 13 million registries)
[3]. BPS constitutes a unique and privileged environment to carry out large-scale RWE studies. Actually, one of the BPS missions is facilitating the discovery of new biomedical knowledge by means secondary use of clinical data
[4], paying special attention to the evaluation of impact in personal data protection
[5].
The Andalusian SARS-CoV-2 genomic surveillance project
[6] set the ground for the implementation of a clinical circuit for controlling the COVID-19 pandemic as well as other potential future emergent viruses. This project engaged the 16 main tertiary hospitals in Andalusia, along with three research centers with genome sequencing facilities (IBIS, Genyo and CABIMER) and the Bioinformatics Area of the Progress and Health Foundation in a circuit of genomic data production. In parallel, the COVID-19 registries from Public Health and the BPS provide ongoing and retrospective clinical data, respectively, to the Bioinformatics Area, where the data are linked to the genomic data in a circuit of genomic data interpretation. Bioinformatics then provide (i) to the microbiologists at hospitals with information on the lineage, clade and relevant mutations in the virus; (ii) to Public Health with epidemiological data; (iii) to BPS with the viral sequences for further secondary clinical data studies; (iv) to the research community with the viral genome sequences through the European Nucleotide Archive (ENA).
The Andalusian Health Research and Innovation Strategy 2020–2023, presented on 2 September 2020, focused on the improvement of the wellbeing of citizens in the framework of Horizon Europe 2027, including a response to the impact of the challenges of the SARS-CoV2 pandemic. The Regional Health Ministry has been working and collaborating on different initiatives for some time: (a) To promote Digital Clinical records (Diraya®), integrating all the information on people into a Single Health Record and facilitating access to all the services and provisions of the health system, ensuring that all the relevant information is structured. As opposed to systems that merely assemble records, the design of the applications in Diraya shared tables, codes and catalogues; (b) To create the Bioinformatics Research Area
[6], on 14 June 2016, to improve the technological support of personalized medicine, genomics and clinical genetics programs in the SSPA; (c) To create, by resolution of the management direction of the Andalusian Health Service 11 March 2018, the information system called BPS of the Andalusian Public Health System, which integrates clinical and epidemiological data from each patient; (d) The resolution of 9 March 2018, of the Public Business Entity Red.es, by which the Agreement with the Andalusian Health Service is published for the application of Information and Communication Technologies in the management of chronicity and continuity of care in the SSPA; (e) On 27 June 2019, at the meeting of the board of the Progress and Health Foundation (FPS), the creation of the B-D Area in Health of Andalusia was approved as an area integrated into the FPS in order to provide the SSPA of a platform of powerful, safe and data analysis tools oriented to health results and the optimization of healthcare processes based on personalized medicine; (f) During the last quarter of 2020, the coordination of the information and communication technologies (ICT) strategy of the SSPA was promoted; (g) The promotion of research on COVID19 in Andalusia, as of December 6, the number of research studies related to COVID-19, presented and/or evaluated in our Research Ethics Committees in Andalusia has been 277, with participation in 24 clinical trials addressing all the spectrum of COVID-19 from epidemiology, diagnosis, biomarkers, genetics (as a contributing study of the COVID-HGI), therapeutic interventions and vaccination. Some of them granted under the specific support for financing research, development and innovation (R+D+i) in COVID-19; (h) The Ministry of Health and Families, through its General Secretariat for Research, Development and Innovation in Health, has also set up three working groups: (1) prospective studies on the evolution of the pandemic; (2) personalized medicine in Covid-19; and (3) supplement and nutritional intervention against the SARS-Cov2 virus.
Implementing personalized medicine in Covid19 included developing actions to define by means of BD and AI the interaction of genomics, epigenetics, metagenomics and viral sequencing in the development of events such as infection, severe disease, response to treatment and response to vaccination. A joint instruction was carried out on January 2020 from the General Secretariat for Research, Development and Innovation in Health and the Management Directorate of the Andalusian Health Service for the Management of samples in the approach to Personalized Medicine in COVID-19. Healthcare professionals will also have access to SARS-CoV-2 virus complete sequencing study by electronic biochemical request (MPA). The San Cecilio Clinical Hospital for Eastern Andalusia and Virgen del Rocio University Hospital for Western Andalusia were established as reference centers for receiving viral samples (Figure 1A) and sequencing them, respectively (Figure 1B), and the Bioinformatics Area process sequencing data (Figure 1C), joint with COVID registry metadata (Figure 1D), previously collected from the Hospitals (Figure 1E), and reporting back relevant epidemiological information to the COVID registry (Figure 1F) and information on lineages and variants to the Hospitals for supporting clinical decisions (Figure 1G). As previously stated, the clinical data of the Andalusian Health System is stored in the BPS (Figure 1H), but in this case, viral genomes are also stored in BPS (Figure 1I) linked to the rest of the patient’s clinical data, offering an unprecedented opportunity for large-scale secondary studies and implementation in clinical practice (Figure 1). Finally, the Bioinformatics Area submits the viral sequences to the ENA database, which is available to the scientific community. Since February, more than 2000 whole viral genomes have been sequenced, allowing the construction of a resource that depicts the evolution of SARS-CoV-2 along time and across the geography of Andalusia
[6]. This systematic genomic surveillance system has allowed following the increase of the B.1.1.7 since February to become a majority or to detect new VOCs, such as the Brazilian lineage P1 or the South African B.1.351, and VOIs such as the Ugandan variant A.23.1 or others.
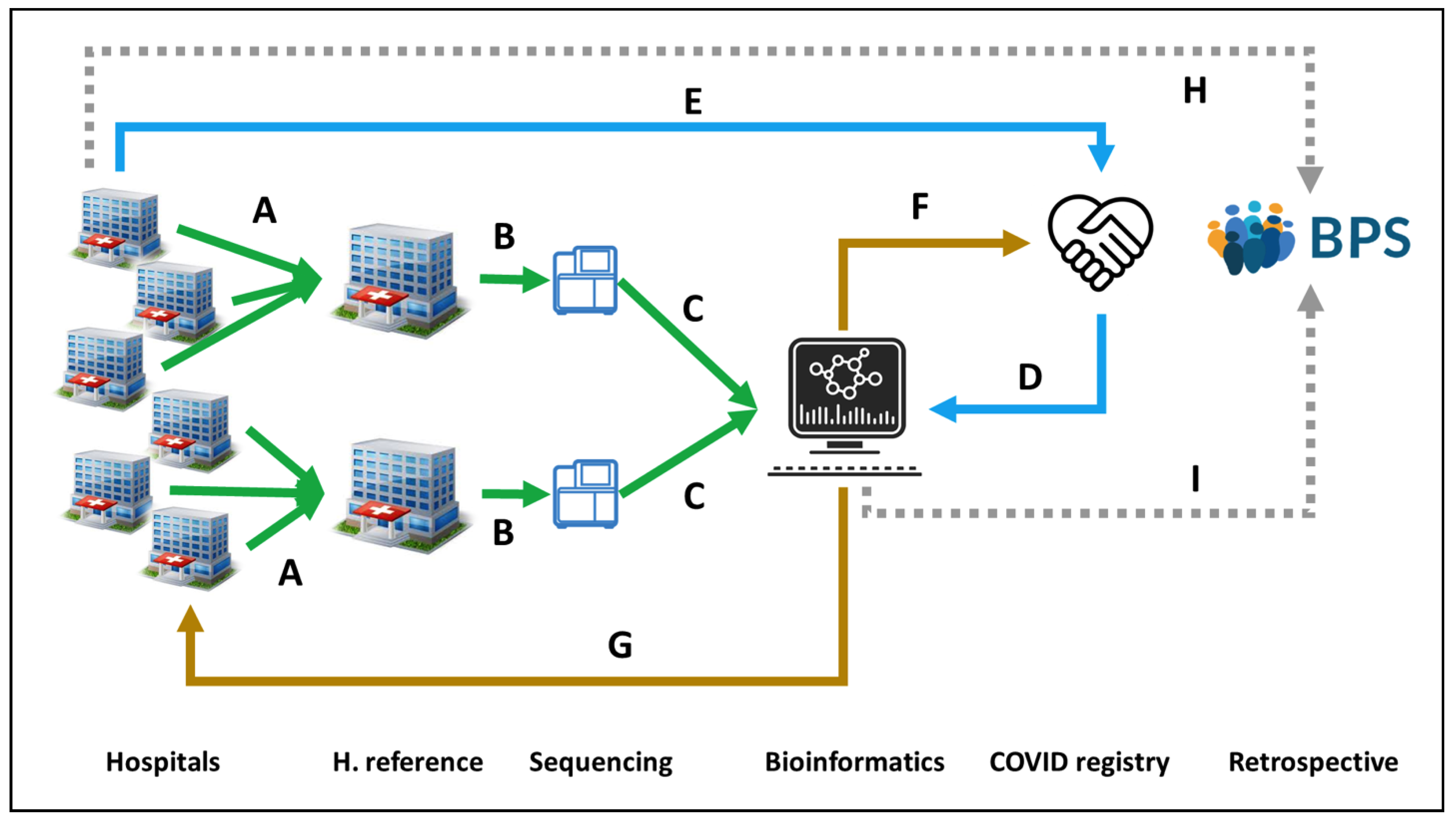 Figure 1.
Figure 1. Circuit for COVID19 genomic surveillance. (
A) Two reference Hospitals, San Cecilio and Virgen del Rocio, collect SARS-CoV-2 samples from Eastern and Western Andalusia, respectively. (
B) Samples are sent to GENYO or IBIS sequencing facilities. (
C) Sequencing data are sent to the Bioinformatics Area for processing and (
D) linking to metadata from the COVID registry, (
E) previously collected from the Hospitals. (
F) Bioinformatics reports relevant epidemiological information to the COVID registry and (
G) information on lineages and variants to the Hospitals for supporting clinical decisions. (
H) Clinical data on COVID-19 patients recorded in the Hospitals is stored in the BPS. (
I) Viral genomes are also stored in BPS linked to the rest of the patient’s clinical data for further secondary use.
3. Concluding Remarks
The pandemic has pushed us to a new scenario promoting association and relationship between governments and the scientific community at the same time that emerged multidisciplinary teams to take care of this complex disease together with telemedicine to guarantee health care keeping at home. Deep sequencing, bioinformatic area and clinicians working on personalized medicine could help to better understand the interaction between the virus and the host. These tools should be available for physicians able to include in their everyday decision-making process. The increasing need for personalized medicine supported by scientific and objective data, big data and AI systems to create algorithms based on individual variables (genomic), the host and the guest (pathogen and patient subject). Public and private investment for the generation and transfer of knowledge could support the development of high-quality translational and collaborative research to face a threatening situation similar to this terrible pandemic.

 Figure 1. Circuit for COVID19 genomic surveillance. (A) Two reference Hospitals, San Cecilio and Virgen del Rocio, collect SARS-CoV-2 samples from Eastern and Western Andalusia, respectively. (B) Samples are sent to GENYO or IBIS sequencing facilities. (C) Sequencing data are sent to the Bioinformatics Area for processing and (D) linking to metadata from the COVID registry, (E) previously collected from the Hospitals. (F) Bioinformatics reports relevant epidemiological information to the COVID registry and (G) information on lineages and variants to the Hospitals for supporting clinical decisions. (H) Clinical data on COVID-19 patients recorded in the Hospitals is stored in the BPS. (I) Viral genomes are also stored in BPS linked to the rest of the patient’s clinical data for further secondary use.
Figure 1. Circuit for COVID19 genomic surveillance. (A) Two reference Hospitals, San Cecilio and Virgen del Rocio, collect SARS-CoV-2 samples from Eastern and Western Andalusia, respectively. (B) Samples are sent to GENYO or IBIS sequencing facilities. (C) Sequencing data are sent to the Bioinformatics Area for processing and (D) linking to metadata from the COVID registry, (E) previously collected from the Hospitals. (F) Bioinformatics reports relevant epidemiological information to the COVID registry and (G) information on lineages and variants to the Hospitals for supporting clinical decisions. (H) Clinical data on COVID-19 patients recorded in the Hospitals is stored in the BPS. (I) Viral genomes are also stored in BPS linked to the rest of the patient’s clinical data for further secondary use.