Triple negative breast cancer (TNBC) is a heterogeneous tumor characterized by early recurrence, high invasion, and poor prognosis. Currently, its treatment includes chemotherapy, which shows a suboptimal efficacy. However, with the increasing studies on TNBC subtypes and tumor molecular biology, great progress has been made in targeted therapy for TNBC. The new developments in the treatment of breast cancer include targeted therapy, which has the advantages of accurate positioning, high efficiency, and low toxicity, as compared to surgery, radiotherapy, and chemotherapy. Given its importance as cancer treatment, we review the latest research on the subtypes of TNBC and relevant targeted therapies.
1. Introduction
Breast cancer is the most common malignancy among women with an incidence rate of 10,492 in 100,000 cases in 2015, according to reports of the Ministry of Health and Welfare. The high morbidity and mortality rates cause a financial burden with regards to health insurance [1]. Several risk factors for breast cancer were investigated, including alcohol intake (relative risk: 1.10; 95% confidence interval: 1.06–1.14) [2], obesity (hazard ratio: 1.58, 95% confidence interval: 1.40–1.79) [3], age (hazard ratio: 10.1, 95% confidence interval: 8.49–11.94) [4], early menarche, and late menopause. Despite several years of bench to bedside investigations, breast cancer remains a challenge for all physicians, with the detailed mechanism of its tumorigenesis still unclear. Its molecular and biological attributes were investigated in detail to better understand the development and pathogenesis of breast cancer, including oncogenes, the tumor suppression genes, and downstream pathways that promote tumor proliferation, differentiation, and distant metastasis. The treatment of breast cancer requires a multidisciplinary approach involving surgery, radiation, and medical oncology to control tumor cell proliferation and metastasis. The new treatments for breast cancer are also discussed in this article, including the use of PARP, MAPK, CHK1, CDK 4/6 inhibitor, anticytotoxic T-lymphocyte-associated protein 4 (anti-CTLA4) antibodies, and anti-programmed cell death protein 1 (anti-PD1)/ligand 1 (anti-PD-L1) antibodies. We believe that elucidating the mechanism of breast cancer will provide an important foundation for therapeutic intervention. This article provides an update on research and clinical results using the molecular biology of tumorigenesis and summarizes current treatments to provide a new strategy for physicians.
2. Therapeutic Strategy for TNBC
2.1. HER2 Inhibitor
Since the first discovery of
HER-2 in breast cancer, its importance in the occurrence and development of breast cancer has gradually been recognized. Current targeted therapy research focuses on blocking the
HER-2 signaling pathway. Trastuzumab, a recombinant DNA-derived humanized monoclonal antibody, is a novel drug for the treatment of metastatic breast cancer. Trastuzumab has achieved good results in both
HER-2-positive early and late-stage breast cancer treatments. Trastuzumab is used alone or in combination with chemotherapy for patients with advanced breast cancer. Trastuzumab treatment can prolong patient survival with low side effects. In a further in-depth study, the results of a combination chemotherapy regimen using trastuzumab with docetaxel or taxane resulted in overall survival of patients that was higher than that obtained using chemotherapy alone
[5]. Therefore, trastuzumab combined with paclitaxel is the first-line treatment for
HER-2 positive breast cancer patients. The investigators conducted a four-year follow-up study showing that patients who received trastuzumab in the observation group were more likely to have a higher survival rate (including disease-free survival) than those who did not receive treatment
[6]. Since the patient’s treatment depends on his or her personal will, it is impossible to carry out rigorous statistical analysis, but patients receiving treatment have increases in disease-free survival and overall survival.
HER-2-targeted therapy has not benefited patients with low
HER-2 expression in TNBC treatment strategies; however, combination therapy may be efficacious. The primary analysis of a phase IIb trial investigating the HER2-derived vaccine nelipepimut-S (NPS) did not benefit the intention-to-treat population (NPS;
n = 55 vs. placebo;
n = 44), but a subgroup analysis showed benefit in patients with TNBC
[7]. Another study enrolled 136 people receiving NPS/GM-CSF and 139 people receiving placebo/GM-CSF. The results of the study confirmed that concomitant administration of trastuzumab and NPS with GM-CSF was safe and had no additional overall toxicity. The combination of
HER-2-targeted NPS and trastuzumab was safe. In
HER-2 low-expressing breast cancer, no significant differences in DFS were observed in the intention-to-treat analysis
[8]; however, significant clinical benefits were observed in TNBC patients. These findings warrant further investigation in a phase III randomized trial.
Targeting RTK Signaling
Lapatinib is an RTKI that reversibly blocks
EGFR and
HER-2 [9], inhibits downstream
MAPK/Erk1/2 and
PI3K/AKT pathways
[10], and enhances trastuzumab-dependent cell-mediated cytotoxicity
[11]. In March 2007, the FDA approved lapatinib in combination with capecitabine for
HER-2-positive breast cancer previously treated with anthracyclines, paclitaxel, and trastuzumab
[12] In February 2010, the FDA approved lapatinib in combination with letrozole for the first-line treatment of hormone receptor (HR)-positive and
HER-2 overexpressing postmenopausal metastatic breast cancer
[13].
Lenatinib is a TKI that irreversibly inhibits
HER-1,
HER-2, and
HER-4. Early clinical studies have shown that lenatinib exerts more effective inhibition than lapatinib in possible resistance pathways in
HER-2-positive patients previously treated with trastuzumab or anti-HER-2. In July 2017, the FDA approved lenatinib for extended postoperative treatment with trastuzumab adjuvant therapy in patients with early-stage
HER-2-positive breast cancer
[14].
Tucatinib is a new oral selective TKI that is more selective for
HER-2 than
EGFR [15]. Recently, the FDA announced the granting of a New Drug Priority Approval Application (NDA) for tucatinib in combination with trastuzumab and capecitabine for the treatment of locally advanced unresectable or metastatic
HER-2-positive breast cancer, including patients with brain metastases.
Pazopanib is a novel multitarget TKI that targets
VEGFR-1,
VEGFR-2,
VEGFR-3,
PDGFR/β, and
c-Kit, and has been shown to inhibit tumor growth and angiogenesis in in vitro studies
[16].
2.2. VEGF Inhibitor
TNBC is a highly proliferating tumor that requires the involvement of angiogenesis in its development, invasion, and metastasis processes. The expression level of vascular endothelial growth factor (
VEGF) is an independent prognostic factor in early breast cancer. High expression of
VEGF suggests high tumor malignancy, easy recurrence and metastasis, short disease-free survival, and low overall survival.
VEGF-A is highly expressed in TNBC cells, so TNBC is generally considered to be sensitive to VEGF-targeting drugs
[17]. Platelet-derived growth factor (PDGF) is one of the angiogenic factors. Abnormal activation of
PDGF or
PDGFR induces tumor angiogenesis and promotes migration and invasion of tumor cells. Overexpression of
VEGF in tumor tissues can promote the migration and proliferation of vascular endothelial cells, promote angiogenesis, increase tumor growth, and significantly increase vascular permeability, allowing tumor cells to enter the blood vessels for tumor invasion and metastasis, providing more favorable conditions
[18]. Currently, antiangiogenic drugs include bevacizumab and ramucirumab, and tyrosine kinase inhibitors such as sunitinib and sorafenib. These antiangiogenic drugs are likely to have higher antitumor activity in TNBC. In a phase II study of paclitaxel, bevacizumab, and gemcitabine in the treatment of
HER2-negative breast cancer, the clinical benefit rate in the TNBC subgroup was 84.6%, and approximately 82.5% of patients achieved a total survival of 18 months
[19]. In another phase III trial, RIBBON-2 in the TNBC subgroup, the bevacizumab combination group, and the chemotherapy group showed a reduction in the risk of disease progression by 51%. It also increased progression-free survival by 3.3 months and overall survival by 5.3 months
[20]. These results demonstrate the efficacy of antiangiogenic drugs in inhibiting tumors in TNBC.
2.3. PARP Inhibitor
PARP is a key enzyme for repairing broken DNA single strands, and in wild-type cells containing
BRCA1/2, broken DNA double strands can be repaired by homologous recombination. However, in BRCA1/2-deficient cells, the homologous recombination function fails and therefore PARP is relied upon to repair the broken DNA single strand. Therefore, PARP inhibitors have the ability to prevent self-repair in
BRCA1/2 mutated breast cancer cells and accelerate apoptosis of tumor cells, thereby enhancing the efficacy of chemotherapy as well as radiotherapy
[21].
As a novel drug, PARP inhibitors can cause double-strand breaks in BRCA-mutant breast cancer cells, leading to cell death due to synthetic lethality
[22]. Olaparib is an effective PARP-1 and PARP-2 inhibitor
[23] and was the first to be used by the FDA and the European Medicines Agency (EMEA) (Table 1). A PARP inhibitor was clinically approved for treatment of relapsed high-grade serous ovarian cancer. Olapani was first used in the clinical study of BRCA-related breast cancer,
ClinicalTrials.gov Identifier: NCT00494234. Patients received olaparib (100 mg/time, 2 times/d (
n = 27) and 400 mg/time, twice/d (
n = 27)), and the results indicated a dose-dependent objective response rate (ORR) of 22% (100 mg group) and 41% (400 mg group). The 400 mg group had a median progression-free survival of 5.7 months (95% CI: 4.6–7.4), and that of the 100 mg group was 3.8 months (95% CI: 1.9–5.5)
[24]. After a preliminary determination of the therapeutically effective dose, the OlympiAD study became the latest basis for FDA-approved olrapani, which included
BRCA-mutant HER2-negative advanced breast cancer patients. The study group (
n = 205) received olaparib (300 mg/second, 2 times/d monotherapy) while the control group (
n = 97) only received capecitabine, eribulin, or vinorelbine in three cycle regimen single-agent chemotherapy. The study showed that the study group and the control group were 7.0 and 4.2 months (HR = 0.58, 95% CI: 0.33 to 0.80,
p < 0.001). Therefore, the study indicated that olaparib has significant advantages in terms of efficacy and safety compared to other chemotherapeutic drugs
[25].
Table 1. Targets and targeted drugs for triple negative breast cancer.
| Signaling Pathway |
Target |
Target Drugs |
Ref. |
| DNA repair pathway |
PARP |
Olaparib, Veliparib, Rucaparib, Niraparib, Talazoparib |
[26] |
| EGF signaling |
EGFR |
Cetuximab, Panitumumab, Erlotinib, Gefitinib |
[27][28] |
| Angiogenesis pathway |
VEGF |
Bevacizumab, Ramucirumab, Sunitinib, Sorafenib |
[19][28][29] |
| PI3K/AKT/mTOR signaling |
PDGF, PI3K, mTOR |
Sunitinib, Sorafenib, BEZ235, Everolimus, Bez235 |
[29][30] |
| MAPK signaling |
ERK, mTOR |
Bez235, MEK162 |
[31] |
| JAK/STAT signaling |
JAK2 |
Ruxolitinib |
[32] |
| Cell cycle pathway |
CHK1 |
UNC01, AZD7762, PF477736, SCH900776, LY2606368 |
[33] |
| Src tyrosine kinase signaling |
Src |
Dasatinib |
[34][35] |
| Human epidermal growth factor receptor 2 |
HER2 |
Lapatinib, Neratinib, Tucatinib, Poziotinib, Pyrotinib, Trastuzumab, Pertuzumab |
[36][37][38][39][40] |
| Fibroblast growth factor receptor signaling |
FGFR |
Gefinitib, Afanitib, Erlotinib |
[41][42][43] |
Intractable TNBC requires more effective treatment strategies, so new drugs are being developed to sensitize sporadic TNBC to PARP inhibition. A clinical trial (
ClinicalTrials.gov Identifier: NCT01623349) is evaluating olaparib and BKM120 (buparlisib) or BYL719 (PI3 kinase inhibitor) in advanced sporadic TNBC and high-grade plasma ovarian cancer
[44]. Another clinical trial (
ClinicalTrials.gov Identifier: NCT01434316) combined veliparib and dinaciclib (CDK inhibitor), in which CDK inhibition was assumed to sensitize TNBC to PARP inhibition
[45]. After the phase I dose ascertainment portion, the study will enroll patients with
BRCA-mutated and nonmutated advanced breast cancer in a dose expansion cohort. Another PARP inhibitor, talazoparib (BMN 673), effectively modulates
PARP transcription. In addition, veriparib (veliparib), a novel potent PARP-1 and PARP-2 inhibitor, had a 51% partial remission rate in the treatment group in a clinical phase II trial of breast cancer treatment, which was significantly higher than the standard chemotherapy group (26%)
[46]. This study suggests that veriparib is more effective than standard therapy for TNBC with a high risk of recurrence. In addition, multicenter clinical trials are evaluating the efficacy and safety of PARP inhibitors in combination with chemotherapy, immunosuppressants, and inhibitors of other targets (e.g.,
VEGFR,
HSP90,
P13K/AKT/mTOR, etc.) for the treatment of TNBC
[47]. Regimens such as olaparib in combination with VEGFR or P13K inhibitors can effectively inhibit the rapid proliferation and growth of TNBC cells by affecting the blood oxygen supply to tumor cells and blocking molecules required for cell growth
[48].
2.4. CHK1 Inhibitor
Checkpoint kinase 1 (
CHK1) belongs to the serine/threonine protein kinase family and plays an important role in the regulation of cell cycle arrest by checkpoints caused by DNA damage or the presence of un-replicated DNA. The CHK1 inhibitor UNC01 abolished the production of G2 checkpoints dependent on DNA damage in cisplatin treatment and increased the sensitivity of cisplatin by nearly 60-fold. In addition, the TCGA database analysis found that the p53 mutation rate was very high in TNBC. The CHK1 inhibitor (UCN01 or AZD7762) increases the sensitivity of TNBC xenografts with
p53 mutations to chemotherapy and achieves good preclinical effects
[33]. These all indicate that
CHK1 is a very promising target in patients with p53 mutations in TNBC. Based on the recognition that CHK1 is a drug target, other CHK1 inhibitors, AZD7762, PF-477736, SCH900776, and LY2606368, are undergoing phase I and II clinical trials.
2.5. EGFR Inhibitor
EGFR is a transmembrane tyrosine kinase growth factor receptor involved in cell proliferation and differentiation in normal tissues. It also participates in the adhesion and movement of normal tissue cells and initiates many downstream cell signal transduction pathways
[49][50].
EGFR is a member of the
ErbB family of membrane tyrosine kinase receptors. Studies have shown that most TNBCs have overexpressed
EGFR, and early TNBC patients with
EGFR overexpression usually have worse overall survival and disease-free survival than normal expression patients. Therefore,
EGFR overexpression is often associated with poor prognosis in TNBC
[51][52]. Currently, the targeted therapeutic drugs that block the
EGFR signaling pathway in breast cancer research are mainly anti-EGFR monoclonal antibodies (cetuximab). A phase II open randomized clinical trial evaluated the efficacy of platinum citrate cetuximab in patients with TNBC. The combination group had a 10% increase in complete release rate compared with the single-agent group, progression-free survival was extended by 2.2 months, and overall survival was extended by 3.5 months, but cetuximab may cause adverse reactions
[28]. These cytological and clinical trials have demonstrated the enormous potential of EGFR inhibitors in the targeted therapy of TNBC.
2.5.1. Targeting of the PI3K/AKT/mTOR Signaling Pathway
Distortion of the
PI3K/AKT/mTOR signaling pathway is common in TNBC and has become a potential pathway for TNBC resistance to chemotherapy; phosphorylation of mTOR often suggests a poor prognosis in early TNBC
[53][54]. Everolimus is an oral mTOR inhibitor that inhibits the downstream signaling of mTOR molecules in cells and arrests the cell cycle in the G1 or S phase, thereby inhibiting
PI3K/AKT/mTOR pathway activity. A phase II clinical trial was performed in patients with TNBC (
n = 50) treated with paclitaxel fluorouracil + epirubicin + cyclophosphamide (T-FEC), followed by randomization into the everolimus group (
n = 23) and blank control group (
n = 27). The results showed that the recurrence rate in the two groups was 47.8% and 29.6%, respectively, and the pCR rate was 30.4% and 25.9%, respectively
[29]. Therefore, the study concluded that the pCR rate in patients with TNBC after combined treatment with everolimus did not improve significantly. In addition to monotherapy studies, a phase II neoadjuvant clinical study published in 2017 randomized TNBC patients into two groups; the study group (
n = 96) was administered everolimus combined with cisplatin + paclitaxel, while the control group (
n = 49) was administered placebo treatment. The results showed that the combination of everolimus with TNBC neoadjuvant therapy did not improve pCR or clinical response rates, but increased the incidence of adverse events
[55]. Thus, inhibition of RTK upstream of
mTOR leads to rebound activation of
AKT [56]; conversely, inhibition of
AKT activity initiates
FOXO-dependent transcription and RTK activation
[57]. Although blocking PI3K activity reduces
AKT activation, it also leads to enhanced
MAPK signaling
[58]. In conclusion, this evidence is a good theoretical basis for studying dual inhibitors of the
PI3K/AKT/mTOR pathway, which can control the activation of the target pathway and respond to its feedback loop, thereby preventing or delaying resistance. For example, combined mTOR and AKT inhibitors have shown synergistic efficacy in allograft models of basal-like patient origin
[59]. A number of PI3K/mTOR inhibitors continue to be developed clinically, including gedatolisib (PF-05212384), which has therapeutic activity in breast cancer and acceptable safety and tumorigenic activity
[60].
2.5.2. Targeting of MAPK Signaling Pathway
Excessive activation of
MAPK is involved in abnormal proliferation and apoptosis of cancer cells, which in turn contributes to TNBC malignancy. Ras and Raf mutations are infrequent in TNBC; instead, activation of
MAPK signaling pathways is often thought to be caused by multiple mechanisms of upstream receptor tyrosine kinase activation or by activation or mutation of upstream proteins, such as
PI3K/AKT/mTOR and related proteins
[61]. Previous studies have shown that flutamide and C1-1040 have synergistic effects in the trastuzumab model, causing a decrease in ERK phosphorylation levels during combination therapy with trastuzumab
[62]. One study found that ERK inhibitor in combination with Forskoli increased the sensitivity of TNBC cells to adriamycin
[63]. In addition, PAD1 is used to treat metastatic breast cancer by regulating
MEK1–ERKl/2–MMP2 signaling in TNBC
[64]. A growing body of preclinical evidence supports targeting the
Ras/MAPK cell signaling pathway in TNBC subtypes, although large genomic surveys (TCGA public database) suggest that typical mutations in this pathway are rare
[65][66]. Because of the early spread of TNBC, targeted treatment in the neoadjuvant setting may offer the effective therapeutic punch needed to eliminate micrometastatic disease and reduce mortality.
2.5.3. Targeting the JAK2/STAT3 Signaling Pathway
In patients with TNBC,
JAK1, and
JAK2 are usually overexpressed. The JAK/STAT pathway is involved in important biological processes such as cell proliferation, differentiation, apoptosis, and immune regulation. Phosphorylated
STAT3 is found in more than 50% of breast tumors and is associated with invasive phenotypes and poor prognosis
[67]. Previous studies have found that abnormalities in the
IL-6/JAK2/STAT3 pathway play a crucial role in TNBC
[68]. This suggests
JAK2/STAT3 to be a potential therapeutic target. The JAK1/2 inhibitor ruxolitinib is now approved for the treatment of myelofibrosis and is expected to improve the treatment of TNBC as a single-targeted drug
[69].
2.5.4. Targeting of the Src Signaling Pathway
Src tyrosine kinase is overexpressed in TNBC and is involved in disease progression. Preclinical studies have shown that the combination of Dasatinib and cisplatin, a combination of
Src,
c-kit, and
PDGFRβ, can inhibit the growth of basal-like tumor cells or TNBC tumor cells
[70][71][72]. In a phase II clinical trial using Dasatinib, anthracyclines, and paclitaxel in combination for treatment of metastatic TNBC, disease control rates were found to be 9.3%, partial response rates were 4.3%, and progression-free survival was around 8.3 weeks
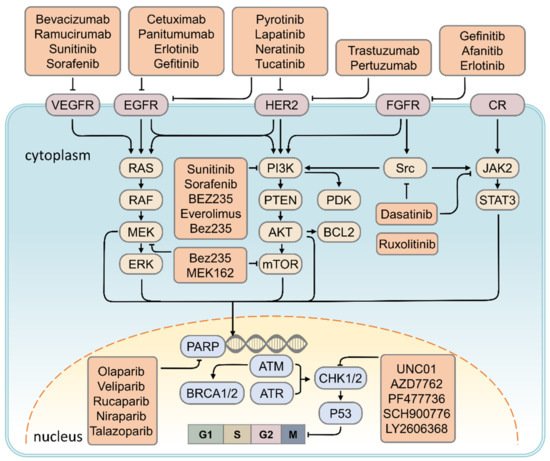
[34][35]. (Figure 2)
Figure 2. Schematic drawing presents the detail signaling pathways of breast carcinogenesis. All signaling pathways represented in this diagram have been found to be operative in the breast cancer cell context, whereas evidence to date supports a predominant role of HER2 in BRCA1/2 signaling and EGFR–PARP cross-talk in breast cancer cells. CR: cytokine receptor.