1000/1000
Hot
Most Recent

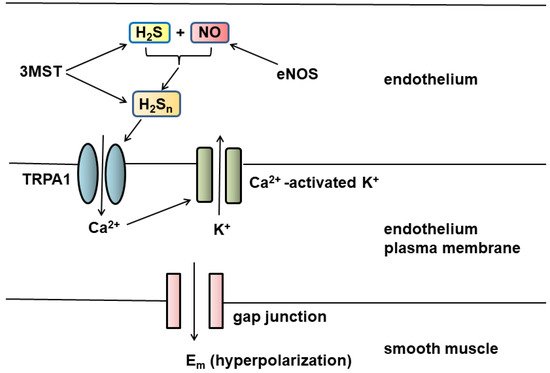
We discovered H2S as a signaling molecule which is produced by enzymes to modulate the synaptic transmission and relax vasculature. The cytoprotective effect, anti-inflammatory activity, energy formation, and oxygen sensing by H2S have been subsequently demonstrated. Two additional pathways for the production of H2S with 3-mercaptopyruvate sulfurtransferase (3MST) from l- and d-cysteine have been identified. We also discovered that hydrogen polysulfides (H2Sn, n ≥ 2) are potential signaling molecules produced by 3MST. H2Sn regulate the activity of ion channels and enzymes, as well as even the growth of tumors. S-Sulfuration (S-sulfhydration) proposed by Snyder is the main mechanism for H2S/H2Sn underlying regulation of the activity of target proteins.
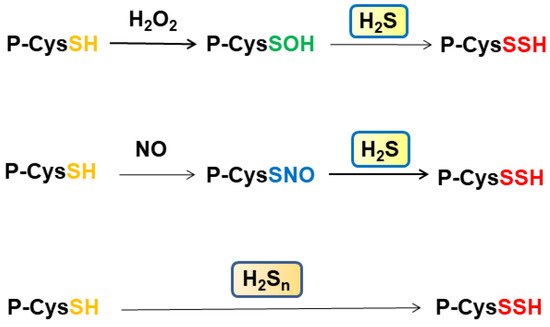
Some cysteine residues are oxidized by H2O2 to generate S-nitrosylated cysteine residues, and some others are S-nitrosylated by NO. These oxidized cysteine residues are S-sulfurated by H2S rather than H2Sn (Figure 2). Cys150 and Cys156 of GAPDH may be in the different oxidation state as described previously [26][64]. Zivanovic et al. demonstrated that the activity of manganese superoxide dismutase is suppressed through S-sulfenylation by H2O2, while the activity is recovered by H2S, which S-sulfurates the S-sulfenylated cysteine residues [65]. The same group showed that epidermal growth factor (EGF) activates its receptor in which the levels of S-sulfenylated cysteine residues are increased at the early phase, and those of S-sulfurated residues are increased at late phase when the expression of H2S producing enzymes is enhanced. H2S S-sulfurates those S-sulfenylated cysteine residues to regulate their activity (Figure 2).
Another role of S-sulfuration is that it enables proteins to recover their functions from over-oxidization. Sulfinic (Protein-CysSO2H) and sulfonic acids (Protein-CysSO3H) are not reduced back to Protein-CysSH by thioredoxin and deteriorate the protein function. In contrast, S-sulfurated proteins P-CysSSO2H and P-CysSSO3H can be reduced by thioredoxin to P-CysSH [65][66].