1. Introduction
Bisphosphonates (BPs) are a significant class of medications that are used in the treatment of diseases linked with low bone mass, including Paget’s disease, osteoporosis, and the inhibition of skeletal-related events (SRE) in individuals with osteolytic bone metastases and multiple myeloma
[1][2]. BP was synthesized for the first time in the 1800s. Its structure in
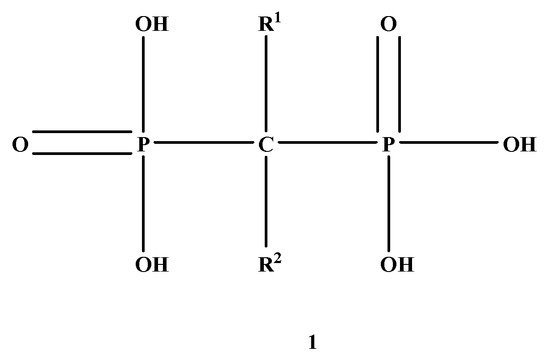
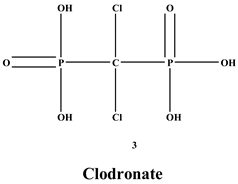
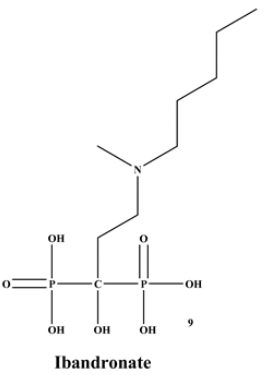
Figure 1 is composed of two phosphate groups binding to a carbon atom (P-C-P), and their potential to bind to the bone surface depends on the P-C-P bond. This bond also improves their binding to the mineralized bone matrix with a subsequent inhibitory effect on bone resorption
[3]. The R1 side chain that contains a hydroxyl group (OH) is attached to the central carbon atom and further enhances its binding affinity
[1]. BPs anti-resorptive activity is ascribed to the R2 side chain. The existence of a primary amine (NH
2) that is attached to the central carbon atom raises strongly anti-resorptive effectiveness
[1]. Hence, the addition of hydroxyl group and/or primary amine group increases the affinity for calcium ions, which results in favored localization of the remedies to the sites of bone remodeling
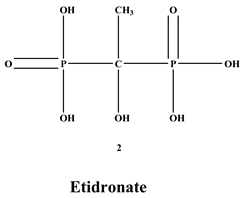
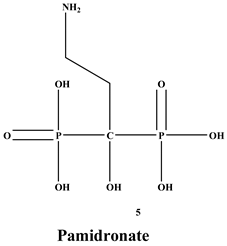
[4]. BPs are classified into two groups which are nitrogen-containing bisphosphonates (N-BPs) e.g., pamidronate, neridronate, alendronate, etc., and non-nitrogen containing bisphosphonates (N-NBPs) e.g., etidronate and clodronate
[5]. They are also classified into three generations based on their therapeutic effects, as shown in
Table 1. The 3rd generation BPs are more effective than the 1st and 2nd generation BPs
[5][6]. The N-NBPs are incorporated into the energy pathways of the osteoclast, resulting in disrupted cellular energy metabolism leading to apoptosis. N-BPs exert their effect on osteoclasts via their inhibition of the mevalonate pathways, resulting in the disruption of intracellular signalling and induction of apoptosis
[7].
Figure 1. Structure of Bisphosphonate.
In the treatment of osteoporosis, BPs are used, and they are active for some pathologies characterized by abnormally high bone resorption, which includes metastatic bone cancer, etc.
[8][9]. BPs have experienced a huge development in the treatment of some bone diseases
[10][11].
Currently, some uses of BPs are:
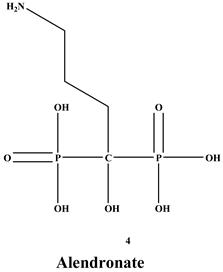
- ✓ In corticosteroid and postmenopausal induced osteoporosis, the most used BPs in these cases, which block pathological fractures, is alendronate [12].
- ✓ They are also used to enhance bone morphology and decrease pain in Paget’s disease [13].
- ✓ In hypercalcaemia of malignancy, its role is to check hypercalcaemia, reducing pain, and preventing the development of osteolytic lesions and fractures [14].
- ✓ In people with bone metastasis, it is used to decrease fractures, hypercalcemia, and relieve pain [15][16].
- ✓ They are used to decrease associated bone pathologies in multiple myeloma, including pain, fractures, and vertebral collapse [17][18].
Despite the efficacy of BPs in the treatment of many bone-related diseases and their relatively good record, BPs also suffer from several limitations such as diarrhea, esophageal erosion, gastric ulcers, rare cases of esophageal cancer, hepatotoxicity, conjunctivitis and hypocalcemia, bone pain, fever, osteonecrosis of the jaw, and femur fracture
[19][20][21][22]. This review focuses on bisphosphonate-based conjugate compounds developed for treating osteoporosis, bone cancer, and cancers that spread to the bones, such as prostate, lung, and breast cancer.
2. Osteoporosis
Osteoporosis is a leading public health challenge, and it has become one of the most dominant chronic diseases in the world
[23]. It is the most widely recognized skeletal disease that is initiated by an imbalance in bone metabolism, leading to a gradual loss of bone mass and attenuation of bone strength, thus disposing to low-energy fractures
[24]. It is common in older adults, more especially postmenopausal women. This disease is characterized by bone loss that results in bone fractures in the spine and hip
[25]. In developed countries such as the United States, the number of individuals with osteoporosis is increasing because of the increasingly aged population. Therefore, the treatment and prevention of osteoporosis are of great concern
[25]. The estimates of women in danger of osteoporotic fractures and the respective economic burden to the health care systems in the next decades are expected to grow continuously
[24]. In 2005, the cost for the treatment of fractures due to osteoporosis in the United States was $16.9 billion; it is predicted that the cost will increase to $25.3 billion by the year 2025
[23]. The pharmacological agents utilized in the management of osteoporosis include medications that precisely inhibit bone loss, decrease the risk of future fractures, and also increase bone strength
[26]. In patients with osteoporosis, BPs are known drugs that inhibit the loss of bone mass. The therapeutic efficacy of BPs is based on their remarkable selectivity to the bone rather than other tissues
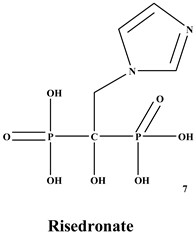
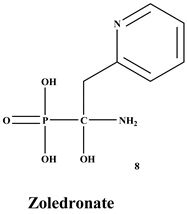
[27]. Many BPs have been approved and utilized to manage osteoporosis, e.g., alendronate, risedronate, zoledronate, ibandronate, etc., in several countries
[26][28]. BPs decrease fracture risk by 30–70% at the spine and increase bone mass. They do the same at varying degrees at other skeletal sites in postmenopausal women
[28][29].
Risk factors of osteoporosis include:
- ✓ Age: the people at high risk are individuals who are age 65 and above.
- ✓ Gender: men are at less risk than women because of the absence of menopause [30].
- ✓ Family ancestry plays a significant role.
- ✓ Drinking too much alcohol and smoking are risk factors for osteoporosis [31].
Bisphosphonates Conjugates for the Treatment of Osteoporosis
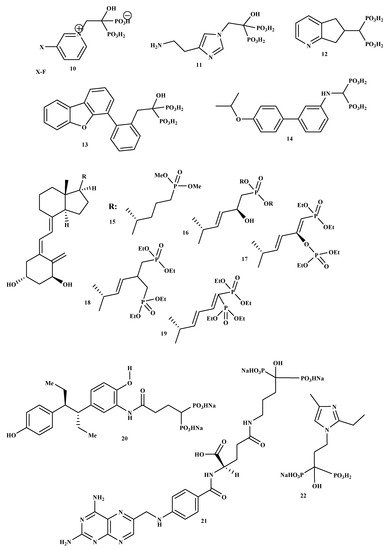
New BP-conjugate anti-osteoporotic agents have been designed and are presented in
Figure 2. Compounds 10, 11, 12, 13, 14 were not effective compared to the drugs that are already utilized to treat osteoporosis
[32]. Therefore, there is still a need to develop drugs that will be effective as anti-resorptive agents for treating osteoporosis. It is reported that vitamin D conjugates for effective for improving good bone health; hence, Steinmeyer et al. synthesized a series of novel vitamin D conjugates by combining bisphosphonate moieties and vitamin D
[33]. The in vitro evaluation of vitamin D conjugates (15, 16 and 18, 19) exhibited moderate activities, while 17 showed a highly moderate vitamin D-like activity in HL 60 and ROS 17/2.8
[33]. Tsushima et al. synthesized and evaluated the effects of a new hybrid compound of estrogen-bisphosphonate, 20
[34]. The results showed that 20 is an effective bone-targeting therapeutic, and it also displayed a strong ability to inhibit bone loss. Therefore, utilizing bisphosphonates for hybrid compounds is a promising approach for the successful delivery of estrogen to the bone
[34]. Xie et al. prepared and characterized a new bone-targeting conjugate, methotrexate-alendronate, 21.
1H-NMR,
13C-NMR,
31P-NMR, and LC-MS were used to characterize the conjugate
[35]. The conjugate compound prevented bone resorption and the formation of osteoclast compared to methotrexate and alendronate utilized alone. Compound 21 potential to adhere to the exposed bone surface and improve drug accumulation in the pathological region for targeted therapy against osteoclastogenesis was significant. Therefore, the results revealed methotrexate-alendronate conjugate as a potential therapeutic for the treatment of rheumatoid arthritis and osteoporosis by targeting osteoclastogenesis
[35]. Motaleb et al. synthesized and characterized a novel zoledronate conjugate, 22, and successfully radiolabeled with 99mTc
[36]. Compound 22 displayed excellent biological activities with high preferential uptake to the bone with clearance from blood and soft tissues, and these results suggest zoledronate conjugate is a promising drug as an anti-osteoporotic agent
[36].
Figure 2. Structures of BP-conjugates 10–22, potential compounds reported for the treatment of osteoporosis.
3. Bone Cancer
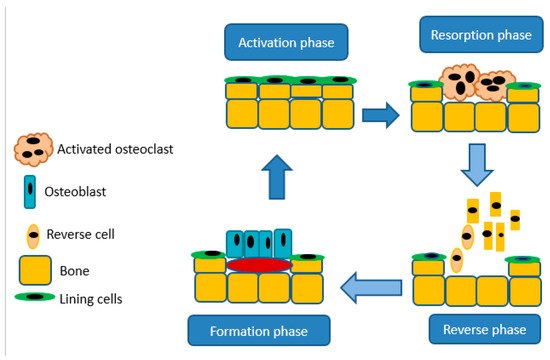
Bones are crucial in the body for movement and structure, mineral storage, protection of organs, and blood cell production, provides a site as bone marrow houses hematopoietic cells for the production of leukocytes, platelets, and red blood cells
[37]. Bones are essential for storing phosphorus and calcium in the body because of their hydroxyapatite crystal structure. Bones continuously undergo cycling of resorption and deposition of the osteoid and hydroxyapatite matrix. Bone releases minerals via resorption by the osteoclasts, the cells accountable for separating bone matrix, whereas osteoblasts develop novel bone through emitting chondroitin, osteocalcin, and collagen to formulate osteoid
[37]. The balance between bone formation and resorption relies upon signals from chemokines, cytokines, and mechanostimulation. Moreover, resorption by osteoclasts is important for the maintenance of typical bone density. An imbalance in these procedures results in hyperplastic bone, including mutual degenerative bone infections
[37]. A schematic illustration of bone resorption and formation is shown in
Figure 3.
Figure 3. A schematic diagram illustrating bone resorption and bone formation.
For cancer patients, it is reported that cancer usually spreads to the bone. Metastatic bone cancer happens mostly in patients with prostate, lung, and breast cancer
[38]. The resulting skeletal events include spinal cord compression, pathologic fractures, and bone pain. Metastasis occurs when cancer cells spread from the primary cancer site to an inaccessible site through the circulatory system forming secondary cancer
[39]. The reported median survival rate for people with metastatic bone resulting from breast, renal, and prostate cancer ranges from 12 to 33 months
[35]. The survival rates for primary lung cancer patients are very low and range from 9.5% to 12% in 12 months
[38]. People with bone metastases can experience some limitations in their daily lives, such as increased medical expenses, decreased quality of life, and the threat of survival
[40].
Osteosarcoma is a well-known diagnosed primary bone malignancy
[41]. Osteosarcoma generally occurs either during active growth phases in adolescence and young adults or often associated with a pre-existing condition, like Paget’s disease (in adults above 65)
[41]. The occurrence of bone cancer in males is higher than in females. However, osteosarcoma in females develops earlier compared to males
[42]. Metastatic cancer affects the skeleton. In particular, cancers with a high incidence and relatively long clinical course include prostate, lung, and breast cancers, and they often metastasize to the bone
[43][44][45]. Most primary cancers and bone metastases exhibit improved osteoclast action and bone resorption
[46][47] that can lead to hypercalcemia, pathological fractures, and pain
[45]. Therefore, techniques to decrease the incidence and morbidity of bone metastases are obviously of tremendous clinical significance
[41]. The treatment that is currently used is the combination of chemotherapeutics and surgery. The combination of high dosages of methotrexate, cisplatin, ifosfamide, and doxorubicin have prompted an increased survival rate. Nonetheless, the utilization of anticancer medications is limited by severe side effects. The poor bone blood supply, drug resistance, and non-specific uptake require the utilization of high toxic doses of anticancer drugs
[41]. Hence, there is a need to develop bone-targeted anticancer agents with reduced and/or no side effects for the treatment and prevention of malignant growth-related bone diseases
[48].
Numerous therapeutic techniques have been developed for the management of malignant-related skeletal complications. These approaches incorporate either anti-neoplastic intervention, for example, chemotherapy, hormonal therapies, radiotherapy, and topical surgeries, or bone helpful treatment such as calcium, analgesics, and vitamin D supplements
[39]. In the treatment of malignant bone metastasis, synthetic anti-resorptive agents are considered to be indispensable
[39]. BPs are known as the most effective of these drugs. They have an established role as palliative treatment in people with skeletal metastases. BPs increases the prevention of cancer cell invasion and adhesion to the bone matrix and induce apoptosis of osteoclasts
[39]. Some studies reported that BPs stimulate osteoblastic bone formation in vitro and in vivo. BPs is one class of pharmacological agents used for the prevention and management of myeloma bone disease. However, agents like anti-RANKL antibody, Denosumab are also utilized in people with bone metastasis resulting from prostate and breast cancer. Denosumab is currently under clinical trials in patients with multiple myeloma
[49]. Anticancer agents, for example, doxorubicin, cisplatin, and methotrexate were, covalently incorporated into the terminal amino group of the amino-BP alendronate utilizing a suitably bio-reversible amide bond. The BP-conjugates displayed prolonged blood circulation, binds to the hydroxyapatite matrix of the bone, and hence released the BP together with the cytostatic agent into the bone micro-environment via the cleavage of the amide linker of the BP-conjugate
[50].
4. Metastatic Cancer That Spreads to the Bones
Bone metastasis is linked with most types of cancers, leading to high mortality and morbidity in cancer patients. However, it is most prevalent in people living with lung, breast, and prostate cancer
[51][52]. Although bone metastasis is common in myeloma, prostate, and breast cancer, according to radiographic appearance, the primary pathogenesis often includes both osteolytic and osteoblastic processes
[53]. About 75% of patients suffer from severe pain during the time of diagnosis of metastatic bone. It may lead to SREs, such as hypercalcemia, pathological fractures, and spinal cord compression resulting in increased medical care cost, shortened survival rate, and decrease quality of life
[52]. Some researchers reported BPs could prevent tumor cell invasion, angiogenesis, proliferation and enhance survival rate in vivo
[54]. BPs have also been utilized to minimize pain in multiple myeloma and reduce skeletal complications
[55]. In addition, BPs can slow down bone-resorption and exert cytotoxic effects on osteoclasts
[56]. In people with cancer, BPs can affect the bone micro-environment to decrease the invasion of the cancer cells
[57]. BPs also exhibit anti-angiogenic and anti-tumor activity
[58][59][60][61][62][63]. However, the biological diversity of bone metastasis and tumors can result in different responses to BP remedies. Moreover, BPs can expose patients to serious side effects such as osteonecrosis of the jaw, hypocalcemia, and nephrotoxicity
[64].
Osteonecrosis of the jaw is one of the most severe side effects. Most cases of bisphosphonate-related osteonecrosis of the jaw have been reported in people with breast cancer and multiple myeloma treated with high doses of intravenous bisphosphonates. Moreover, it has also been reported in people taking bisphosphonates for the treatment of osteoporosis. The risk factors for bisphosphonate-related osteonecrosis of the jaw comprise the usage of higher doses, intravenous bisphosphonates and prolonged duration of administration. Furthermore, the usage of anticancer therapy and glucocorticoids, smoking, cancer, and history of diabetes are additional risk factors
[37][65]. Hypocalcemia is also a common problem associated with bisphosphonate usage; moreover, the incidence can be as high as 18%. Hypocalcemia is more common, secondary to intravenous bisphosphonates, and in people with hypoparathyroidism, an underlying untreated vitamin-D deficiency, poor calcium intake, and hypocalcemia. Vitamin D and calcium deficiency should be treated before administering bisphosphonates, especially intravenous bisphosphonates
[66][67]. Some BPs such as pamidronate, zoledronate, clodronate, and ibandronate have been in clinical use for the management of health problems associated with tumors that cause osteolysis. However, there is increasing evidence from preclinical studies that demonstrate BPs good antitumor activity
[68][69]. Numerous investigations have revealed a decrease in the risk of colorectal and breast cancer among BP users. Therefore, BPs can be clinically effective for the prevention and treatment of tumors
[70].