1000/1000
Hot
Most Recent

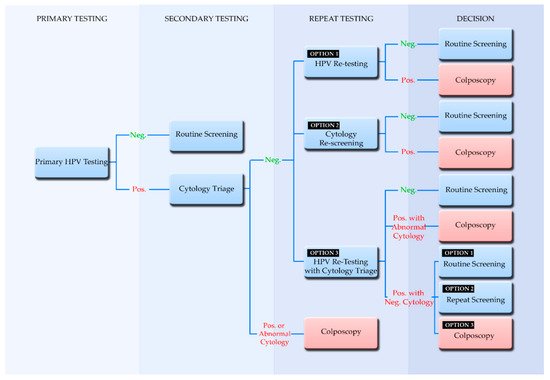
The human papillomavirus is one of the most common sexually transmitted viruses, and an infection from this virus may become persistent, leading to diseases such as cervical cancer. In the past, cytology-based methods such as the Papanicolaou (Pap) test were imperative to identify the disease at a stage where it can be treated. However, since the 1980s where the etiological association of HPV and cervical cancer was identified, new tests began emerging directed towards identifying the virus. Furthermore, as the biology of HPV along with the relationships with its host are elucidated, these tests and treatments further advance. Recently in Europe, there is a movement towards the implementation of HPV testing methodologies in national screening programs to precede cytological testing. These screening strategies are recommended by the European guidelines and the World Health Organization. This review presents the current HPV testing methodologies, their application in organized population-based cervical cancer screening programs based on the most recent European guidelines, and their implementation status in countries in Europe.
| Tests | Hybrid Capture 2 (Qiagen) | GP5+/6+ EIA a | Cobas 4800 HPV Test (Roche) | APTIMA HPV Assay (Hologic) | BD Onclarity HPV Assay |
|---|---|---|---|---|---|
| Type of assay | Signal amplification, hybrid capture | PCR, probe hybridization |
Real-time PCR detection | Transcription mediated amplification, probe hybridization |
Real-time PCR detection |
| Targets | DNA, Whole viral genome | L1 DNA, 150 bp |
L1 DNA 200 bp | E6/E7 mRNA | E6 and E7 DNA |
| HPV Subtypes detected |
16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59 and 68 | 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68 | 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66 and 68. Individual genotyping for: 16, 18 |
16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68. Reflex Partial genotyping for: 16, 18–45 |
33–58; 56–59–66; 35–39–68 f. Individual genotyping for: 16, 18, 31, 45, 51, and 52 |
| Internal Controls Human genes |
NO | NO | Internal human β-globin control | Internal RNA transcript (HPV16 E6/7) control | Internal human β-globin control |
| Capacity Batch size |
88 | 96 samples in 9.5 h e |
96 | Panther system 100 and 250 test /Tigris DTS system 250 | 46 |
| VALGENT Validation |
Standard comparator tests for validation b | Standard comparator tests for validation b | YES | YES | YES |
| US FDA c Validation | YES | NO | YES | YES | YES |
| CE Mark d Validation | YES | YES | YES | YES | YES |
| Uses within a screening program | ASC-US Triage, test-of-cure |
ASC-US Triage, test-of-cure |
ASC-US Triage/co-testing/Primary testing | ASC-US Triage/co-testing | ASC-US Triage/co-testing/Primary testing |