1000/1000
Hot
Most Recent

Small Leucine-Rich Proteoglycans (SLRPs) are key extracellular matrix proteins that play a role in many fundamental biological processes involved in the maintenance of retinal homeostasis.
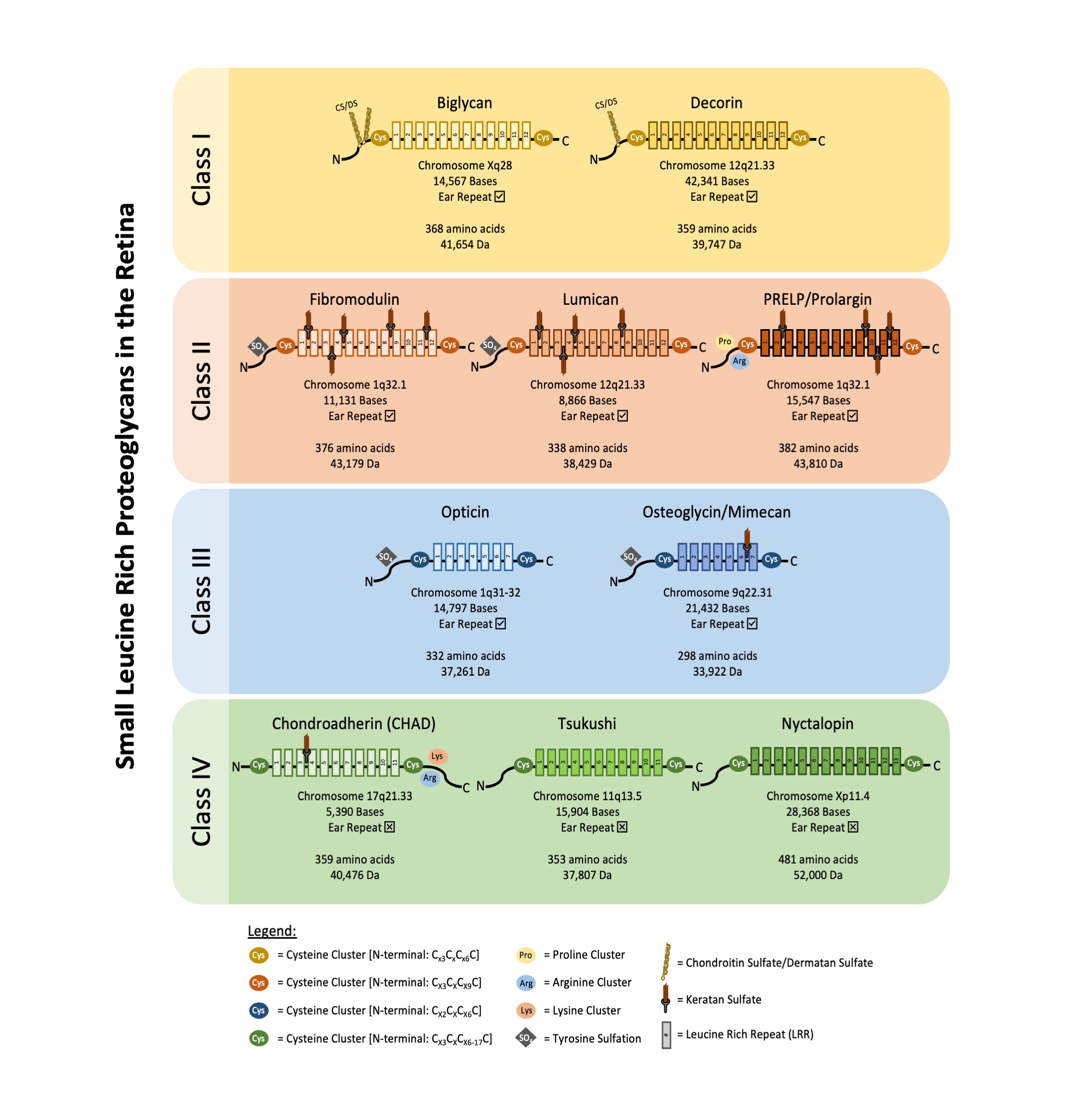
SLRPs can be broadly divided into five distinct classes based on their number of LRRs, amino acid residues at the N-terminus and their chromosomal organization. In canonical classes I–III, a capping motif, comprising of two terminal LRR and an “ear repeat”, can be found. These ear repeats maintain the protein core’s structural conformation and influences its ligand binding ability [5]. On the other hand, Class IV and V SLRPs do not have ear repeats [6]. Figure 1 summarizes the gene–protein information for each SLRP that has been found in the retina. Table 1 summarizes the distribution of known SLRPs in the retina.
|
Biglycan |
Decorin |
Fibromodulin |
Lumican |
PRELP/ Prolargin |
Opticin |
Osteoglycin/ Mimecan |
Chondro-adherin (CHAD) |
Tsukushi |
Nyctalopin |
|
|---|---|---|---|---|---|---|---|---|---|---|
|
ILM |
Human [7] |
Human [7] |
Human [7] |
Human [7] |
Human [7] |
Human [7] |
Human [7] |
|||
|
NFL |
Human [7], Mice [8] |
Human [7], Mice [8], Rat [9] |
Human [7], Mice [8] |
Human [7] |
Human [7] |
Human [7] |
Human [7] |
|||
|
GCL |
Human [7], Mice [8] |
Human [7], Mice [8], Rat [9] |
Human [7], Mice [8] |
Human [7], Mice [10] |
Human [7] |
Human [7] |
Human [7] |
Rat [12], Chick [13] |
||
|
IPL |
Human [7], Mice [8] |
Human [7], Mice [8], Rat [9] |
Human [7], Mice [8] |
Human [7], Mice [10] |
Human [7] |
Human [7] |
Human [7] |
Mice [14] |
||
|
INL |
Human [7], Mice [8] |
Human [7], Mice [8], Rat [9] |
Human [7], Mice [8] |
Human [7], Mice [10] |
Human [7] |
Human [7] |
Human [7] |
Mice [16] |
Rat [12], Chick [13] |
|
|
OPL |
Human [7], Mice [8] |
Human [7], Mice [8], Rat [9] |
Human [7], Mice [8] |
Human [7], Mice [10] |
Human [7] |
Human [7] |
Human [7] |
Mice [14] |
Mice [12], Rat [12], |
|
|
ONL |
Human [7] |
Human [7], Mice [8], Rat [9] |
Human [7], Mice [8] |
Human [7] |
Human [7], Mice [17] |
Human [7] |
Human [7] |
|||
|
PRL |
Human [7] |
Human [7], Mice [8], Rat [9] |
Human [7], Mice [8] |
Human [7] |
Human [7] |
Human [7] |
Human [7] |
Mice [14] |
Human [11] |
|
|
IPM |
Rat [18] |
|||||||||
|
RPE |
Human [7] |
Mice [17] |
Human [7], Canine [19] |
ILM inner limiting membrane; NFL nerve fiber layer; GCL ganglion cell layer; IPL inner plexiform layer; INL inner nuclear layer; OPL outer plexiform layer; ONL outer nuclear layer; PRL photoreceptor layer; IPM interphotoreceptor matrix; RPE retinal pigmented epithelium.
Fibromodulin, lumican and proline/arginine-rich end leucine-rich repeat protein (PRELP) are class II SLRPs.
Opticin and osteoglycin/mimecan are class III SLRPs.
Opticin may play a role in vitreoretinal adhesion by modulating macromolecular surface coatings of collagen fibrils in the inner limiting membrane [35]. It also prevents pathological angiogenesis by acting as a competitive inhibitor to prevent the adhesion of endothelial cells to collagen [36]. This is supported by another study whereby opticin deficient OIR mice depicted a significant increase in preretinal neovascularization [37]. Opticin may also serve as a potential biomarker for neovascular AMD [38]. Additionally, it has been shown to bind and regulate growth factors in the chick retina [39].
Osteoglycin/mimecan has been found to interact with the leucine-rich protein, B7, which is expressed in the human cornea, iris, retina and sclera [40]. However, little is known about B7’s role in modulating retinal integrity.
Chondroadherin, tsukushi and nyctalopin are class IV SLRPs.
Chondroadherin (CHAD) has been detected in both human [7] and mouse [14] retina. However, its functional roles have yet to be determined.
Tsukushi is expressed in mice inner nuclear layer [16] and suggested to be involved in the regulation and proliferation of Müller glia. However, further evaluation will be required to determine its role in the retina.
Nyctalopin has been found to be associated with transient receptor potential melastatin 1 (TRPM1) [41] and mGluR6 [42]. They form a complex that enables fast signal transmissions for the ON-bipolar cell (BC) response. Another protein, LRIT3 has also been reported to interact with nyctalopin and is required for the localization of nyctalopin to ON BC dendritic tips [43]. Nyctalopin also regulates retinal activity required for the maintenance of axon terminal segregation in the dorsal–lateral geniculate nucleus [44]. Mutations in the nyctalopin gene can result in congenital stationary night blindness [45] and myopia [46].
Apart from the 10 SLRPs discussed above, there are seven other SLRPs yet to be studied in the retina, namely, extracellular matrix 2 (ECM2) and asporin from class I; osteoadherin and keratocan from class II; epiphycan from class III; and podocan and podocan-like-protein-1 (Podnl1) from class V. Further evaluation to determine the role of SLRPs in the retina can potentially expand our understanding of complex retinal diseases and provide new opportunities for the treatment for the vision loss.