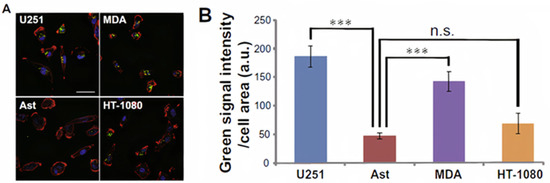
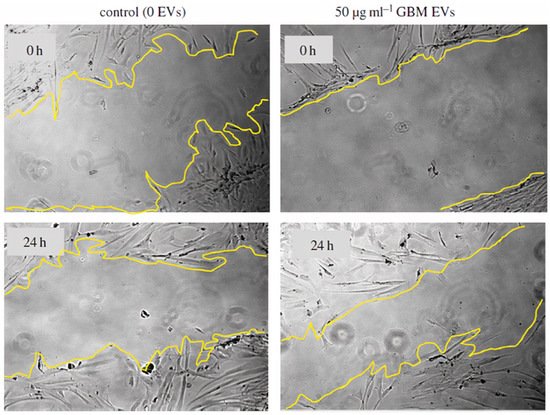
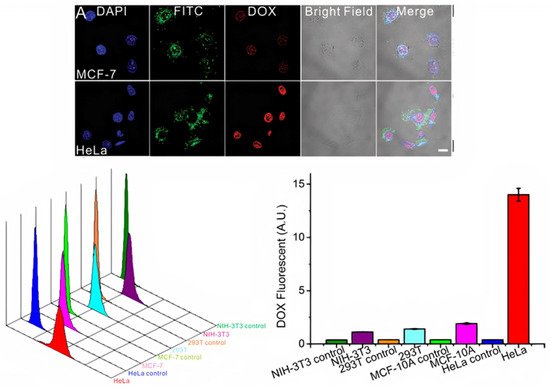
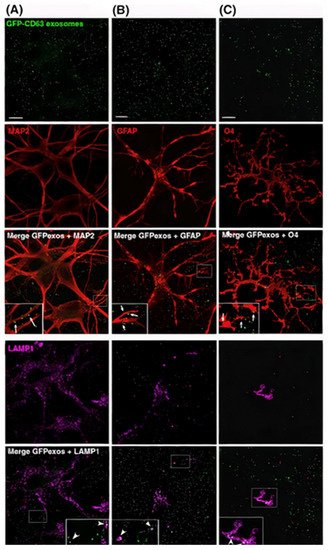
1000/1000
Hot
Most Recent

Exosomes are nanostructural vesicles with a size range of 30–150 nm that have gained increasing attention over the past few years. They have endocytic origin, the endocytic compartment membrane, the plasma membrane, and cytosol contain all exosomal proteins. Exosomes have not been found to contain proteins of the mitochondria, nucleus, endoplasmic reticulum, or Golgi apparatus.