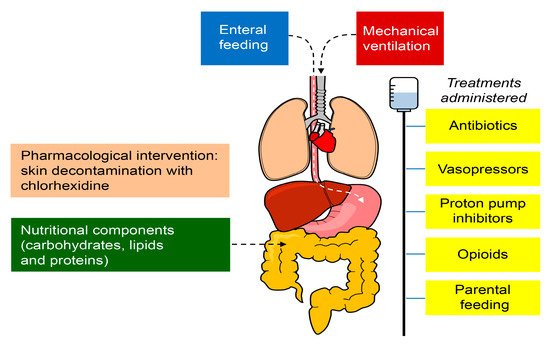
The intestine has long been hypothesized as "the engine" of critical illness, but its clinical importance needs to be better defined. The gut microbiome is severely altered in multiple disease states, including critical illness, where the health-inducing microbiome becomes a disease-promoting pathobiome that makes the patient more vulnerable to nosocomial infections, sepsis, and multiple organ failure
[12]. So far, only a few studies have analyzed the gut microbiome in critically ill patients, and they have confirmed a state of dysbiosis
[13][14][15]. Moreover, recent studies in intensive care unit (ICU) patients observed a gradual worsening of the dysbiosis during their stay in the ICU
[14][16][17][18]. The most relevant changes in the microbiome can be seen in the largest study to date that examined the sequencing of the 16S rRNA gene from multiple body sites (skin, oral, and feces) from 115 ICU patients and compared it with 1242 healthy volunteers
[14]. At the intestinal level there was a low prevalence of the Firmicutes and Bacteroidetes phyla, and a greater richness of Proteobacteria in comparison to healthy individuals. At the genus level, there was a lower prevalence of key commensal genera (such as
Faecalibacterium—an anti-inflammatory organism,
Blautia, and
Ruminococcus), and in some cases, an overgrowth (over 50% relative abundance) of genera with pathogenic properties, such as
Escherichia/
Shigella,
Salmonella,
Enterococcus,
Clostridium difficile, or
Staphylococcus [13][14][15][16][19]. It has been proposed that changes in the
Firmicutes/Bacteroidetes ratio can predict patient outcome
[16], although further work is required to validate these findings.
Overall, critically ill patients admitted to the ICU present a gut microbiome characterized by lower bacterial diversity and large inter-individual variation. A study of 14 ICU patients also reported the emergence of ultra-low-diversity communities in 35% of patients who only presented one to four bacterial taxa
[19]. In previous studies, low microbial diversity has been associated with an increased risk of mortality
[20][21], and the domination of certain pathogens have been identified as an independent risk factor for adverse outcomes
[22][23][24].
Considering all the studies on critically ill patients, Proteobacteria is the dominant phyla, and Firmicutes is reduced, whereas Enterococcus, Staphylococcus, and Enterobacter are increased in septic patients. In these patients with sepsis, the focus often lies on the identification of a single pathogen as the causative agent. However, there is an increasing belief that most infections have “polymicrobial” phenotypes that depend on the microbiome status of the patient. Thus, the initial state of the microbiome can determine both the susceptibility to infection
[25] and its severity
[26]. The composition and functions of the intestinal microbiome of critically ill patients and healthy humans are summarized in
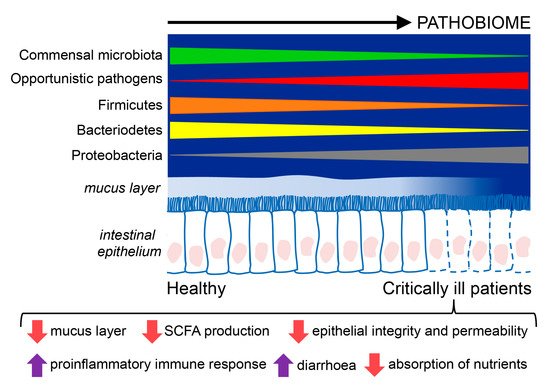
Figure 1.
Figure 1. Composition and functions of the intestinal microbiome in critically ill patients compared to healthy individuals. Critically ill patients exhibit an intestinal disease-promoting microbiome or pathobiome. This pathobiome is characterized by a lower prevalence of the Firmicutes and Bacterioidetes phyla, and a higher prevalence of the Proteobacteria phyla, in contrast to healthy individuals. Furthermore, the intestinal epithelium is altered in critically ill patients, showing reduced reperfusion, that could lessen the hydrophobicity of the mucus layer and favor the translocation of pathogens through gaps between the epithelial cells, and epithelial apoptosis, resulting in poor absorption of nutrients, diarrhea, loss of fecal energy, and lower production of short chain fatty acids (SCFA).
Furthermore, migration of microorganisms between the intestinal and pulmonary microbiome has been reported in critically ill patients. A recently published study highlights the impact of the gut microbiome on the pulmonary microbiome. It was observed that the pulmonary microbiome in both murine sepsis and human acute respiratory distress syndrome (ARDS) was enriched with bacteria associated with the intestine. An operative taxonomic unit of Bacteroides was detected in the bronchoalveolar fluid (BAL) samples from 41% of patients with ARDS compared to 3% in healthy patients. Moreover, the systemic and alveolar levels of tumor necrosis factor (TNF)-α in patients with ARDS were markedly increased by the presence of organisms derived from the intestine in the BAL. However, the precise route by which the intestinal microorganisms reached the lungs of the mice with sepsis has not been identified
[27].