CRS is a chronic inflammatory disease that occurs in the nasal cavity and sinuses and affects 12% of the global population
[22]. The phenotypic classification of CRS is mainly based on the presence or absence of nasal polyps, which can be divided into CRS with nasal polyps (CRSwNP) or CRS without nasal polyps (CRSsNP)
[23]. In contrast to the phenotype, the endotypic classification of CRS mainly represents an individual’s inflammatory mechanisms, rather than a clear entity with a direct biological basis. In a very meaningful study, researchers used 14 different inflammatory markers for hierarchical cluster analysis to determine the putative inflammatory endotype of CRS, and identified ten different clusters, including eosinophils and T helper-(Th) 2 related markers such as interleukin (IL)-5 and immunoglobulin E (IgE), neutrophils, or proinflammatory mediators such as IL-1β, IL-6, IL-8, and myeloperoxidase; Th17/Th22 markers such as IL-17A, IL-22, and tumor necrosis factor-α (TNF-α); and interferon-γ (IFN-γ)
[24]. In recent years, a deeper understanding of the role of microbiota in the human immune system evolved; various inflammatory diseases, such as CRS, were reported to be associated with a significant shift in host microbiota from a healthy state to a diseased state
[25]. Compared with that on other disorders such as asthma, research concerning microbiota and nasal diseases is still in its infancy, and causal relationships involving the existence of microbial communities and the development of CRS cannot be readily explained
[26].
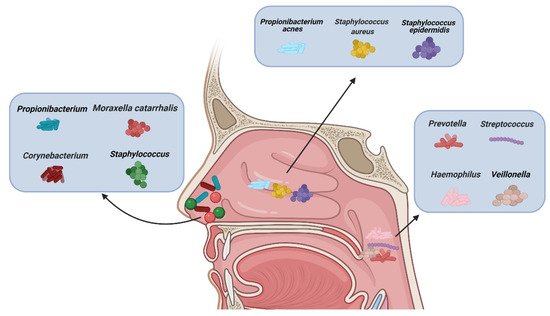
There are different types of microbiota in the upper airway of healthy adults (). The nasal cavity is directly connected to the external environment. Through inhalation, the nasal cavity can directly contact various microbiota, fungal spores, and pollutants
[27]. The microbiota of healthy adults’ anterior nares is mainly composed of
Actinobacteria,
Firmicutes, and
Proteobactera [28]. Researchers examined the anterior nares of 236 healthy adults using nasal swabs and concluded that
Staphylococcus,
Propionibacterium,
Corynebacterium, and
Moraxella were the most common microbiota in their anterior nares
[29]. One study concluded that tissue samples were more suitable for assessing microbiological groups in CRS patients than nasal swabs, because they observed significant differences in the microbiota groups in the nasal swabs, while the differences observed in the tissue samples were smaller
[30]. However, there are some discrepancies about the usefulness of the two methods.
[31]. Other data showed that a tissue biopsy cannot provide additional information compared with multiple swab tests. In more than 90% of their cases, swabs from multiple sites provide comprehensive information about patients’ culturable pathogens. In the middle meatus of healthy adults, the most abundant microbiota were
Staphylococcus aureus (
S. aureus),
Staphylococcus epidermidis, and
Propionibacterium acnes [32]. Using next-generation 454 pyrosequencing of the 16S rRNA gene, Jetté et al. found that
Streptococcus,
Prevotella,
Veillonella, and
Haemophilus were the most common microbiota in the throats of 97 adults
[33]. The common microbiota in CRS patients vary with geographical location. According to data reported,
Cyanobacteria are the dominant phylum in CRS in Missouri, USA, and that the change in microbiota composition between the control group and CRS group is minor
[34]; in contrast, other data show that the abundance of
Verrucomicrobia and
Bacteroides is low and that of
Actinobacteria is high in Colorado, USA
[35]. A Korean study compared a CRS group with a control group and found that the abundance of
Bacteroides in the CRS group was low and that of
Fusobacteria was high
[36].