1000/1000
Hot
Most Recent

Volumetric muscle loss (VML) is the massive wasting of skeletal muscle tissue due to traumatic events or surgical ablation. This pathological condition exceeds the physiological healing process carried out by the muscle itself, which owns remarkable capacity to restore damages but only when limited in dimensions. Upon VML occurring, the affected area is severely compromised, heavily influencing the affected person’s quality of life. Overall, this condition is often associated with chronic disability, which makes the return to duty of highly specialized professional figures (e.g., military personnel or athletes) almost impossible. The actual treatment for VML is based on surgical conservative treatment followed by physical exercise; nevertheless, the results, in terms of either lost mass and/or functionality recovery, are still poor. On the other hand, the efforts of the scientific community are focusing on reconstructive therapy aiming at muscular tissue void volume replenishment by exploiting biomimetic matrix or artificial tissue implantation. Reconstructing strategies represent a valid option to build new muscular tissue not only to recover damaged muscles, but also to better socket prosthesis in terms of anchorage surfaces and reinnervation substrates for reconstructed mass.
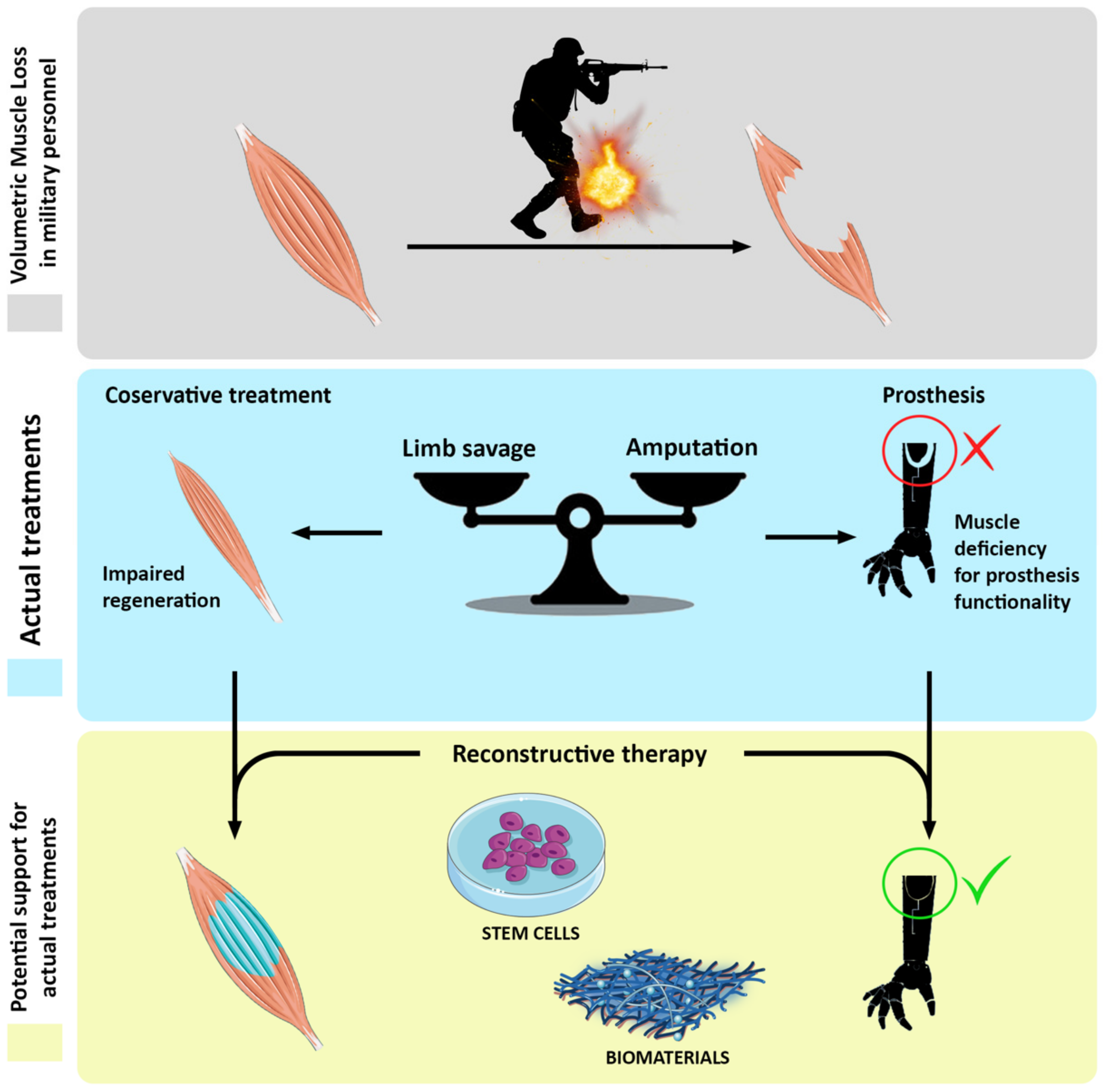 Figure 1. Schematic representation of cell-based reconstructive approach to volumetric muscle loss (VML) recovery.
Figure 1. Schematic representation of cell-based reconstructive approach to volumetric muscle loss (VML) recovery.