1000/1000
Hot
Most Recent

Use of productivity-enhancing technologies (PET: growth hormones, ionophores, and beta-adrenergic agonists) to improve productivity has recently garnered public attention regarding environmentally sustainability, animal welfare, and human health.
|
Class a |
Mode of Action |
Substance b |
Mode of Administration |
|---|---|---|---|
|
Growth hormones |
|||
|
Endogenous/Synthetic |
Increase protein deposition at the expense of fat to increase growth rate and decrease amount of feed required for the animal to gain weight. |
Estradiol-17β, Testosterone, Progesterone/Zearalenone, Trenbolone acetate |
Implants |
|
Melengestrol acetate |
In-Feed |
||
|
Beta-adrenergic agonists |
Redirect nutrients from digestive organs into muscle tissue, thus increasing muscle mass accretion at the expense of fat deposition. |
Ractopamine chloride, Zilpaterol chloride |
In-Feed |
|
Antibiotics c |
|||
|
Ionophores |
Act against Gram-positive bacteria by altering membrane permeability to promote propionate formation in the rumen, which is more energetically favorable than acetate production. |
Monensin, Lasalocid, Salinonmycin |
In-Feed |
|
Macrolides |
Has bacteriostatic effect on both Gram-positive and Gram-negative bacteria, thus reducing microbial competition for nutrients. |
Tylosin, Neomycin |
In-Feed, water, or parenteral |
|
Aminoglycosides |
|||
|
Tetracyclines |
Oxytetracycline, Chlortetracyclic |
a Used in growth promotion by beef producing countries, including countries in North America (US, Canada, Mexico), Australian–New Zealand region, South America (Brazil and Argentina), and Africa (South Africa). Approval of specific products depends on the regulatory framework within each country. b Synthetic derivatives of estrogen, testosterone, and progesterone are zearalenone, trenbolone acetate, and melengesterol acetate, respectively. c Globally not recommended for feed efficiency, except ionophores. However, implementation is subject to local and national legislation or regulation.
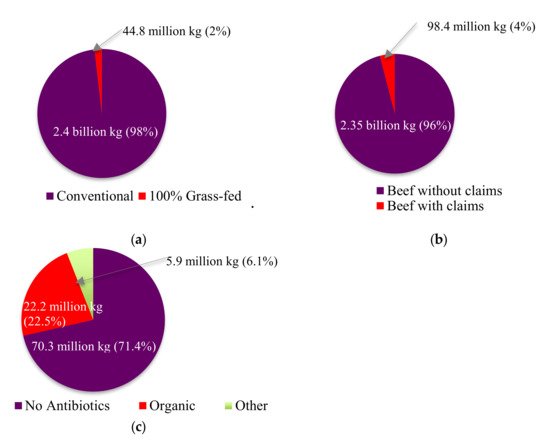
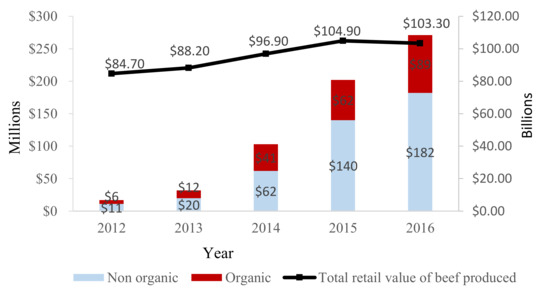
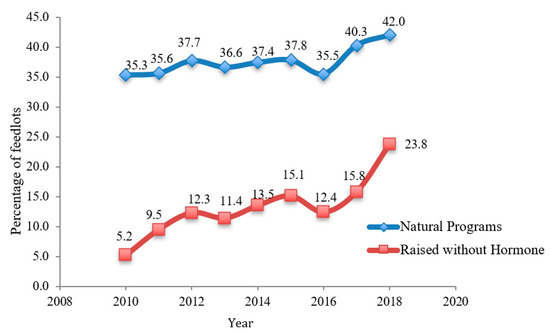
|
Reference |
Summary of Trial Design |
Environmental Indices e,f |
Country |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Methodology a |
Production Stage b |
Treatment c |
Days on Feed |
CO2eq |
Land |
Water |
Energy |
NH3 /Manure Excretion |
||
|
Basarab et al. [49] |
LCA |
Backgrounding and finishing phases |
IMP or control |
Backgrounding: 312 days. Finishing: 146 to 207 days. |
5.8% ↓ |
7.8% ↓ |
NR |
NR |
NR |
Canada |
|
Capper [50] |
LCA |
Backgrounding and finishing phases |
βAA + IMP + MGA + ION (“conventional”); and no additives (“grass-fed” or “natural” animals). |
Backgrounding: 123 to 159 days. Finishing: 110 to 313 days. |
14.8–40.3% ↓ |
18.3–44.7% ↓ |
17.9–75.2% ↓ |
14.9–28.6% ↓ |
17.9–50.5% ↓ N and 20.7–51.4% ↓ P excretions |
US |
|
Capper and Hayes [51] |
LCA |
Backgrounding and finishing phases |
βAA + IMP + ION + MGA; or control. |
Backgrounding: 148 to 159 days. Finishing: 116 to 209 days. |
8.9% ↓ |
9.1% ↓ |
4.0%↓ |
7.1% ↓ |
8.9% and 9.6%, ↓ N and P excretions, respectively. |
US |
|
Cooprider et al. [52] |
Animal trial |
Finishing phase |
βAA + IMP + ION; or control. |
146 to 188 days. |
31.4% ↓ non-CO2 emissions |
NR |
NR |
NR |
NR |
US |
|
Stackhouse et al. [53] |
LCA |
Backgrounding and finishing phases |
IMP + ION only; βAA + IMP + ION; or control. |
Backgrounding: 182 days. Finishing: 121 to 212 days. |
6.6–8.0% ↓ |
NR |
NR |
NR |
7.7–13.5% ↓ NH3 emissions. |
US |
|
Stackhouse-Lawson et al. [54] |
Animal trial |
Finishing phase |
ION only; IMP + ION only; βAA + IMP + ION; or control |
107 days. |
9.6–16.4% ↓CH4 emissions |
NR |
NR |
NR |
30% ↓ NH3 emissions |
US |
|
Webb [55] |
Animal trial and LCA |
Cow–calf, backgrounding, and finishing phases |
ION only; IMP + ION only; βAA + IMP + ION; or control. |
Backgrounding: 91 days, Finishing: 152 to 183 days |
1.1–7.7% ↓ |
NR |
1.0–5.8% ↓ |
1.1–5.5% ↓ |
0.7–5.1% ↓ reactive N |
US |
a Type of study conducted: LCA = Life cycle assessment, with PETs administered during backgrounding and finishing phases only, except Webb [55], who included implanted pre-weaned calves during the cow–calf phase; Animal trial = a study that used steers at the finishing phase. b Assumes a production system comprised of three distinct phases: cow–calf, backgrounding, and finishing. Grain-based diet during finishing phase except where indicated. c IMP = Implants (trenbolone acetate, estradiol, zearalenone); MGA= melengestrol acetate; ION = Ionophores (Monensin); βAA = Beta-adrenergic agonist (zilpaterol chloride and ractopamine chloride). d ADG = average daily gain; G:F = gain:feed. In Stackhouse et al. [53] and Webb [55], linear growth was assumed during the backgrounding phase; and during the finishing phase, ADG was adjusted when days on feed were extended as a consequence of lower feed quality and availability, which were assumed to limit growth. e Where ↓= decrease, ↑ = increase, and NR= not recorded; In all studies, the production indices and environmental parameters for all PET treatments were compared with control (no additives); however, in Capper [50], “conventional” animals (administered PETs) were compared with “natural” or “grass-fed” animals (no PETs administered for either). f Environmental indices were expressed on an intensity basis (per kg of beef); CO2eq = carbon dioxide equivalent; CH4 = methane; NH3 = ammonia; N = Nitrogen; and P = Phosphorus. g The total number of cattle considered under “grass-fed” was 12,510,000 and for “natural” was 8,257,000 animals. h The total number of cattle in the production system without PETs was 3,651,000 animals.