3.1. An Integrated Monitoring Physical Model for Legionella Study and Control in Real Systems
One feasible approach to gain this knowledge, while tracking operational features of the systems, is the combination of complementary monitoring methods, which include (a) online, continuous information and discrete sensing; (b) surface and water monitoring; (c) biofilm and Legionella analysis. Even though the development of such an idea can follow different pathways and certainly requires wider scientific reflection/discussion, we propose, for illustration purposes, an integrated monitoring model for Legionella study at field-based systems (Figure 1). This model aims to catalyze a joint discussion on a renewed Legionella management strategy, which can be optimized under the scope of field studies for later adoption at water utilities. Here, we will only focus on the macro perspective of the model rather than on overviewing specific methodologies, since those will depend on several items, including the sort of water system under study.
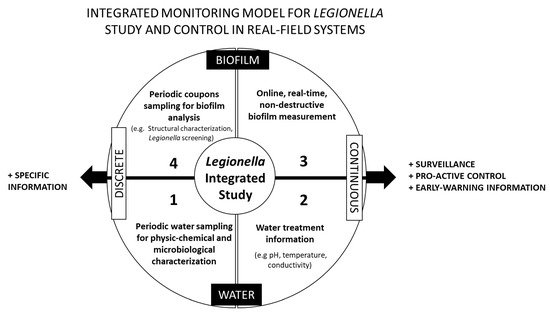
Figure 1. Integrated monitoring conceptual model for Legionella study and control in field-based systems. The model proposes four complementary sets of information: water (1 and 2) and biofilm (3 and 4) monitoring, discretely sampled (1 and 4) and continuously measured (2 and 3). Continuous information will enhance pro-active control and surveillance, based on early-warning information, while discrete information will allow to gain more specific information about Legionella ecology.
The conceptual model proposed in Figure 1 relies on the idea that Legionella control will be as effective as we manage to gain a broader perspective on the overall ecology of Legionella. Surveillance and pro-active control driven with online, continuous measurements are essential for effective Legionella mitigation practices, and specific information is key for enhancing understanding about Legionella overall ecology. Under these assumptions, four complementary sets of information were foreseen.
The first set of information is related to the routine monitoring approach, focused on periodic water sampling for physical, chemical, and microbiological characterization. This also includes
Legionella spp. and
L. pneumophila detection and quantification. Recently, Walker et al.
[83] reviewed current
Legionella testing methods, and LeChevallier
[84] proposed an interesting guidancefor the development of a
L. pneumophila monitoring plan for water utilities. Both works are of great importance to the implementation of improved routine
Legionella monitoring procedures. Furthermore, given the role of protozoa in the overall
Legionella ecology and virulence
[47], it seems to be very important to include their analysis under this first level of monitoring. This also embraces with the findings of Shaheen et al.
[85], who suggest that monitoring free-living amoebae can be useful to predict the ‘possible imminent high occurrence of
Legionella’ in engineered water systems. Protozoa are not detected through traditional bacteriological methods, and the detection of a large diversity of free-living protozoa can be a challenging and laborious task
[58]. This is demonstrated, for example, in the work of Valster et al.
[86], who found that different protozoan communities developed in duplicated samples (samples from different water settings). Nisar et al.
[87] discuss the relevance of molecular techniques such as PCR and fluorescence in situ hybridization (FISH) for
Legionella and protozoan screening in environmental water samples. In this work, the authors also came across the conclusion that, in potable water systems (including hospitals),
Vermamoeba and
Acanthamoeba were the hosts predominantly associated with
L. pneumophila. This also raises the possibility of selecting some specific protozoa indicators that might be linked to
L. pneumophila. For example, the review conducted by Lau et al.
[30] might be a great starting point for this discussion, since it systematizes the protozoa species (mostly amoebae) found to host
Legionella species in drinking water settings.
The second set of information is related to standard water treatment parameters that will directly or indirectly reflect the performance of the control measures
[88], including, for example, pH, conductivity, temperature, flow, critical pumps operation, and biocidal residue (if applicable). This also aligns with the WHO (World Health Organization) guidelines
[23], which state that ‘operationally, control measures, (…) should be monitored online’. The need to reinforce operational monitoring is also stressed in the recently revised European Directive (2020/2181) on the quality of water for human consumption
[26]. An online, real-time dataset of these parameters enables the timely identification and correction of punctual deviations to the established operational limits
[23][26][89], avoiding situations that can favor
Legionella proliferation. For example, Whiley et al.
[90] reported real-time monitoring of the temperature and flow in the thermostatic mixing valves of water distribution networks as an interesting surveillance strategy to detect changes in water quality, as well as to identify hazardous situations regarding different opportunistic pathogens, including
Legionella. This continuous information would be an important complement to well-established water routine sampling, as discussed in previous sections since it raises the opportunity to keep continuously an eye on the system in between samplings and while microbiological analysis is being processed. This information would also serve for registration purposes (an essential asset of a proper
Legionella prevention plan)
[24].
As formerly discussed, the potential of online, continuous, non-destructive biofilm monitoring can be determinant to establish a proactive, informated-based water management
[34]. Flemming
[34] systematized the features of an ideal online, real-time biofouling monitoring sensor able to provide information about the biofilm: location and extent, quantity (mass, thickness), nature of the deposit (organic/inorganic, biological/non-biological, chemical composition), the kinetics of deposit formation, and removal. Additionally, such monitoring tools should be applied to a large monitoring area and should be low cost and easy to handle. Due to this long and very specific list of features, it is very unlikely that a unique sensor meets all these requirements at once. As such, combining different monitoring tools into an ‘all-in-one’ solution is probably the most feasible way to strengthen the arguments for their routine implementation. This ‘all-in-one’ setup should combine a selection of tools that are suitable for real-field operation and that provide distinct (but complementary) output information about biofilm deposits.
Regarding
Legionella prevention, it seems plausible to accept that both the biofouling extent and nature (biotic/abiotic) of the attached layers are important parameters to assess. Measuring biofilm build-up/removal kinetics can provide important insights on ‘how fast is the biofilm being formed/removed’ and ‘how far will the stabilization plateau be achieved’. This concept is somewhat similar to the ‘Biofouling Formation Potential’ described by van der Kooji et al.
[61], yet applied to a different measuring unit. Those two indicators (kinetics and maximum biofilm amount) will provide information about the biofilm formation potential of the system and the biofilm extent, respectively. Both the ‘stabilization plateau’ and ‘threshold of interference’
[91], as well as biofilm kinetics, depend on the particular water system and its specific operating conditions
[34]. As such, for a given system, at a given representative location, an increased build-up rate or an unexpected sloughing-off event (which can bring
Legionella back into the bulk water) are certainly examples of early-warning calls that something in the standard operation has changed (even though that can be a planned change). Similarly, removal rates can be used to assess the efficacy of implemented countermeasures. For example, Pereira et al.
[92] reported the use of a surface sensor technology
[93] to monitor in real-time the formation/removal of biofouling layers, identifying proactively processual changes in the bypass of a cooling water system.
Evaluating the nature (biotic/abiotic) of the biofilm layer can be important for assessing and adjusting the efficacy of microbial control programs
[79], with the aim of keeping microbial growth at the surface under control. For example, the commercially available Alvim sensor
[94]—an online, electrochemical sensor—was successfully used in industrial water settings to follow the biofilm growth and to optimize cleaning procedures. Monitoring the nature of the deposit will be particularly relevant in finding out how biotic and/or abiotic attached layers affect
Legionella persistence. Another promising tool is the OnGuard
TM analyzer, which has been successfully used to optimize the biocidal program of a cooling water system, based on the detection of biofouling formation/removal kinetics
[95]. This analyzer can also provide information about the nature of the attached deposit
[95].
To gain detailed information that can enhance
Legionella ecology understanding, surface online monitoring must be complemented with biofilm discrete sampling, followed by a detailed analysis and characterization, including
Legionella screening. For that, the inclusion of biofilm sampling probes (or coupons), which can be periodically removed over time, might be a suitable approach. Some overviews on biofilm formation devices suitable for industrial application can be found in the works of
[96] or
[97], for example. Some interesting solutions for biofilm formation studies are the Flow Cell system
[93][98][99] or the Modified Robbins Devices
[100], which are very well characterized in the laboratory in terms of operation and hydrodynamics and have been successfully used in the study of biofilms in full-scale water systems.
The work of Azeredo et al.
[32] is a good starting point to choose which analytical techniques for biofilm characterization best fits a study’s purposes. Apart from the standard methods focused on biofilm physiology and the composition of the attached layers, we emphasize the role that structural characterization plays in the control of
Legionella. Several arguments support this suggestion: (a) protozoa have a significant impact on ‘shaping’ biofilm architectures
[58], (b) biofilm structure affects the efficacy of countermeasures
[101], (c) sloughing-off events are more likely to occur when heterogeneity increases
[102]. As such, evaluating structural changes in real-field systems can inform on biofilm and protozoa interactions, with a visible influence on
Legionella control.