1. Olive Leaves
Olive leaves represent an agricultural waste by-product obtained during the harvesting of olive trees for table olives and olive oil production chains
[1][2]. It has been estimated that pruning alone produces annually 25 kg of waste represented by branches and leaves per olive tree
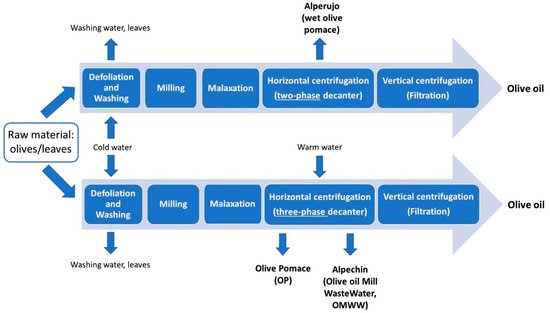
[3]. A considerable amount of olive leaves is also discarded during the olive drupes washing process at the beginning of the olive oil production chain (see ), reaching a value ranging from 8%
[4] to 10%
[5] of the total weight of olives subjected to milling. In general, olive leaves are not included in the definition of olive mill solid waste
[6] but, together with olive stones, they are described as solid residue
[7]. The use of solid residue is of great economic and social importance for the Mediterranean area, as it is accumulated in large amounts
[7]. Although limited by their pungency, olive leaves are traditionally used in many countries as feed for livestock or simply disposed by burning. Olive leaves represent a potential source of bioactive compounds, as indirectly proved by the use of their extracts in the context of folk medicine for centuries, with the first medicinal application attested in history dating back to the ancient Egyptian civilization
[1]. During centuries, the use of olive leaves preparations in traditional medicine has spread in many different countries
[8] (see ). Recently, preparations based on olive leaves, in the form of liquid extracts or tablets, have been commercialised as natural supplements against diabetes, high blood pressure, cardiovascular diseases, urinary tract infections, chronic fatigue symptoms and to improve immune system
[9]. Olive leaves preparations also find their application in the cosmetic industry for their anti-ageing activity, and due to their antibiotic and antiparasitic properties as supplements for animal health
[10]. Moreover, olive leaves extracts may be used as additives to increase food shelf-life, safety and functionality for their antioxidant and antimicrobial features
[11][12]. Interestingly, their probiotic properties have been recently found to promote the
Lactobacillus casei survival during cold storage of cheese
[13]. Their addition to beer
[14] and to a Southern Italy traditional cereal-based baked product known as
taralli [5] has been found to increase the inherent polyphenols content of these products. It is also worth noting that phenolic compounds and tocopherols, normally found in olive oils, play a protective role against oxidative stress
[15] and are able to extend the extra virgin olive oil shelf-life due to their antioxidant properties
[16]. Since the use of synthetic antioxidants may lead to health risks, recent papers have featured that olive leaves, having a high antioxidant activity
[17][18][19] due to their phenolics, can exhibit strong preventive effects against olive oil oxidation. The addition of olive leaves during oil extraction process, specifically during olive milling, has thus been recently evaluated in detail and found to lead to the enrichment in chlorophyll
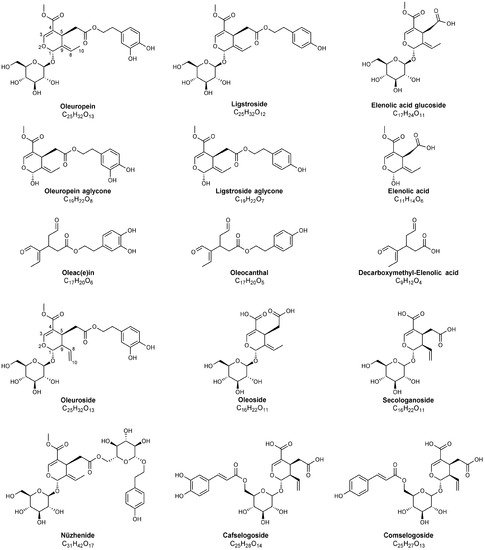
[20][21], carotenoids, flavonoids, and in the derivatives of oleuropein, which is the main secoiridoid occurring in olive leaves (see )
[22].
Figure 1. Layout of the main steps of olive oil production involving two- or three-phase horizontal centrifugation and the main by-products obtained.
Table 1. Main uses of olive leaves in traditional medicine occurring worldwide.
| Country |
Assumption Form of Olive Leaf-Based Products |
Route |
Treatments |
References |
| Arabic countries |
Dried plant fumigation |
Nasal |
Abortifacient, and treatment of cystitis and sore throat |
[23] |
| Brazil |
Herbal tea of the fresh leaves |
Oral |
To induce diuresis, and treatment of hypertension |
[24] |
| Canary Islands |
An infusion prepared from the fresh or dried leaves |
Oral/rectal |
Treatment of diabetes; hypertension and haemorrhoids |
[25] |
| France |
Powdered dried leaves as hard capsules |
Oral |
To promote urinary and digestive elimination functions |
[26] |
| Germany |
Extract with ethanol 96% v/v as liquid or coated tablet |
Oral |
Treatment of atherosclerosis and hypertension |
[26] |
| Italy |
Infusion of the dried leaf
Infusion of the fresh leaf |
Oral |
Treatment of hypertension and anti-inflammatory; for wound healing, emollient for ingrown nails and to restore epithelium |
[27][28] |
| Morocco |
Leaves and essential oil from the leaves |
Oral
Topical |
Treatment of stomach and intestinal disease and as a mouth cleanser; treatment of constipation and liver pain; tonic for hairs |
[29] |
| Trinidad |
Hot water extract of the leaf |
Oral |
To increase milk supply of nursing mother |
[30] |
| Ukraine |
Hot water extract of dried plant |
Oral |
Treatment of bronchial asthma |
[31] |
The promising antimicrobial and antioxidant hallmarks of phenols contained in olive leaves by-products have also led to produce bio-active films for food packaging by integrating olive leaf extract or powder in plastic polymers
[32][33].
Figure 2. Chemical structures of major secoiridoids identified in olive leaves, pomace and in olive oil mill wastewater using MS techniques. Note that hemiacetalic forms of elenolic acid and of oleuropein and ligstroside aglycones are reported; dialdehydic open-structure isoforms are indicated for oleac(e)in, oleocanthal and decarboxymethyl-elenolic acid. The carbon atoms numbering typically adopted for secoiridoids has been reported for oleuropein and oleuroside to emphasize the positional isomerism occurring for these compounds, related to the position of the exocyclic C=C bond.
2. Olive Pomace and Olive Oil Mill Wastewater
According to Decision 2000/532/EC of the European Union Commission and to the Eurostat database classification
[34], waste from agricultural activities (i.e., corn, wheat, fruit, vegetables, rice, pomace, olive wastes) are included in the agricultural livestock waste (ALW) category. Just to give an idea of the amount of ALW in 2016 resulting from olive oil production, 22 and 14 thousands of tons of olive pomace were generated, respectively, in Apulia and Sicily, two of the major olive oil producers in Italy
[35]. Such amounts are not surprising, since approximately 800 kg of pomace and 200 kg of wastewater are on average obtained for 200 kg of extra virgin olive oils. To avoid endangering human health and to protect the environment, a rational waste management including prevention, reuse, recycling, recovery, and disposal is crucial also for such by-products
[36].
As previously mentioned, the type of horizontal centrifugation adopted during olive oil production can determine the main features of OP, which is humid and semisolid, with a moisture content between 50 and 70%, when two-phase decanters are used, whereas moisture is decreased if three-phase decanters (35–40%) or traditional press mills (20–25%) are employed
[37]. The average rough composition of OP has been reported as follows: water (60–70%), lignin (13–15%), cellulose and hemicellulose (18–20%), olive oil retained in the pulp (2.5–3%) and mineral solids (2.5%)
[38]. Major organic compounds are sugars (3%), volatile fatty acids (C2–C7) (1%), poly-alcohols (0.2%), proteins (1.5%), polyphenols (0.2%) and pigments (0.5%)
[38]. The profile vitamin E occurring in OP includes α-tocopherol, β-tocopherol, α-tocotrienol and γ-tocopherol, being α-tocopherol the major form (>2.6 mg/100 g). The lipid fraction is particularly rich in oleic acid (ca. 75%), followed by palmitic, linoleic, and stearic acids; hydroxytyrosol and comselogoside represent around the 80% of the total phenolics in OP
[39].
Since OP consists essentially of olive pulp, olive stone and vegetation water, it includes many of the valuable components of the olive fruit so it can be subjected to biorefining, i.e., to the extraction of valuable compounds and energy. OP is mostly used for the recovery of pomace oil by solid-liquid extraction with hexane, followed by distillation and solvent recycling. The crude oil is then refined and typically blended with a small amount of virgin olive oil. Furthermore, after OP is evaporated and thermally concentrated, it can be applied to cultivated soils as herbicide, insecticide, and compost. Indeed, due to the polysaccharide occurrence, dried OP represents a potential source of gelling pectic material. As an example, Lama-Munoz et al. isolated sugars from
alperujo by ethanol/water precipitation, obtaining various oligosaccharide fractions as pectin, neutral and acidic xylo-oligosaccharides, and xyloglucan oligosaccharides with diverse applications as gelling agents, stabilizers and emulsifiers for the food industry
[40].
Alperujo compost at different doses was evaluated by Tortosa et al.
[41] as an organic manure, mixed with a minimal amount of inorganic fertilizers, for pepper growth in greenhouse cultivations. The authors speculated that the organic matter from
alperujo compost positively affects pepper oxidative metabolism by increasing the antioxidative enzymatic activities. Hence, the yield and physiological growth of plants would be improved or rather comparable to standard nutrient solutions. OP is also considered a source of bioethanol, biogas, and methane
[42]; residuals of olive stones in OP can be the substrate to obtain activated carbon that can be employed as fuel for the generation of heat and electricity
[7]. Moreover,
alperujo oil has been used as a non-edible substrate with a high-free fatty acid content for the synthesis of biodiesel through an enzymatic path based on recombinant 1,3-positional specific
Rhizopus oryzae lipase, avoiding the generation of glycerol as a co-product
[43]. Another fascinating alternative of OP valorization is its use as a substrate for growing microorganisms of biotechnological interest
[44]. This strategy offers the opportunity of producing high value-added products, such as enzymes, biopolymers, and pigments with a concomitant reduction of organic wastes. Ghilardi et al. explored the possibility to produce carotenoids by a strain of
Rhodotorula mucilaginosa using different media derived from
alperujo as a cheap substrate
[44]. According to the medium used, it was possible to obtain a mixture of carotenoids enriched in torulene, torularhodin and/or neurosporene, thus showing that
alperujo can be used to produce carotenoids exploitable at industrial scale, as additives in pharmaceutical, food and feed products
[44].
OP has been applied even in the construction field. De la Casa et al.
[45] reported the addition of
alperujo to the ceramic paste of bricks, lowering density and thermal conductivity. The OP residue was also used as a substitute for clay in 1.25, 2.5 and 5% (wt) of artificial lightweight aggregate manufactures
[46]. The results indicated that the addition of OP in the mixture is beneficial in terms of environmental impact compared to that related to aggregates made with clay.
Besides current uses, including composting, soil amendment, animal feed, OP can be considered a valuable source of bioactive substances with well-recognized benefits for human health and well-being. As aforementioned, it is rich in phenolic compounds and triterpenic acids, for which numerous biological activities, including anti-inflammatory, antitumor, antimicrobial, antioxidant, antidiabetic, and cardio-protective activities, have been reported
[47]. Among bioactive phenols, hydroxytyrosol (HT) stands out among compounds with highest added value that can be recovered from the solid by-product, due to its high oxidative stability and antioxidant activity and it is currently used as a therapeutic agent, dietary supplement or natural ingredient in food and feed industries. For instance, various contents (65–195 μg) of HT were added to 100 g of fresh prepared mayonnaise and several quality parameters as free acidity, peroxide value and concentration of conjugated dienes, as well as the content of polyphenols and squalene in the lipid fraction
[48], were evaluated
[49]. The authors demonstrated that HT improves the mayonnaise hydrolytic stability reducing the formation of oxidation by-products during storage at room temperature and in the dark up to four weeks. Antioxidant-rich extracts from olive mill pomace related to tree different cultivars of Southern Italy (
Nocellara,
Roggianella and
Carolea) were dissolved in a commercial pear juice using inulin as a carrier system of bioactive compounds
[50]. In vitro evaluation highlighted outstanding antioxidant features of fortified juice in terms of both antioxidant and scavenging performances, representing an attractive source of functional foods. Very recently, Di Nunzio et al.
[51] prepared bakery products (biscuits and breads) using a variety of flours and fermentation protocols also enriched with defatted OP. To assess the effects in a biological system, the digested fractions were supplemented to intestinal cultured enterocytes cells, used as model system, and the secretion of cytokines was measured. OP extracts were also marketed as feed additives, especially fish feed
[52], while an olive oil bioactive extract was tested in fish diet, favouring growth of rainbow trout and gilthead sea bream
[53]. Other important examples of the use of by-products for functional and food applications have been summarized by Nunes et al.
[54] in a more focused review.
As shown in , OMWW (also called
alpechin) is a secondary product of the olive oil extraction process generated from three-phase decanters. It has a distinctive odour, pH = 4.0–5.5, and high electrical conductivity (6000–16000 mS/cm); it also contains large amounts of suspended solids and high concentrations of polyaromatic compounds
[55]. Despite the fact that three-phase centrifugation causes the reduction of natural antioxidants in olive oil and a considerable volume of OMWW, it is currently largely used for oil production, having high working capacity and enabling the automation of industrial plants, leading to a lessening of manual labour and olive-processing costs
[56]. The physicochemical features of OMWW largely depends on the oil extraction and processing methods, climate, as well as olives maturity, cultivar and origin
[57][58][59]. From the chemical composition point of view, OMWW consists mainly of water (80–92%), and contains 3–15% organic matter (olive oils, carbohydrates, lipids, pectin, organic acids, polysaccharides, phenols, tannins, and lignin) and minerals
[60]. Phenolic compounds, sugars, and organic acids are the main components of the OMWW organic fraction, while, among minerals, the potassium ion content is relatively high (see in Ref.
[61]). Long-chain fatty acids contained in OMWW represent a pollution concern, being toxic to soil micro-organisms and plants
[57] it is considered the most polluting waste generated by the agri-food industries
[60]. Although it has been spread for many years into soil or nearby streams and rivers, OMWW can be very harmful to soil microflora, plants and freshwater species
[62] since it is characterized by high values for key pollution parameters, such as biological oxygen demand (BOD
5, 40–95 g/L) and chemical oxygen demand (COD, 50–180 g/L) and high concentrations of phenols and flavonoids (from 0.5 to 24 g/L), having strong antimicrobial and phytotoxic properties
[63]. At present, more than 50 different phenolic compounds, in particular hydrophilic species such as phenolic alcohols and acids and, at in a minor amount, secoiridoids, have been identified in OMWW
[64][65][66][67]. Due to its inherent features and with the aim of giving value to a waste, different employments of OMWW have been evaluated in the last 30 years.
The effects of spreading OMWW on soil properties and crops was reviewed by Barbera et al.
[68]; within the European Union each State has established different limits (for example, in Italy the legal limit is 80 m
3/ha) and the authors, by using an holistic approach, concluded that direct application of OMWW exerts a temporary positive effect on soil physical properties, if some outlined restrictions based on soil characteristics are applied. Moreover, they emphasized that polyphenols are the most limiting factor for spreading OMWW on soils because of their antimicrobial and phytotoxic effects
[68].
Due to the well-known beneficial effects of phenols contained in OMWW, its potential application for the preparation of functional beverages has been also explored
[56]. However, some issues were evidenced, such as the stability of bioactive constituents during preservation procedures, processing, and storage that require appropriate thermal and light-exposure conditions. Moreover, as already discussed, the OMWW composition largely depends on specific factors, which makes a “standardization” of its phenolic extract not straightforward. Olive oils enriched with phenolic compounds extracted from OMWW have been also proposed
[69][70][71]; despite further studies are needed, this is an intriguing example of the possibility to create added value by using a waste of olive oil production. As reviewed by Caporaso et al.
[71], a similar approach was used for milk and derivatives and also for meat-based products, such as lard and hamburgers.