1. Introduction
Diabetes and its effect on fetal health are significant to the developmental origins of health and disease (DOHaD) hypothesis. Globally, around 223 million women currently live with diabetes, 60 million of whom are of reproductive age
[1]. In addition to pre-existing diabetes, gestational diabetes mellitus (GDM), a form of maternal diabetes typically first diagnosed during weeks 24–28 of pregnancy, currently affects around 1 in 6 births worldwide, equating to approximately 16.8 million pregnancies
[1][2][3]. The diagnostic criteria for GDM vary widely in different countries and in turn have led to heterogeneity in screening and trial designs, making it difficult for comparative judgement and unified consensus on its effect on maternal and fetal health
[4]. If maternal hyperglycaemia is poorly controlled, this accelerates intrauterine growth and increases the risk of macrosomia, in which birth weight is > 4 kg, or the fetus being born large for gestational age (LGA), in which birth weight is above the 90th percentile. This may cause birth trauma for mother and baby by increasing the risk of preeclampsia, neonatal hypoglycaemia, shoulder dystocia, late stillbirth, or the need for caesarean section or neonatal intensive care
[5][6][7]. Although GDM ceases post-parturition, these women are predisposed to an estimated sevenfold increased risk of type 2 diabetes mellitus (T2DM) within 5–10 years post-pregnancy
[8][9][10]. Diabetes currently represents 10% of the National Health Service budget and, with the ever increasing prevalence of diabetic pregnancies, including diabetic risk factors such as the obesity epidemic and advanced maternal age
[11][12], it is now paramount to refine diagnostic and treatment strategies to improve outcomes for mother and baby.
Insulin therapy is a standard treatment for diabetes to restore glucose homeostasis; however, this therapy is associated with increased maternal weight gain and hypoglycaemia. As rates of diabetes continue to rise, the cost, storage, and administration requirements for insulin have proven to be of increasing concern
[13][14], particularly in developing countries where these storage requirements may not be feasible
[14]. As such, metformin has been advanced as an alternative first-line therapy for T2DM and GDM in many countries
[15][16].
2. Metformin in Pregnancy
Metformin is an oral synthetic guanidine analogue known as a ‘glucophage’ due to its glucose-lowering abilities by reducing gluconeogenesis and insulin resistance
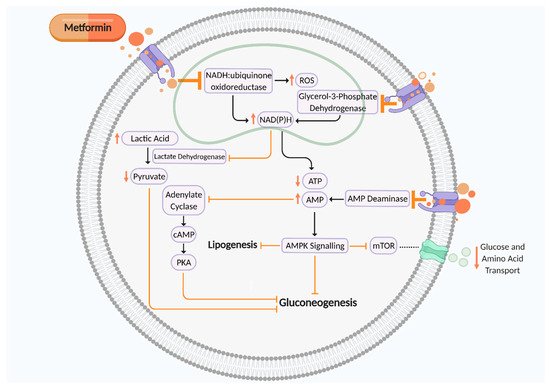
[15][16]. Metformin is a mitochondrial complex I (NADH:ubiquinone oxidoreductase) inhibitor which is transported into the cell to directly influence cellular respiration (Figure 1). Complex I (NADH:ubiquinone oxidoreductase) oxidises NADH synthesised from one carbon metabolism, glycolysis, fatty acid β-oxidation, and the tricarboxylic acid (TCA) cycle for adenosine triphosphate (ATP) production via the electron transport chain
[17][18][19]. Thus, metformin-induced suppression of complex I increases NADH accumulation and ROS production and reduces ATP synthesis, thereby elevating the AMP:ATP ratio. This activates AMP-activated protein kinase (AMPK) and leads to inhibition of gluconeogenesis, therefore maintaining glycaemic control
[17]. Metformin can also reduce gluconeogenesis by inhibiting AMP deaminase, which further contributes to elevated cellular AMP levels, thus in turn inhibiting adenylate cyclase and cAMP–PKA signalling. Metformin-induced suppression of mitochondrial glycerol 3 phosphate dehydrogenase (G3PDH) also augments cytosolic NAD(P)H concentration, leading to reduced pyruvate levels and a suppression of gluconeogenesis. However, activation of AMPK signalling inhibits mammalian target of rapamycin (mTOR) activity, a nutrient sensor which regulates amino acid transport and glucose storage
[17][20][21][22][23] (Figure 1). This mechanism of action leads to improved insulin sensitivity by augmenting insulin receptor tyrosine kinase activity, amplifying glycogenesis and suppressing glycogenolysis, inhibiting lipolysis, enhancing glucose transporter GLUT4 recruitment and activity, and suppressing the activity of hepatic glucose 6 phosphatase. Metformin also heightens insulin release due to enhanced glucagon-like peptide-1 (GLP-1) activity
[24].
Figure 1. Putative mechanism of action of metformin on cellular metabolism and mitochondrial aerobic respiration to suppress gluconeogenesis. Metformin is an inhibitor of mitochondrial complex I (NADH:ubiquinone oxidoreductase), AMP deaminase, and mitochondrial glycerol 3 phosphate dehydrogenase (G3PDH), which all contribute towards suppression of cellular gluconeogenesis to maintain glycaemic control. ROS, reactive oxygen species; NAD(P)H, nicotinamide adenine dinucleotide phosphate; ATP, adenosine triphosphate; AMP, adenosine monophosphate; cAMP, cyclic AMP; PKA, protein kinase A; mTOR, mammalian target of rapamycin. Black arrows indicate cellular pathway. Orange arrows indicate putative effects of metformin. Figure created using Biorender.com.
Metformin’s glucose-lowering activity certainly demonstrates beneficial outcomes for maternal health, as it decreases maternal weight gain, inflammation, atherothrombosis, and cardiovascular disease mortality, all of which are diabetic co-morbidities
[15][25]. However, studies suggest that its short- and long-term effects on the metabolic health of the offspring may not be as favourable.
Whilst metformin therapy has been shown to significantly reduce the incidence of LGA, it has been reported that it may decrease birth weight to the extreme as metformin use in pregnancy is associated with an increased rate of small for gestational age (SGA) births; that is, those with a birth weight below the 10th percentile or two standard deviations below the mean weight for gestational age
[5][7][26]. Notably, SGA offspring exposed to metformin in utero have shown signs of ‘catch-up growth’ during childhood. In the Metformin in Gestational Diabetes: The Offspring Follow Up (MiG: TOFU) study, at two years of age, metformin-exposed offspring demonstrated higher subcutaneous adiposity and larger mid-upper arm circumferences and bicep and subscapular skinfolds than insulin-exposed offspring
[27]. By nine years of age, they presented with significantly higher body mass index (BMI) and larger arm and waist circumferences, triceps skinfolds, and abdominal fat volumes compared to insulin-exposed offspring
[28]. A follow-up study of children exposed to metformin in utero in pregnancy complicated by polycystic ovarian syndrome also revealed they had higher BMIs at four years old than placebo-treated pregnancies
[29]. Another randomised controlled trial showed that infants exposed to metformin during GDM pregnancy were markedly heavier at 12 and 18 months of age compared to insulin-exposed infants
[30]. A murine study examining the effects of gestational metformin exposure from days E0.5 to E17.5 also showed that dams exposed to metformin manifested lower fetal weight on E18.5 than untreated dams. When fed a high-fat diet later in development, metformin-exposed fetuses were heavier than untreated fetuses and demonstrated increased mesenteric fat and liver weight. These findings, combined with gene set enrichment analysis of differentially expressed genes in the metformin and untreated murine offspring, reveal that metformin may induce transgenerational effects by way of fetal programming
[31]. Accordingly, these studies suggest that metformin therapy in pregnancy may increase the risk of childhood obesity and thus is likely to predispose offspring to cardiometabolic diseases during adulthood. It is therefore crucial to develop our understanding of metformin’s mechanistic activity and its effects on the balance between maternal health and adverse fetal outcomes.
Studies from the literature around the effects of metformin treatment on the placenta suggest that metformin alters placental gene expression and function (Table 1), although the mechanisms remain unclear.
Table 1. Current literature on the impact of metformin on placental gene expression and function
[32][33][34][35][36][37][38][39][40][41][42][43].
| Reference |
Model |
Effects Demonstrated by
Metformin |
Significance |
| Clinical studies |
Jamal et al. 2012
[32] |
Pregnant women with PCOS treated with metformin |
- ⇔ on birth weight
- ↓ uterine artery pulsatility index |
Metformin adversely affected uteroplacental circulation |
| Ex vivo or in vitro human placental studies |
Jiang et al. 2020
[33] |
Human GDM and T2DM placental explants cultured and treated with metformin (ex vivo) |
Male human placental explants:
- AMPK activation
- ↑ H3K27 acetylation
- ↓ DNMT1 protein abundance
- ↓ PGC-1α promoter methylation and ↑ PGC-1α mRNA expression |
Effects of metformin may be fetal sex-dependent
Metformin may improve placental efficiency by facilitating placental mitochondrial biogenesis |
Brownfoot et al. 2020
[34]
Cluver et al. 2019
[35]
Kaitu’u-Lino 2018
[36]
Brownfoot et al. 2016
[37] |
Human primary tissues exposed to metformin; placental explants, endothelial cells and placental villous explants, whole maternal vessels, maternal omental vessel explants (in vitro and ex vivo) |
- ↓ sFlt-1 and sEng secretion from primary endothelial cells, preterm preeclamptic placental villous explants and villous cytotrophoblast cells
- ↓ VCAM-1 mRNA expression in endothelial cells
- ↑ whole maternal blood vessel angiogenesis
- ↓ sFlt mRNA expression
- ↓ TNFα-mediated endothelial cell dysfunction |
Metformin enhances placental angiogenesis and reduces endothelial dysfunction by decreasing endothelial and trophoblastic antiangiogenic factor secretion via mitochondrial electron transport chain inhibition
Metformin is being trialled as a medication for preeclampsia (trial number PACTR201608001752102) |
Szukiewicz et al. 2018
[38] |
Human placental lobules perfused with metformin under normoglycemic or hyperglycaemic conditions
(ex vivo) |
- ↓ CX3CL1 and TNFα secretion
- ↑ placental CX3CR1 protein expression
- ↓ placental NFκB p65 protein |
Metformin has anti-inflammatory effects in the placenta |
Correia-Branco et al. 2018
[39] |
HTR-8/SVneo extravillous trophoblast cell line exposed to metformin
(in vitro) |
- ↓ proliferation
- ↑ apoptosis
- Inhibited folic acid uptake
- Inhibited glucose uptake
- Effects of metformin were prevented by inhibition of mTOR, JNK, and PI3K pathways |
Metformin impairs placental development and nutrient transport via PI3K, mTOR, JNK, and PI3K pathways |
Arshad et al. 2016
[40] |
Human placental explants; from healthy pregnancy, non-treated diet-controlled GDM pregnancy, and metformin-treated GDM pregnancy (ex vivo) |
- ↓ similar morphology in metformin-treated GDM placenta and non-treated healthy placenta, except for increased cord width
- ↓ placental width in metformin-treated GDM placenta compared to non-treated GDM placenta
- ↓ chorangiosis, placental thickness, and syncytial knots in metformin-treated placenta compared to non-treated GDM placenta |
Metformin may improve placental morphology by restoring diabetic placental hallmarks to characteristics similar to healthy placenta |
Han et al. 2015
[41] |
Human first trimester trophoblasts treated with or without metformin (in vitro) |
- ↓ trophoblast cytokine and chemokine release in normal and high glucose culture concentrations
- No antiangiogenic or antimigratory effects |
Metformin may potentially decrease placental glucose-induced inflammatory response |
| In vivo rodent studies |
Jiang et al. 2020
[33] |
Mice treated with maternal metformin and high-fat diet |
Improved placental efficiency in males:
- ↓ PGC-1α promoter methylation and ↑ PGC-1α expression
- ↑ TFAM expression
Improved glucose homeostasis in male offspring |
Metformin may improve placental efficiency by facilitating placental mitochondrial biogenesis
Metformin may be protective to the offspring by suppressing epigenetic changes evoked by maternal diabetes |
Wang et al. 2019
[42] |
Pregnant mice fed an isocaloric diet (control), high-fat diet, or high-fat diet plus metformin
(in vivo) |
- ↓ placental weight compared to control
- Partially rescued high-fat diet induced ↓ in placental and fetal weight
- ↑ VEGF and MMP-2 protein expression |
Metformin improves high fat diet-induced reduction in placental and fetal growth, potentially by modulating placental vasculature |
Alzamendi et al. 2012
[43] |
Pregnant rats fed a normal or high-fructose diet, treated with metformin
(in vivo) |
- ↓ fetal weight
- ⇔ on placental weight or blood vessel area
- Improved fructose diet induced ↓ blood vessel area |
Metformin reduces fetal weight in mice fed a normal diet
Metformin prevents high fructose diet-induced placental dysfunction |
Dark grey is table heading; pale grey titles demonstrate whether the study was clinical, ex-vivo or in vitro human placental, or in-vivo rodent studies. ⇔ no change; ↓reduction; ↑ increase. AMPK, AMP-activated protein kinase; DNMT, DNA methyltransferase; PGC-1α, peroxisome proliferator-activated receptor-gamma coactivator 1α; TFAM, mitochondrial transcription factor A; sFlt-1, soluble fms-like tyrosine kinase-1; sEng, soluble endoglin; VCAM-1, vascular cell adhesion molecule 1; TNFα, tumour necrosis factor alpha; VEGF, vascular endothelial growth factor; MMP-2, matrix metalloproteinase-2; NF-κB, nuclear factor kappa B; mTOR, mammalian target of rapamycin; JNK, c-Jun N-terminal kinase; PI3K, hosphatidylinositol-3-kinase.
2.1. Transplacental Transport of Metformin
As the interface between maternal and fetal circulations, the placenta transports nutrients to the developing fetus. There is also evidence that metformin is transported across the placenta to the fetal circulation. In metformin-exposed pregnancies, serum samples from umbilical cord, placental, and fetal tissues have demonstrated metformin concentrations to be equal or greater than maternal levels, suggesting active transport of metformin from the maternal circulation across the placenta and into fetal tissue
[44][45][46]. Metformin is a hydrophilic cation, has a half-life of 5 h, and is not metabolised in humans, but recent evidence suggests that metformin bioavailability, volume of distribution, and clearance may be significantly increased in pregnancy, dependent on dose
[47][48]. The mechanisms of how pregnancy alters metformin clearance remain to be established. Although metformin can cross the placenta, it is undetermined how metformin influences placental metabolism to influence gene expression and whether fetal tissues handle metformin in the same way.
The transporter responsible for metformin uptake from the maternal circulation into the placenta is yet to be determined and requires further research. Studies have reported norepinephrine transporter (NET), serotonin transporter (SERT), and organic cation transporter novel type 2 (OCTN2) to be localised on the maternal interface of the placenta at the syncytiotrophoblast apical membrane, which could be responsible
[19][49][50][51]. OCT3 has been demonstrated to be the key transporter for fetal metformin uptake and distribution, localised on the fetal interface of the placenta at the syncytiotrophoblast basal membrane and fetal capillaries. Indeed, OCT3-/- pregnant mice show attenuated fetal metformin exposure
[19]. However, it is apparent that placental OCT3 expression increases with gestational age, as a murine study demonstrated that placental OCT3 mRNA and protein expression increased by 37-fold and 56-fold, respectively, at day 15 of gestation, and by 46-fold and 128-fold, respectively, at day 19
[52]. Thus, it is possible that metformin may not be reaching fetal tissues with significant concentration until late gestation. Moreover, these findings suggest that, unlike insulin
[13], metformin can cross the placenta
[21][28] and reach fetal tissue, which could potentially influence fetal growth and programming.
2.2. Impact of Metformin on Placental Nutrient Transport and Nutrient Bioavailabilty
It has been demonstrated that metformin influences fetal growth and nutrient bioavailability by inhibiting mitochondrial complex I, leading to activated AMPK signalling and inhibition of placental mTOR signalling (Figure 1). Attenuated placental mTOR signalling is associated with restricted fetal growth
[53]. This mechanism of action may potentially explain the significant relationship between SGA births and metformin exposure in pregnancy. mTOR is highly expressed in the human placenta syncytiotrophoblast layer and mTOR complex 1 (mTORC1) signalling plays a major role in placental nutrient sensing, thus significantly influencing fetal nutrient availability and metabolism. Trophoblast mTORC1 regulates System A and System L amino acid transporters for amino acid uptake, essential for fetal metabolism
[54][55]. Preliminary in vitro models of human trophoblast cells with silenced mTORC1 have also demonstrated that placental mTORC1 may regulate a circulating factor or factors, which could influence fetal growth
[55][56]. mTORC1 signalling is regulated by placental insulin and IGF I, and fetal glucose, amino acid, and oxygen levels. Diabetes may increase mTORC1 activation due to elevated maternal nutrient and ATP concentrations
[54][55]. During early gestation, the embryo mainly expresses immature mitochondria. As gestation develops, the placenta and fetus increase their expression of mature mitochondria, which are more susceptible to metformin inhibition. With this in mind, it is possible metformin may not adversely affect offspring growth until after the first trimester
[21].
Transplacental metformin exposure may restrict placental and fetal growth by reducing nutrient bioavailability which could influence fetal programming. Evidence suggests metformin can influence the status of several vitamins and micronutrients, including vitamins B
1, B
12, and D, folic acid, and magnesium
[15][20][57][58][59][60][61][62]. Whilst all of these nutrients are important for fetal growth and development, folate and vitamin B
12 are co-factors of one carbon metabolism, essential for cell growth, metabolism, and production of the methyl donor S-adenosyl-methionine (SAM). Furthermore, exposure to metformin and maternal deficiency of both folate and vitamin B
12, during pregnancy, lead to similar changes to placental and fetal growth and offspring health
[63][64][65][66][67]. We therefore postulated that a potential mechanism by which metformin influences placental and fetal growth and offspring risk of cardiometabolic complications is by affecting the balance between folate and vitamin B
12 levels and, therefore, perturbing one carbon metabolism.