1000/1000
Hot
Most Recent

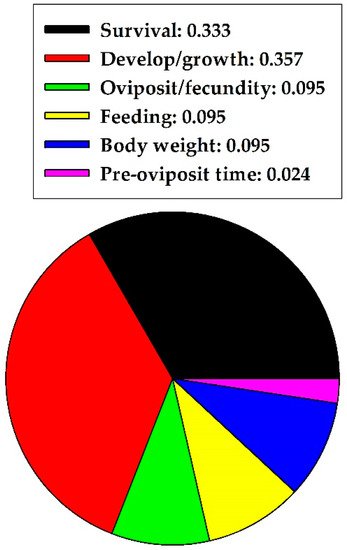
Quercetin is one of the most abundant flavonoids in terrestrial plants and pollen. In living plants, quercetin can function as a secondary metabolite to discourage insect herbivory. Literature on insect-quercetin interactions was searched and data synthesized to test the hypothesis that quercetin can become an effective biocide to reduce herbivory. The USDA, National Agricultural Library, DigiTop Navigator platform was used to search the literature for harmful versus nonharmful effects of quercetin on insect behavior, physiology, and life history parameters. Quercetin effects were evaluated on herbivores in five insect orders. Quercetin was significantly more harmful to Hemiptera, Diptera, and Lepidoptera but significantly more nonharmful to Coleoptera. Harmful and nonharmful effects to Orthoptera were indistinguishable. Quercetin had significantly more harmful (than nonharmful) effects on herbivores when data from the five insect orders were combined. Quercetin concentration (mg/mL) did not significantly affect these results. This study suggests that quercetin could prevent herbivory but field experiments are necessary to substantiate these results.
| Category | Bioassay Method | 1 Effects on Behavior and Life History | 2 Effective Concn. | Reference |
|---|---|---|---|---|
| Herbivore: Hemiptera; true bugs |
||||
| Macrosiphum rosae, nymphs and adults (Aphididae) |
Treated red rose (Rosa) foliage | Survival (−−) | 1 mg/mL | [1] |
| Acyrthosiphon pisum, nymphs and adults (Aphididae) |
In artificial diet | Development (−−), Pre-oviposition time (−−), Fecundity (−−), Survival (−−) |
1–10 mg/mL, 0.1–10 mg/mL, 1–10 mg/mL, 0.01–10 mg/mL |
[2] |
| Sitobion miscanthi, adults (Aphididae) |
Innate resistance in wheat ears in field | Fecundity (−−) | 0.199 mg/mL | [3] |
| Tupiocoris notatus, nymphs (Miridae) |
Treated tobacco (Nicotiana) leaves | Attractancy (++) | 0.09 μg | [4] |
| Herbivore: Coleoptera; beetles |
||||
| Callosobruchus chinensis, eggs and adults (Bruchidae) |
On filter paper and in plastic jar | Survival (−−), Oviposition (−−) |
5.0 mg/mL, 5.0 mg/mL |
[5] |
| C. chinensis, adults | On glass beads | Oviposition (oo) | 0.001–1.0 mg/mL | [6] |
| Tribolium castaneum, adults (Tenebrionidae) |
On wheat wafer discs | Feeding (−−) | 2.0 mg/mL | [7] |
| Melolontha melolontha, larvae (Scarabaeidae) |
In potted soil, in field | Survival (oo) | 20.0 mg/mL | [8] |
| Popillia japonica, adults (Scarabaeidae) |
In artificial diet | Feeding (++) | 30.2 mg/mL | [9] |
| P. japonica, adults | In artificial diet | Feeding (++) | 0.302–3.02 mg/mL | [10] |
| Carpophilus hemipterus, larvae and adults (Nitidulidae) |
In artificial diet | Feeding (++) | 0.025 mg/mL | [11] |
| Leptinotarsa decemlineata, larvae (Chrysomelidae) |
In artificial diet plus insecticide | Survival (−−) | 0.1 mg/mL | [12] |
| Phaedon brassicae, adults (Chrysomelidae) |
Treated filter paper | Feeding (−−) | 3.02 mg/mL | [13] |
| Oulema oryzae, adults (Chrysomelidae) |
Treated filter paper | Feeding (−−) | 3.02 mg/mL | [13] |
| Plagiodera versicolora, adults (Chrysomelidae) |
Treated filter paper | Feeding (++) | 3.02 mg/mL | [13] |
| Altica oleracea, adults (Chrysomelidae) |
Treated filter paper | Feeding (+ +) | 3.02 mg/mL | [13] |
| Altica nipponica, adults | Treated filter paper | Feeding (++) | 3.02 mg/mL | [13] |
| Anthonomus grandis, larvae and adults (Curculionidae) |
In artificial diet | Feeding (oo), Oviposition (oo), Body weight (++) |
1–10 mg/mL, 1–10 mg/mL, 6 mg/mL |
[14] |
| A. grandis, adults | Treated filter paper | Feeding (++) | 0.5 mg/mL | [15] |
| Epilachna paenulata, larvae (Coccinellidae) |
Treated squash (Curcubita) leaves | Feeding (++), Survival (−−) | 0.01 μg/cm2, 10–100 μg/cm2 |
[16] |
| E. paenulata, larvae | Treated squash (Cucurbita) leaves | Feeding (oo), Body Weight (oo), Survival (oo) |
0.1–50.0 μg/cm2, 0.1–50.0 μg/cm2, 0.1–50.0 μg/cm2 | [16] |
| Herbivore: Lepidoptera; moths/butterflies |
||||
| Helicoverpa armigera, larvae (Noctuidae) |
In artificial diet; leaf-dip toxicity test | Development (−−), Pesticide sensitivity (oo) |
0.1% (w/w), 0.1% (w/w) |
[17] |
| Spodoptera litura, larvae (Noctuidae) |
Toxicity test | Development (−−), Survival (−−) |
0.005 mg/mL, 0.005 mg/mL |
[18] |
| Helicoverpa armigera, larvae (Noctuidae) |
In artificial diet | Development (−−), Survival (−−), Pesticide sensitivity (oo) |
16 mg/g, 16 mg/g, 16 mg/g |
[19] |
| Helicoverpa armigera, larvae (Noctuidae) |
Ingested with liquid solution | Development (−−), Survival (−−) |
3 mg/g, 3 mg/g |
[20] |
| Spodoptera frugiperda, larvae (Noctuidae) |
Treated foliage (Lettuce) | Feeding (++), Feeding (−−) |
0.01 μg/cm2, 100 μg/cm2 |
[21] |
| Chilesia rudis, larvae (Arctiidae) |
Treated foliage (cultivated Murtilla) | Feeding (++) | 0.005 mg/mL | [22] |
| Lymantria dispar, larvae (Lymantriidae)(from Quercus forest) |
In artificial diet | Survival (−−), Body weight (−−) |
2% (w/w), 2% (w/w) |
[23] |
| Bombyx mori, larvae (Bombycidae) |
In artificial diet | Body weight/Weight gain (−−) | 0.1% (w/w) | [24] |
| Ostrinia nubilalis, larvae (Pyralidae) |
In artificial diet | Development (−−) Survival (−−) |
1 mg/g | [25] |
| Heliothis virescens, larvae (Noctuidae) |
In artificial diet | Development (−−) | 0.25% (w/w) | [26] |
| Heliothis virescens, larvae Helicoverpa zea, larvae (Noctuidae) |
In artificial diet | Body Weight (−−) Feeding (oo) |
0.10% (w/w) | [27] |
| Pectinophora gossypiella, larvae Heliothis virescens, larvae Helicoverpa zea, larvae (Noctuidae) |
In artificial diet | Body Weight (−−) Development (−−) |
0.10% (w/w), P. gossypiella; 0.10% (w/w), H. virescens; 0.20% (w/w), H. zea |
[28] |
| Heliothis virescens, larvae Helicoverpa zea, larvae (Noctuidae) |
In artificial diet | Development (−−) Survival (−−) |
0.20% (w/w), H. virescens; 0.80% (w/w), H. zea | [29] |
| Herbivore: Diptera; true flies |
||||
| Bactrocera cucurbitae, adults (Tephritidae) |
On substrate (pumpkin) | Oviposition (−−) | 0.125 mg/mL | [30] |
| B. cucurbitae, eggs, larvae, and pupae | Dipped in test solution | Development (−−), Development (−−), Development (−−) |
3.125 mg/mL, 0.125 mg/mL, 0.005 mg/mL |
[31] |
| Rhagoletis pomonella, larvae (Tephritidae) |
In artificial diet | Development (−−) | 1.0 mg/mL | [32] |
| Drosophila melanogaster, larvae (Drosophilidae) |
In artificial diet | Development (++) | 1.75% (w/w) | [33] |
| D. melanogaster, adults | In artificial diet | Fecundity (++) | 5% (w/w) | [34] |
| Lycoriella pleuroti, larvae (Sciaridae) |
In artificial culture media | Survival (−−) | 0.1–0.3% (w/w) | [35] |
| Herbivore: Orthoptera; grasshoppers |
||||
| Calliptamus abbreviatus, nymphs (Acrididae) |
Sprayed on alfalfa foliage, field cages | Development (−−) Survival (−−) |
0.10 mg/mL | [36] |
| Oedaleus asiaticus, nymphs (Acrididae) |
Sprayed on natural host plant foliage, field cages | Development (−−) Survival (−−) |
0.10–10 mg/mL | [37] |
| Melanoplus sanguinipes, nymphs (Acrididae) |
In artificial diet | Body weight (oo) Survival (oo) |
0.125–4.0% (w/w) | [38] |